Protective effects of flavonoids against intracerebral and subarachnoid hemorrhage (Review)
- Authors:
- Published online on: July 4, 2024 https://doi.org/10.3892/etm.2024.12639
- Article Number: 350
-
Copyright: © Dong et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
1. Introduction
Intracerebral hemorrhage and subarachnoid hemorrhage
Intracerebral hemorrhage (ICH) is a deleterious form of stroke that results from cerebral vascular rupture and hemorrhage. It commonly occurs in the deep basal ganglia region of the brain and is associated with a high incidence and mortality rate (1). This subtype of hemorrhagic stroke comprises 10-20% of all documented stroke cases worldwide, based on statistical data from 1970 to 2008(2). A systematic review and meta-analysis published in 2010 showed that the overall incidence of ICH was 24.6 per 100,000 person-years; among them, incidence of ICH per 100,000 person-years was 51.8 in East and Southeast Asian individuals, which was much higher compared with that in white individuals in Oceania, Europe and northern Manhattan (24.2), black individuals in Africa, Caribbean and northern Manhattan (22.9) and Hispanic individuals in Brazil, northern Manhattan, Caribbean and northern Chile (19.6); moreover, the incidence rate of ICH was higher in the elderly, with an incidence per 100,000 person-years of ICH in individuals >55 years old (36.5-196.0), which was much higher compared with that in individuals aged ≤54 (1.9-19.1) (3).
ICH is a devastating condition with a reported mortality of ~40% within the first month (3). Despite the temporary lack of comprehensive epidemiological data on ICH of the last five years, it is undeniable that, having being recognized as one of the most challenging conditions for the prognosis of patients with stroke (4), ICH is likely to remain a serious public health problem that cannot be ignored in the near future, even after the progress in medicine in the past few years. There are two types of damage caused by ICH: Primary injury and secondary injury (5). In ICH, the primary injury occurs during the initial stage. It is characterized by physical damage to the brain cell structure caused by the hematoma (6). The increased intracranial pressure from the hematoma mass leads to compression of the brain region, causing mechanical damage (7). Following this, a series of reactions occur, including neuroinflammation and oxidative stress (OS), leading to further damage such as blood-brain barrier (BBB) dysfunction and cerebral edema. This is referred to as a secondary injury (4). They both seriously harm the health of patients with ICH.
Subarachnoid hemorrhage (SAH) has also attracted considerable interest in the study of hemorrhagic stroke in recent years. It is a type of neurological emergency that arises from the rupture of diseased blood vessels found either at the brain's surface or at the base of the brain. The blood flows directly into the subarachnoid space (8), resulting in the third most prevalent subtype of stroke (9) mainly attributed to aneurysm rupture (10). It may have severe consequences for patients, such as cognitive decline and secondary cerebral ischemia (11). It occurs at a relatively young age, despite comprising only 5% of all strokes (12). Unlike ICH, SAH starts very quickly, and symptoms can peak in a few minutes, with a thunderclap headache which can peak within just 1 minute after SAH (13). The complex pathophysiological cascade that occurs after SAH is primarily triggered by increased intracranial pressure (ICP) and blood components (14). These factors can lead to unpredictable diffuse brain damage, which is challenging to detect until it reaches an irreversible stage (15).
Two areas in the pathophysiology of SAH have attracted significant attention in scientific research: Early brain injury (EBI) and cerebral vasospasm (CVS) (16). Among them, EBI after SAH refers to secondary brain injury within 72 h after SAH, including microcirculation dysfunction, neuroinflammation damage, BBB disruption, cerebral edema, neuronal death and oxidative cascade (17). The EBI after SAH is associated with several pathogenic mechanisms, including inflammation, OS, and ferroptosis (18,19). Moreover, CVS is a significant contributor to the high mortality and morbidity associated with SAH (20). CVS usually occurs on day 3 after SAH, peaks on days 6 and 8, lasts 2 to 3 weeks (17) and has lasting and serious consequences after SAH. After a review of the relevant literature, it was observed that the EBI and CVS after SAH have similar mediating pathways, including OS and inflammation. This suggests that flavonoid compounds have the potential to protect against a number of significant injury modes in SAH.
