Blackcurrant extract promotes differentiation of MC3T3‑E1 pre‑osteoblasts
- Authors:
- Published online on: June 19, 2024 https://doi.org/10.3892/br.2024.1810
- Article Number: 121
-
Copyright: © Nanashima et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Blackcurrant (Ribes nigrum L.) contains polyphenols, particularly four anthocyanins: Cyanidin-3-glucoside, cyanidin-3-rutinoside, delphinidin-3-glucoside and delphinidin-3-rutinoside. Additionally, it contains large amounts of vitamins A, C and E, as well as small amounts of each of the B vitamins. Moreover, it contains abundant minerals, such as calcium, iron, magnesium, phosphorus, potassium and zinc (1). These compounds elicit beneficial health effects, including increased blood flow, cancer suppression and prevention of glaucoma, eye strain and lifestyle-related diseases, such as obesity and diabetes mellitus (2,3).
Phytoestrogens are plant-derived substances that exhibit effects similar to those of endogenous estrogens. Multiple phytoestrogens, including isoflavones and resveratrol, have been identified (4,5). Decreased estrogen secretion associated with increased age and menopause increases risk of developing disorders, including decreased blood vessel function, dyslipidemia and osteoporosis (6-8). Our previous studies showed that blackcurrant extract (BCE) and its anthocyanins exert phytoestrogenic activity via signaling through estrogen receptors α and β (9,10) and alleviate some menopausal symptoms, including arteriosclerosis, hair loss, skin aging and dyslipidemia (11-14).
Bone is remodeled by constantly being resorbed and formed by osteoblasts. Notably, imbalance between bone resorption and formation causes a decrease in bone density, leading to osteoporosis (15). Moreover, bone remodeling is a complex process involving several hormones, including estrogen (16,17). Osteoporosis refers to a condition in which bone mass decreases and bone structure deteriorates, weakening bone strength and increasing susceptibility to fractures (18,19). Considering that estrogen regulates bone metabolism, osteoporosis is more likely to occur following menopause-associated decrease in estrogen, a condition called postmenopausal osteoporosis (20).
During early stage osteoporosis, patients present almost no symptoms, being difficult to diagnose this condition. Thus, it is important to consume foods rich in calcium, such as fish, dairy products and seaweed during menopause. The intake of phytoestrogens, such as equol, which is produced by metabolism of soy isoflavones contained in soybeans and soybean foods by intestinal bacteria, is effective in preventing postmenopausal osteoporosis (21,22). Blackcurrant has also been shown to reduce the risk of osteoporosis and improve trabecular bone mass in young mice (23,24). Additionally, blackcurrant alleviates osteoporosis in humans, and clinical trials are currently ongoing to validate its efficacy (25).
Estrogen acts on both osteoclasts and osteoblasts; its deficiency in menopause is hypothesized to accelerate bone resorption by osteoclasts and decrease bone mass (26). Although blackcurrant has been shown to inhibit osteoclastogenesis, studies on its effects on osteogenesis are lacking (27). Additionally, the mechanism by which blackcurrant alleviates osteoporosis remains unclear. As osteoblasts are sensitive to estrogen, the present study aimed to investigate the phytoestrogenic effects of BCE on osteoblast proliferation and differentiation using mouse pre-osteoblastic MC3T3-E1 cells. Notably, these cells produce large amounts of collagen, differentiate into osteoblasts and ultimately form bone (28-30).
In osteoblasts, expression of differentiation markers, such as collagen type I (Col-I), alkaline phosphatase (Alp), bone γ-carboxyglutamate protein (Bglap) and runt-related transcription factor 2 (Runx2), increase depending on the extent of differentiation (31). Osteoblasts in the late stage of differentiation produce mineralized deposits (calcified nodules) that can be stained with Alizarin Red (32,33). To the best of our knowledge, the present study is the first to assess the health effects of BCE on osteoblast differentiation.
Materials and methods
Reagents and cell culture
BCE powdered extract was CaNZac-35 (Koyo Mercantile Co., Ltd.), containing a high concentration of polyphenols and anthocyanins (37.6 and 38.0% w/w, respectively) (10). 17β-estradiol (E2) was purchased from Sigma-Aldrich (Merck KGaA). The mouse pre-osteoblast cell line MC3T3-E1 was obtained from the Health Science Research Resources Bank (Osaka, Japan). MC3T3-E1 cells were maintained in α-MEM (FUJIFILM Wako Pure Chemical Corporation) supplemented with 10% (v/v) FBS (Sigma-Aldrich; Merck KGaA), 100 U/ml penicillin and 100 µg/ml streptomycin (FUJIFILM Wako Pure Chemical Corporation). Cell culture experiments were conducted at 37˚C in a humidified incubator under 5% CO2.
Cell treatment
MC3T3-E1 cells (1x104 cells/well) were seeded in six replicates in 96-well plates and cultured overnight in α-MEM supplemented with 10% (v/v) FBS. The medium was replaced with phenol red-free α-MEM supplemented with 5% (v/v) charcoal-stripped FBS (Thermo Fisher Scientific, Inc.). Cells were cultured for 48 h at 37˚C in the presence or absence of 0.2, 1.0, 5.0 or 20.0 µg/ml BCE or 10 nM E2. E2 was used as a positive control to examine the phytoestrogenic effect of BCE on MC3T3-E1 cells (32). Treatment duration and the E2 concentration of 10 nM were selected based on previous studies (29,33). The morphology of MC3T3-E1 cells was analyzed under a phase-contrast microscope (CK40; Olympus Corporation; magnification, x100) with an Anyty™ digital microscope camera (3R-DKMCO4; Three R Solution Corp. Japan).
Proliferation assay
Quantification of cell proliferation was performed using Cell Counting Kit-8 (CCK-8; Dojindo Laboratories, Inc.) according to the manufacturer's instructions. CCK-8 solution was added to the wells, and incubated for 30 min at 37˚C. Absorbance was measured at 450 nm using a Benchmark microplate reader (Bio-Rad Laboratories, Inc.).
Alp assay
MC3T3-E1 cells (5x104 cells/well) were seeded in duplicate in 24-well plates and cultured overnight as aforementioned. The growth medium was replaced with phenol red-free α-MEM, supplemented with 5% (v/v) charcoal-stripped FBS. The cells were cultured for 72 h with BCE or 10 nM E2 as aforementioned, based on previous studies (30,34). After washing with 0.1 M Tris-HCl buffer (pH, 9.8), 100 µl 0.1% (v/v) Triton X-100 in 0.1 M Tris-HCl buffer (pH, 9.8) was added to the medium and the plates were stored at -80˚C. The plates were rapidly thawed at 37˚C and assayed using the LabAssay™ ALP kit (FUJIFILM Wako Pure Chemical Corporation). Protein concentrations were determined using a Takara BCA Protein Assay kit (Takara Bio, Inc.). Absorbance relative to Alp activity and protein concentrations were measured at wavelengths of 405 and 570 nm, respectively, using a Benchmark microplate reader (Bio-Rad Laboratories, Inc.).
Quantification of total collagen
MC3T3-E1 cells (1x105 cells/well) were seeded in duplicate in 12-well plates and cultured overnight in α-MEM supplemented with 10% (v/v) FBS. The growth medium was replaced with differentiation medium [phenol red-free α-MEM supplemented with 5% (v/v) charcoal-stripped FBS and Osteoblast-Inducer Reagent (Takara Bio, Inc.)], according to the manufacturer's protocol. Cells were cultured in the presence or absence of BCE or 10 nM E2, as aforementioned, and the medium was replaced every 3 days. Collagen staining was performed on day 14 using a Sirius Red/Fast Green Collagen Staining kit (Iwai Chemicals Co. Ltd.), according to the manufacturer's instructions. Cells were washed with PBS, followed by the addition of 0.5 ml Kahle fixative at 22˚C for 10 min. Dye solution was added to culture plates and incubated at 22˚C for 30 min. Cells were rinsed with 0.5 ml distilled water until the solution was colorless. Digital images were acquired using a phase-contrast microscope (CK40; magnification x40) with an Anyty™ digital microscope camera. Following microscopy, the dye was eluted. The optical density (OD) of the eluted dye solution was measured at 540 and 605 nm using a spectrophotometer (U-5100; Hitachi High-Technologies Corporation). The calculation formulas followed the kit manufacturer's instructions: Collagen (µg/section)=OD540-(OD605 x 0.291)/0.0378; non-collagen protein (µg/section)=OD605/0.00204.
Reverse transcription-quantitative PCR (RT-qPCR)
MC3T3-E1 (2x105 cells/well) were seeded in 6-well plates and cultured overnight as aforementioned until 80% confluent. The medium was replaced with phenol red- and serum-free α-MEM with or without BCE or 10 nM E2, as aforementioned. After incubating at 37˚C for 24 h, cells were washed twice with PBS. Total RNA was extracted using the RNeasy Mini kit (Qiagen GmbH), according to the manufacturer's instructions. RNA was reverse-transcribed into cDNA using PrimeScript RT Master Mix (Takara Bio, Inc.), according to the manufacturer's protocol. Col-I, Alp, Bglap and Runx2 mRNA expression levels were quantified via RT-qPCR using TB Green Premix Ex Taq II (Tli RNaseH Plus; Takara Bio, Inc.). Thermocycling conditions were as follows: 30 sec at 95˚C, followed by 40 cycles of 5 sec at 95˚C and 30 sec at 60˚C. Transcription levels were normalized to β-actin (Actb). Primers (5'→3') were as follows: Col-I forward, GAGCGGAGTACTGGATCG and reverse, GCTTCTTTTCCTTGGGGTT (31); Alp forward, GATCATTCCCACGTTTTCACATT and reverse, TTCACCGTCCACCACCTTGT (31); Bglap forward, GCGCTCTGTCTCTCTGACCT and reverse, AAGCAGGGTCAAGCTCACAT (31); Runx2 forward, AGCGGCAGAATGGATGAGTC and reverse, ACCAGACAACACCTTTGACG (35) and Actb forward, CATCCGTAAAGACCTCTATGCCAAC and reverse, ATGGAGCCACCGATCCACA (36). PCR specificity was determined using melting curve analysis. All samples were analyzed in duplicate. Relative gene expression was calculated using the 2-ΔΔCq method (37).
Mineralization assessment
Calcified nodule formation was assessed using Calcification Evaluation set (PG Research), according to the manufacturer's instructions. Briefly, 1x105 cells/well were seeded in duplicate in 12-well plates containing differentiation medium (Osteoblast-Inducer Reagent). Cells were treated as aforementioned with BCE or 10 nM E2 on day 21 and the medium was replaced every 3 days. At the end of treatment, medium was removed and wells were washed with PBS. Cells were fixed with neutral buffered 10% formalin at 22˚C for 10 min, followed by washing with distilled water. The cells were stained with 1.0 ml Alizarin Red solution at 22˚C for 30 min. Micrographs were acquired using a fluorescence microscope (FSX100; Olympus Corporation; magnification, x40). After removing the water, 0.5 ml calcified nodule lysate [5% (v/v) formic acid] was added and the plates were stirred for 10 min at 22˚C to elute the dye. The absorbance of the eluate was measured at 450 nm using a spectrophotometer (U-5100).