Flavonoids
Flavonoids, which have 2-phenylchromone as their fundamental structural unit, are among the most abundant secondary metabolites found in plants. They can be found in herbs and different dietary sources, including fruits, vegetables, teas and grains, in glycosides or a number of free forms (21). Flavonoid compounds consist of a shared C6-C3-C6 backbone where two aromatic rings are linked by 3-carbon bridges, typically forming a phenyltropane configuration (22). Several studies have shown that natural flavonoid compounds have neuroprotective effects in brain diseases (23,24), suggesting that understanding the functions and mechanisms of flavonoids in preventing neuroinflammation may hold immense significance in the advancement of nutritional guidelines and therapeutic approaches for brain diseases.
2. Protective effects of flavonoid compounds against ICH
Flavonoids protect against brain damage after ICH by inhibiting inflammation and OS
The potential of flavonoids to protect against ICH through their superior anti-inflammatory and antioxidant properties has been the main focus of recent studies.
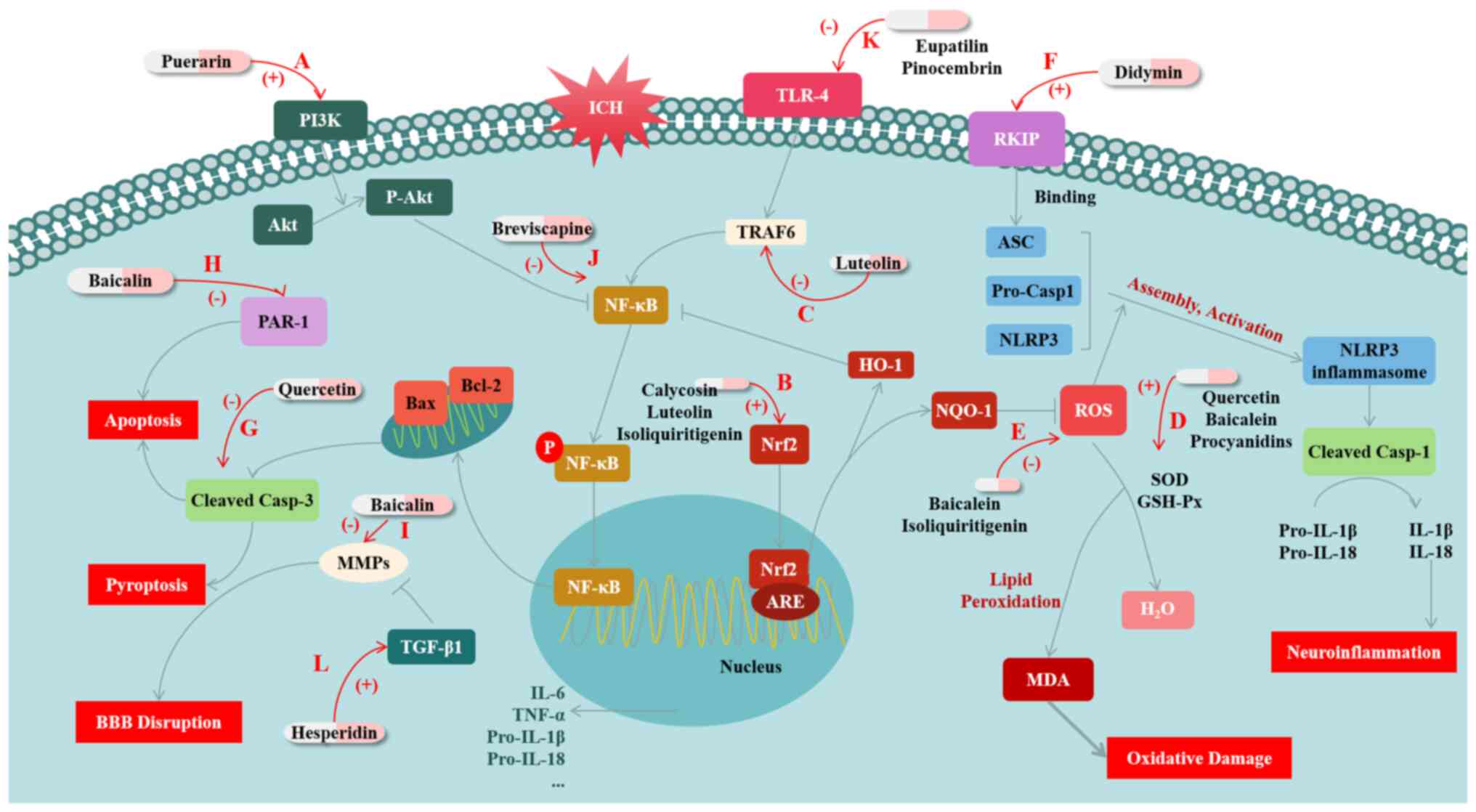
Puerarin is a natural isoflavone extracted from the roots of pueraria species. A previous study has documented the protective effects of puerarin against various diseases such as Alzheimer's disease and cerebral ischemic disease (23). The PI3K/Akt signaling pathway is involved in regulating the NF-κB pathway. The activation of PI3K/Akt signaling has been shown to effectively decrease the levels of phosphorylated NF-κB p65 and to inhibit the production of inflammatory cytokines such as TNF-α and IL-1β. Zeng et al (24) showed that puerarin can activate PI3K/Akt signaling, thereby inhibiting inflammatory reactions caused by the NF-κB pathway and ultimately attenuating the EBI after ICH. Specifically, puerarin activates the PI3K/Akt pathway and reduces NF-κB activation, significantly increasing the expression level of Bcl-2 and inhibiting the expression of Bax and Caspase-3, thereby inhibiting ICH-induced activation of apoptosis signaling (24). At the same time, the inflammatory factors that are promoted by NF-κB are downregulated, reducing the inflammatory damage after ICH. In addition, puerarin also reduces the levels of 8-OHdG and 3-NT after ICH, inhibiting the production of reactive oxygen species (ROS) and thus alleviating OS damage after ICH (24).