Animals and treatments
Ovariectomized (OVX) and sham-operated female Sprague-Dawley rats (OVX, n=6; sham-operated rats, n=6; age; weight, ~240 g; 12 weeks; CLEA Japan, Inc.) were housed in plastic cages in air-conditioned rooms (23˚C; 50% humidity) under a 12/12-h light/dark cycle at the Institute for Animal Experiments of Hirosaki University Graduate School of Medicine (Hirosaki, Japan). All experimental procedures were approved by the Animal Research Committee of Hirosaki University (approval no. G18003). Our previous study showed that 3% BCE has phytoestrogenic effects in rats (10). All rats were fed AIN-93M diet, with or without 3% BCE (Oriental Yeast Co., Ltd.) and were divided into three groups (n=3/group): Sham, OVX and OVX + 3% BCE. All rats had free access to food and water. After 3 months, the animals were euthanized by anesthesia with isoflurane (induction, 5%; maintenance, 3%), followed by decapitation, then femurs were excised and fixed in 10% (v/v) formaldehyde at 22˚C for 1 week, demineralized in 10% (v/v) EDTA-2Na (pH, 7.2) at 4˚C for 3 weeks and embedded in paraffin. Femur sections (4 µm) were routinely passed through xylene and descending ethanol series before Alp staining.
Alp staining
To measure femoral Alp activity, femurs were stained with a TRAP/ALP kit (FUJIFILM Wako Pure Chemical Corporation), according to the manufacturer's instructions. Nuclei were stained with hematoxylin at 22˚C for 5 min. Specimens were examined and photographed using a fluorescence microscope (FSX100; Olympus Corporation; magnification, x40).
Statistical analysis
All data are expressed as mean ± SD from four independent experiments. Graphs were generated using GraphPad Prism (version 7.03; Dotmatics). Significant differences were determined using Kruskal-Wallis analysis and Steel post hoc test via Bell Curve in Excel software (version 3.2; Social Survey Research Information Co., Ltd.). P<0.05 was considered to indicate a statistically significant difference.
Results
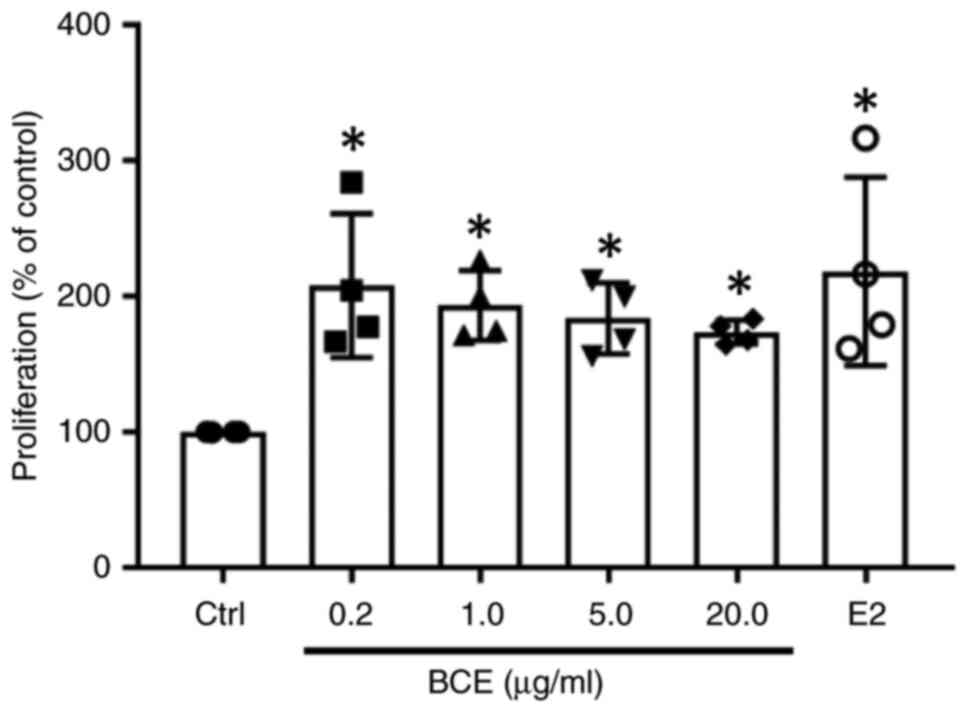
BCE promotes MC3T3-E1 cell proliferation
The present study investigated the effect of BCE on MC3T3-E1 cell proliferation. Cells were treated with 0.2, 1.0, 5.0 or 20.0 µg/ml BCE, which showed phytoestrogenic effects in our previous studies (12,13) or 10 nM E2 as a positive control. Cell proliferation increased significantly (P<0.05) after treatment with all concentrations of BCE or 10 nM E2 (Fig. 1). However, there was no notable difference in cell morphology following treatment with BCE or E2 (Fig. S1).
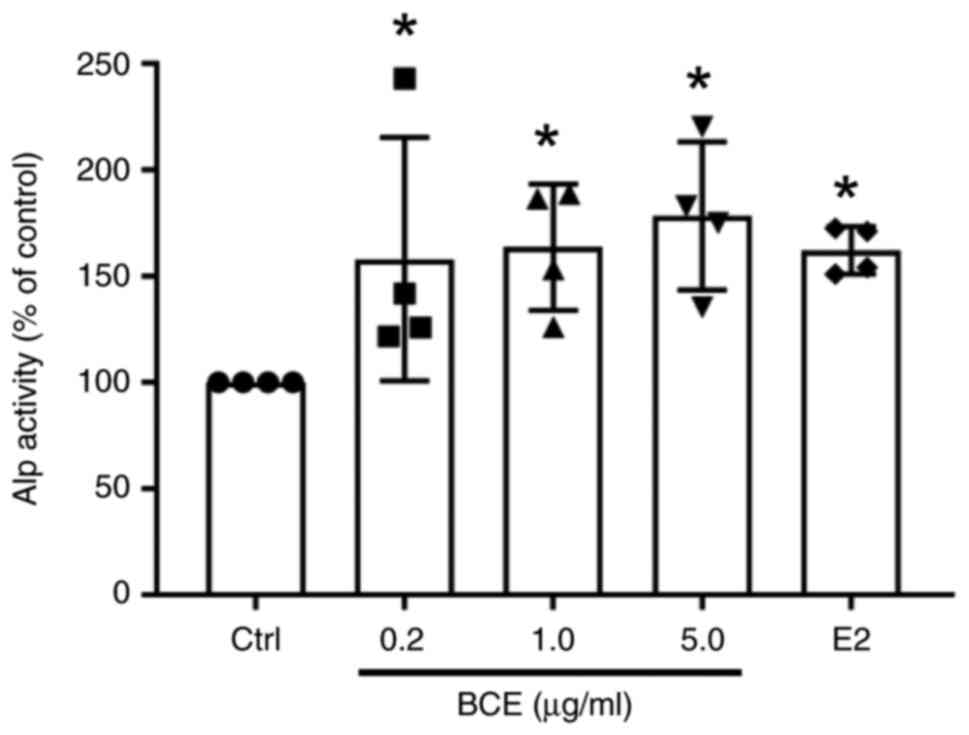
BCE enhances Alp activity in MC3T3-E1 cells
MC3T3-E1 cells were treated with 0.2, 1.0 or 5.0 µg/ml BCE or 10 nM E2 and cultured for 72 h. Alp activity increased significantly (P<0.05) in a dose-dependent manner following treatment with BCE and increased significantly (P<0.05) upon treatment with E2 (Fig. 2).
BCE enhances total collagen production in MC3T3-E1 cells
MC3T3-E1 cells were treated with 0.2, 1.0 or 5.0 µg/ml BCE or 10 nM E2 and cultured for 14 days. Total collagen was quantified using Sirius Red staining. Total collagen increased significantly (P<0.05) after treatment with all concentrations of BCE and 10 nM E2 (Fig. 3).
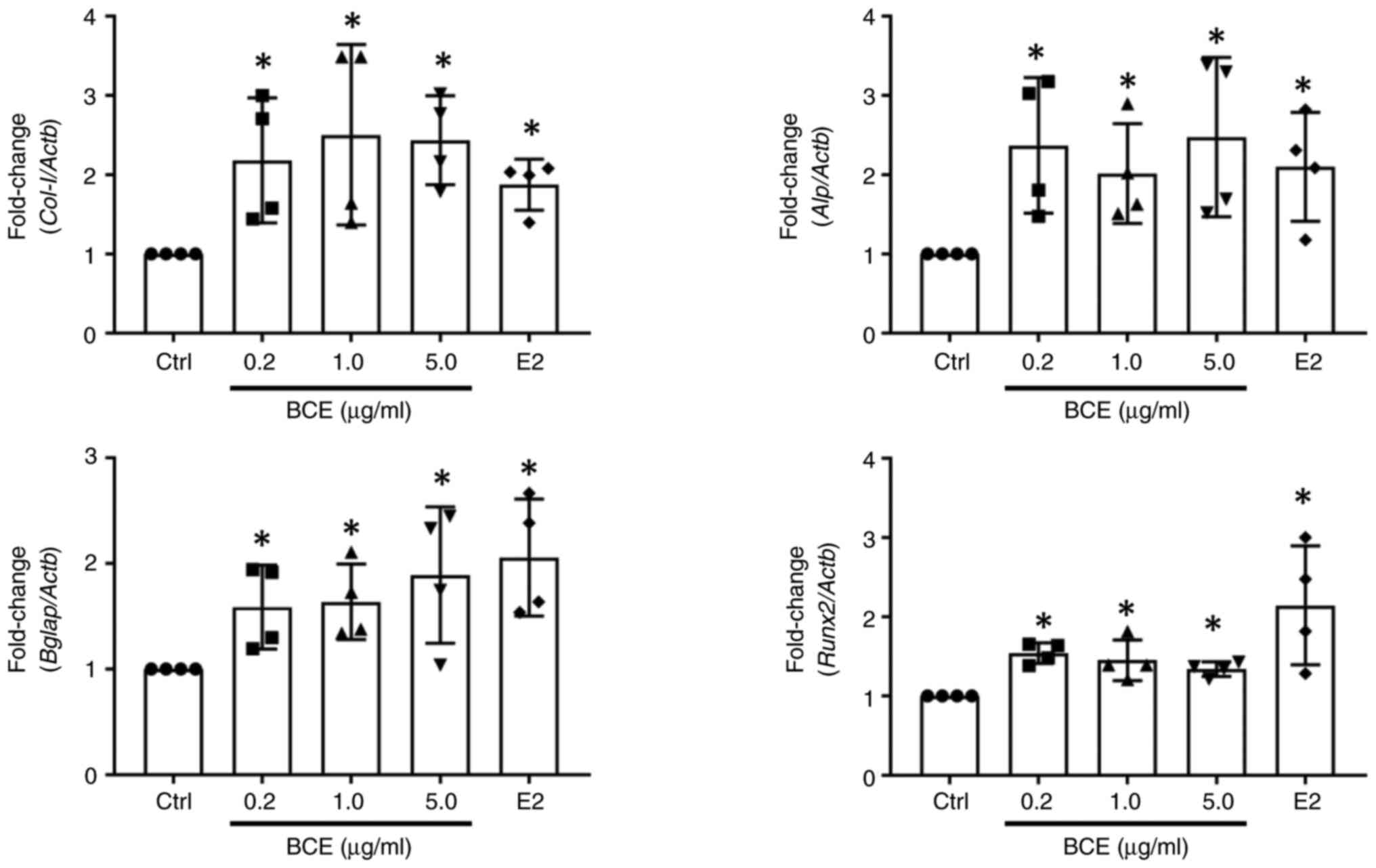
BCE upregulates expression of osteogenic markers in MC3T3-E1 cells
MC3T3-E1 cells were incubated with 0.2, 1.0 or 5.0 µg/ml BCE or 10 nM E2 for 24 h. Bone differentiation marker expression was measured using RT-qPCR. Col-I, Alp, Bglap and Runx2 expression increased significantly (P<0.05) following treatment with all concentrations of BCE and 10 nM E2 (Fig. 4).
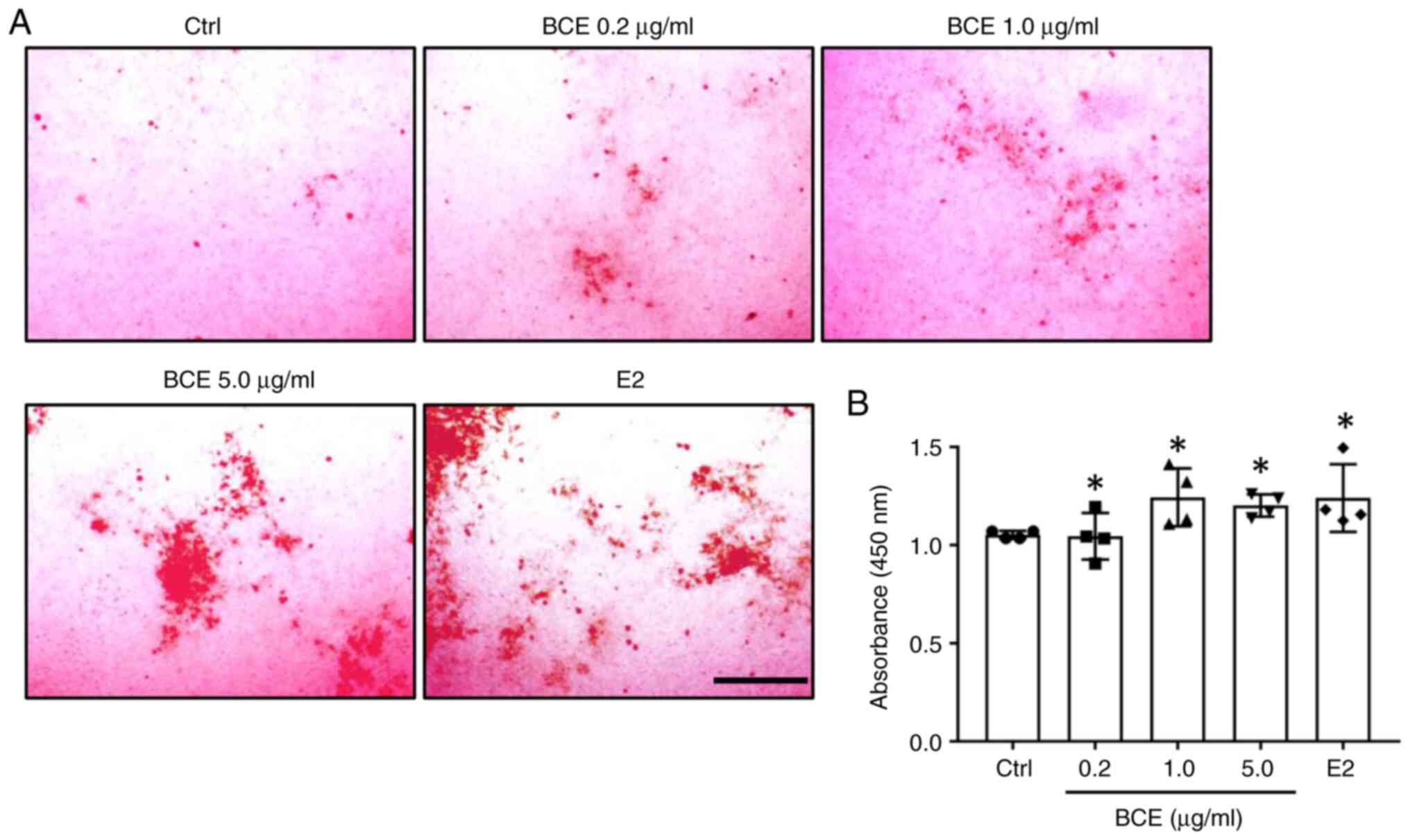
BCE promotes mineralization in MC3T3-E1 cells
MC3T3-E1 cells were treated with 0.2, 1.0 or 5.0 µg/ml BCE or 10 nM E2 and cultured for 21 days. Cells were stained with Alizarin Red to assess the extent of mineralization, which revealed the presence of calcified nodules (Fig. 5A). Levels of calcified nodules increased significantly (P<0.05) following treatment with all concentrations of BCE and 10 nM E2 (Fig. 5B).
BCE induces osteoblast differentiation in vivo
We investigated whether BCE induces osteoblast differentiation in the femoral tissue of OVX rats, which were used as a menopausal model. As Alp activity increases in the region surrounding the epiphysis, where active osteoblast differentiation occurs, rat femurs were stained to assess Alp activity. Compared with that in the Sham group, Alp staining intensity was weak in the epiphyses of rats in the OVX group. However, Alp staining intensity was stronger in the epiphyses of OVX rats treated with 3% BCE than in those of untreated OVX rats (Fig. S2, arrows).
Discussion
BCE inhibits osteoclast differentiation (27); however, its effects on osteoblasts are unknown. Therefore, the present study focused on the effect of BCE on differentiation of MC3T3-E1 pre-osteoblasts into osteoblasts.
Administration of phytoestrogens or 10 nM E2 significantly enhances MC3T3-E1 cell proliferation (28,38). Here, BCE or E2 treatment increased the proliferation of MC3T3-E1 cells, which was consistent with previous findings (28,38). Overall, the increase in MC3T3-E1 cell proliferation may be due to the phytoestrogen activity of BCE.
Estrogen activity has been shown to increase Alp activity and collagen secretion in MC3T3-E1 cells (31). In the present study, BCE increased Alp activity and collagen secretion in MC3T3-E1 cells in a concentration-dependent manner. Col-I, Alp, Bglap and Runx2 are known osteoblast differentiation markers (31). Here, BCE and E2 increased the expression of all four genes, which may be due to phytoestrogen activity.
Consistent with the present findings, several studies have shown that anthocyanin induces osteoblast differentiation, with increased expression of Col-I, Alp, Bglap and Runx2 (33,39,40). Runx2 is a transcription factor required for osteoblast differentiation and is expressed at an early stage of differentiation (41). By contrast, Col-I, Alp and Bglap are the earliest biomarkers of mature osteoblast differentiation (42,43). In addition to increased expression of these genes, there was an increase in Alp activity and collagen protein secretion in the present study. Furthermore, there was an increase in the levels of calcified nodules. Collectively, these results suggested that BCE induced osteogenesis and differentiation of pre-osteoblasts into osteoblasts.
Our previous study revealed the half-maximal inhibitory concentration for estrogen receptor α is ~10 nM for E2 and ~5 µg/ml for BCE; relative binding affinity of BCE is 0.06% relative to E2(10). Therefore, BCE may have significantly lower estrogenic activity in MC3T3-E1 cell than 10 nM E2.
Although BCE has been shown to be effective in animal models of osteoporosis (23,24,27), to the best of our knowledge, there has been no study on the effect of BCE on osteoblast differentiation in vivo. As the present in vivo experiment was simple, more detailed studies using microcomputed tomography, are required. Additionally, it is key to isolate osteoblasts from BCE-treated and untreated OVX rats to examine the effect of BCE on osteoblast differentiation. Moriwaki et al (27) reported that bilberry, BCE and their anthocyanins prevent osteoporosis by inhibiting excessive osteoclastogenesis. Although osteoblasts were also used in the aforementioned study, BCE treatment caused no change in cell differentiation. Research findings indicate that estrogen and phytoestrogen enhance osteoblast differentiation (30,31,34); however, results may differ depending on the estrogen sensitivity of osteoblasts. In the present study, phytoestrogen effects were observed in estrogen-sensitive MC3T3-E1 cells. Experiments using estrogen receptor inhibitors were not performed in the present study as our previous studies demonstrated that BCE has phytoestrogenic effects (9,10). However, it is unclear which molecules of BCE exhibit phytoestrogenic effects on MC3T3-E1 cells. Therefore, detailed component analysis of BCE and experiments using estrogen receptor inhibitors against its candidate molecules are necessary in future. Additionally, it is important to verify the effect of different compounds in BCE on osteoclasts. Polyphenols include anthocyanin-rich plants and attenuate osteoporosis (24,27). Notably, their mechanism is reported to include the activation of signaling via the Wnt/β-catenin, transforming growth factor-β/bone morphogenetic protein 2, mitogen-activated protein kinase and PI3K/AKT pathways (44,45). The present study focused on phytoestrogen activity. However, the intracellular signaling mechanism is complex, as phytoestrogens also activate PI3K/AKT, Src/ERK1/2, and nuclear factor κB via the estrogen receptor (46,47). Moreover, BCE likely interacts with these factors via estrogen receptors (10). A microarray analysis of breast cancer MCF7 cells with high expression of estrogen receptors showed that BCE activates multiple pathways (10).
Menopause is associated with increased risk of osteoporosis owing to decreased estrogen secretion. Although hormone replacement therapy is available for treatment of osteoporosis, it is associated with complications, including increased risk of breast cancer (48,49). Research findings indicate that the intake of phytoestrogens, such as soy isoflavones and equol, prevents and ameliorates osteoporosis (21,22). However, as phytoestrogens are not suitable for people who are allergic to soybeans, the anti-osteoporotic effect of blackcurrant is key. Pre-clinical and clinical trials are required to validate the present findings in humans.
In conclusion, BCE and E2 promoted cell proliferation, increased Alp activity and collagen production, upregulated expression of osteoblast differentiation markers, and enhanced mineralization and differentiation of pre-osteoblasts into osteoblasts. Overall, although BCE has phytoestrogenic effects, the present data suggest that it also affected osteoblast differentiation. Collectively, the results suggested that BCE may ameliorate osteoporosis during menopause.
Supplementary Material
Morphology of MC3T3-E1 cells. The morphology did not change after treatment with BCE and 10 nM E2 for 48 h. Scale bar, 500 μm. Ctrl, control; BCE, blackcurrant extract; E2, 17β-estradiol.
Light micrographs of alkaline phosphatase staining of the femur. Representative alkaline phosphatase staining images of the femur sections of rats in (A) sham, (B) OVX and (C) OVX + 3% BCE groups. Arrows indicate epiphyses. Scale bar, 200 μm. OVX, ovariectomized; BCE, blackcurrant extract.
Acknowledgements
Not applicable.
Funding
Funding: The present study was partially supported by the Japan Society for the Promotion of Science KAKENHI (grant no. 20K02402).
Availability of data and materials
The data generated in the present study may be requested from the corresponding author.
Authors' contributions
NN, IO and KH designed the study, performed the experiments and analyzed the data. NN wrote the manuscript. IO edited the manuscript. All authors have read and approved the final manuscript. NN and KH confirm the authenticity of all the raw data.
Ethics approval and consent to participate
The present study was approved by the Animal Research Committee of Hiroaki University (Hirosaki, Japan; approval no. G18003).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Golovinskaia O and Wang CK: Review of functional and pharmacological activities of berries. Molecules. 26(3904)2021.PubMed/NCBI View Article : Google Scholar | |
|
Cortez RE and Gonzalez de Mejia E: Blackcurrants (Ribes nigrum): A review on chemistry, processing, and health benefits. J Food Sci. 84:2387–2401. 2019.PubMed/NCBI View Article : Google Scholar | |
|
Gopalan A, Reuben SC, Ahmed S, Darvesh AS, Hohmann J and Bishayee A: The health benefits of blackcurrants. Food Funct. 3:795–809. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Lephart ED: Phytoestrogens (resveratrol and equol) for estrogen-deficient skin-controversies/misinformation versus anti-aging in vitro and clinical evidence via nutraceutical-cosmetics. Int J Mol Sci. 22(11218)2021.PubMed/NCBI View Article : Google Scholar | |
|
Nguyen M and Osipo C: Targeting breast cancer stem cells using naturally occurring phytoestrogens. Int J Mol Sci. 23(6813)2022.PubMed/NCBI View Article : Google Scholar | |
|
Jia M, Dahlman-Wright K and Gustafsson JA: Estrogen receptor alpha and beta in health and disease. Best Pract Res Clin Endocrinol Metab. 29:557–568. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Lobo RA: Metabolic syndrome after menopause and the role of hormones. Maturitas. 60:10–18. 2008.PubMed/NCBI View Article : Google Scholar | |
|
Prisby RD: Mechanical, hormonal and metabolic influences on blood vessels, blood flow and bone. J Endocrinol. 235:R77–R100. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Nanashima N, Horie K and Maeda H: Phytoestrogenic activity of blackcurrant anthocyanins is partially mediated through estrogen receptor beta. Molecules. 23(74)2017.PubMed/NCBI View Article : Google Scholar | |
|
Nanashima N, Horie K, Tomisawa T, Chiba M, Nakano M, Fujita T, Maeda H, Kitajima M, Takamagi S, Uchiyama D, et al: Phytoestrogenic activity of blackcurrant (Ribes nigrum) anthocyanins is mediated through estrogen receptor alpha. Mol Nutr Food Res. 59:2419–2431. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Horie K, Nanashima N and Maeda H: Phytoestrogenic effects of blackcurrant anthocyanins increased endothelial nitric oxide synthase (eNOS) expression in human endothelial cells and ovariectomized rats. Molecules. 24(1259)2019.PubMed/NCBI View Article : Google Scholar | |
|
Nanashima N and Horie K: Blackcurrant extract with phytoestrogen activity alleviates hair loss in ovariectomized rats. Molecules. 24(1272)2019.PubMed/NCBI View Article : Google Scholar | |
|
Nanashima N, Horie K, Maeda H, Tomisawa T, Kitajima M and Nakamura T: Blackcurrant anthocyanins increase the levels of collagen, elastin, and hyaluronic acid in human skin fibroblasts and ovariectomized rats. Nutrients. 10(495)2018.PubMed/NCBI View Article : Google Scholar | |
|
Nanashima N, Horie K, Yamanouchi K, Tomisawa T, Kitajima M, Oey I and Maeda H: Blackcurrant (Ribes nigrum) extract prevents dyslipidemia and hepatic steatosis in ovariectomized rats. Nutrients. 12(1541)2020.PubMed/NCBI View Article : Google Scholar | |
|
Zhang Y, Liang J, Liu P, Wang Q, Liu L and Zhao H: The RANK/RANKL/OPG system and tumor bone metastasis: Potential mechanisms and therapeutic strategies. Front Endocrinol (Lausanne). 13(1063815)2022.PubMed/NCBI View Article : Google Scholar | |
|
Cheng CH, Chen LR and Chen KH: Osteoporosis due to hormone imbalance: An overview of the effects of estrogen deficiency and glucocorticoid overuse on bone turnover. Int J Mol Sci. 23(1376)2022.PubMed/NCBI View Article : Google Scholar | |
|
Tunheim EG, Skallevold HE and Rokaya D: Role of hormones in bone remodeling in the craniofacial complex: A review. J Oral Biol Craniofac Res. 13:210–217. 2023.PubMed/NCBI View Article : Google Scholar | |
|
Okagu IU, Aham EC, Ezeorba TPC, Ndefo JC, Aguchem RN and Udenigwe CC: Osteo-modulatory dietary proteins and peptides: A concise review. J Food Biochem. 46(e14365)2022.PubMed/NCBI View Article : Google Scholar | |
|
Sfeir JG, Drake MT, Khosla S and Farr JN: Skeletal aging. Mayo Clin Proc. 97:1194–1208. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Zhou T, Gai Z, Gao X and Li L: The potential mechanism of exercise combined with natural extracts to prevent and treat postmenopausal osteoporosis. J Healthc Eng. 2021(2852661)2021.PubMed/NCBI View Article : Google Scholar | |
|
Harahap IA and Suliburska J: Probiotics and isoflavones as a promising therapeutic for calcium status and bone health: A narrative review. Foods. 10(2685)2021.PubMed/NCBI View Article : Google Scholar | |
|
Mayo B, Vázquez L and Flórez AB: Equol: A bacterial metabolite from the daidzein isoflavone and its presumed beneficial health effects. Nutrients. 11(2231)2019.PubMed/NCBI View Article : Google Scholar | |
|
Sakaki J, Melough M, Lee SG, Kalinowski J, Koo SI, Lee SK and Chun OK: Blackcurrant supplementation improves trabecular bone mass in young but not aged mice. Nutrients. 10(1671)2018.PubMed/NCBI View Article : Google Scholar | |
|
Zheng X, Mun S, Lee SG, Vance TM, Hubert P, Koo SI, Lee SK and Chun OK: Anthocyanin-rich blackcurrant extract attenuates ovariectomy-induced bone loss in mice. J Med Food. 19:390–397. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Nosal BM, Sakaki JR, Macdonald Z, Mahoney K, Kim K, Madore M, Thornton S, Tran TDB, Weinstock G, Lee ECH and Chun O: Blackcurrants reduce the risk of postmenopausal osteoporosis: A pilot double-blind, randomized, placebo-controlled clinical trial. Nutrients. 14(4971)2022.PubMed/NCBI View Article : Google Scholar | |
|
Oursler MJ, Landers JP, Riggs BL and Spelsberg TC: Oestrogen effects on osteoblasts and osteoclasts. Ann Med. 25:361–371. 1993.PubMed/NCBI View Article : Google Scholar | |
|
Moriwaki S, Suzuki K, Muramatsu M, Nomura A, Inoue F, Into T, Yoshiko Y and Niida S: Delphinidin, one of the major anthocyanidins, prevents bone loss through the inhibition of excessive osteoclastogenesis in osteoporosis model mice. PLoS One. 9(e97177)2014.PubMed/NCBI View Article : Google Scholar | |
|
Ahmad Hairi H, Jamal JA, Aladdin NA, Husain K, Mohd Sofi NS, Mohamed N, Mohamed IN and Shuid AN: Demethylbelamcandaquinone B (Dmcq B) is the active compound of marantodes pumilum var. Alata (Blume) kuntze with osteoanabolic activities. Molecules. 23(1686)2018.PubMed/NCBI View Article : Google Scholar | |
|
Kim M, Lim J, Lee JH, Lee KM, Kim S, Park KW, Nho CW and Cho YS: Understanding the functional role of genistein in the bone differentiation in mouse osteoblastic cell line MC3T3-E1 by RNA-seq analysis. Sci Rep. 8(3257)2018.PubMed/NCBI View Article : Google Scholar | |
|
Luo D, Kang L, Ma Y, Chen H, Kuang H, Huang Q, He M and Peng W: Effects and mechanisms of 8-prenylnaringenin on osteoblast MC3T3-E1 and osteoclast-like cells RAW264.7. Food Sci Nutr. 2:341–350. 2014.PubMed/NCBI View Article : Google Scholar | |
|
Jiang X, Chen W, Shen F, Xiao W, Guo H, Su H, Xiu J and Sun W: Pinoresinol promotes MC3T3-E1 cell proliferation and differentiation via the cyclic AMP/protein kinase A signaling pathway. Mol Med Rep. 20:2143–2150. 2019.PubMed/NCBI View Article : Google Scholar | |
|
Czekanska EM, Stoddart MJ, Richards RG and Hayes JS: In search of an osteoblast cell model for in vitro research. Eur Cell Mater. 24:1–17. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Mao W, Huang G, Chen H, Xu L, Qin S and Li A: Research progress of the role of anthocyanins on bone regeneration. Front Pharmacol. 12(773660)2021.PubMed/NCBI View Article : Google Scholar | |
|
Kim H, Tabata A, Tomoyasu T, Ueno T, Uchiyama S, Yuasa K, Tsuji A and Nagamune H: Estrogen stimuli promote osteoblastic differentiation via the subtilisin-like proprotein convertase PACE4 in MC3T3-E1 cells. J Bone Miner Metab. 33:30–39. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Cai W, Sun B, Song C, Liu F, Wu Z and Liu Z: Resveratrol induces proliferation and differentiation of mouse pre-osteoblast MC3T3-E1 by promoting autophagy. BMC Complement Med Ther. 23(121)2023.PubMed/NCBI View Article : Google Scholar | |
|
Norikura T, Sasaki Y, Kojima-Yuasa A and Kon A: Glyoxylic Acid, an α-keto acid metabolite derived from glycine, promotes myogenesis in C2C12 cells. Nutrients. 15(1763)2023.PubMed/NCBI View Article : Google Scholar | |
|
Livak KJ and Schmittgen TD: Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.PubMed/NCBI View Article : Google Scholar | |
|
Xiao HH, Fung CY, Mok SK, Wong KC, Ho MX, Wang XL, Yao XS and Wong MS: Flavonoids from Herba epimedii selectively activate estrogen receptor alpha (ERα) and stimulate ER-dependent osteoblastic functions in UMR-106 cells. J Steroid Biochem Mol Biol. 143:141–151. 2014.PubMed/NCBI View Article : Google Scholar | |
|
Hu B, Chen L, Chen Y, Zhang Z, Wang X and Zhou B: Cyanidin-3-glucoside regulates osteoblast differentiation via the ERK1/2 signaling pathway. ACS Omega. 6:4759–4766. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Park KH, Gu DR, So HS, Kim KJ and Lee SH: Dual role of cyanidin-3-glucoside on the differentiation of bone cells. J Dent Res. 94:1676–1683. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Matsushita Y, Ono W and Ono N: Growth plate skeletal stem cells and their transition from cartilage to bone. Bone. 136(115359)2020.PubMed/NCBI View Article : Google Scholar | |
|
Fusaro M, Crepaldi G, Maggi S, D'Angelo A, Calo L, Miozzo D, Fornasieri A and Gallieni M: Bleeding, vertebral fractures and vascular calcifications in patients treated with warfarin: Hope for lower risks with alternative therapies. Curr Vasc Pharmacol. 9:763–769. 2011.PubMed/NCBI View Article : Google Scholar | |
|
Shao X, Cao X, Song G, Zhao Y and Shi B: Metformin rescues the MG63 osteoblasts against the effect of high glucose on proliferation. J Diabetes Res. 2014(453940)2014.PubMed/NCBI View Article : Google Scholar | |
|
Setchell KDR and Lydeking-Olsen E: Dietary phytoestrogens and their effect on bone: Evidence from in vitro and in vivo, human observational, and dietary intervention studies. Am J Clin Nutr. 78 (3 Suppl):593S–609S. 2003.PubMed/NCBI View Article : Google Scholar | |
|
Torre E: Molecular signaling mechanisms behind polyphenol-induced bone anabolism. Phytochem Rev. 16:1183–1226. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Arnal JF, Lenfant F, Metivier R, Flouriot G, Henrion D, Adlanmerini M, Fontaine C, Gourdy P, Chambon P, Katzenellenbogen B and Katzenellenbogen J: Membrane and nuclear estrogen receptor alpha actions: From tissue specificity to medical implications. Physiol Rev. 97:1045–1087. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Canivenc-Lavier MC and Bennetau-Pelissero C: Phytoestrogens and health effects. Nutrients. 15(317)2023.PubMed/NCBI View Article : Google Scholar | |
|
Dinger J, Bardenheuer K and Heinemann K: Drospirenone plus estradiol and the risk of serious cardiovascular events in postmenopausal women. Climacteric. 19:349–356. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Dinger J, Do Minh T and Heinemann K: Impact of estrogen type on cardiovascular safety of combined oral contraceptives. Contraception. 94:328–339. 2016.PubMed/NCBI View Article : Google Scholar |