In addition, a previous study has revealed that luteolin, which is classified as a flavonoid compound and has anti-inflammatory properties, has been found to stimulate the Nrf2 pathway and facilitate the nuclear translocation of Nrf2 after ICH (25). The study has shown that luteolin can prevent ICH by stimulating the p62/Keap1/Nrf2 pathway and substantially increasing the expression levels of its downstream antioxidant proteins heme oxygenase 1 (HO-1) and NQO1(26). NQO1 regulates ROS production to inhibit oxidative stress (5), while increased expression of HO-1 can inhibit the activation of NF-κB, thereby inhibiting NF-κB-mediated inflammatory responses (27). Further, luteolin inhibits the activation of the TLR4/TRAF6/NF-κB signaling pathway by binding to TRAF6. This mechanism of action reduces neuroinflammation, thereby providing a protective effect against ICH by attenuating the overproduction of inflammatory cytokines, the inflammatory cascade, apoptosis and structural and functional disruption of BBB may result (28).
Baicalein is a bioactive flavonoid with anti-inflammatory and antitumor activities (29). Based on a report, ICH rats treated with baicalein showed significant reductions in lesion volume and brain water content. On the one hand, its action was manifested as decreasing the expression levels of pro-inflammatory cytokines (IL-1β, IL-4, IL-6 and TNF-α), and inhibiting apoptosis. On the other hand, it also increased the activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px), while decreasing the malondialdehyde (MDA) level in the brain tissues of rats (30). MDA is an important marker in oxidative stress, and the enhanced activity of SOD and GSH-Px can strongly inhibit the production of MDA by lipid peroxidation to improve oxidative damage after ICH (31). The results of the research indicated that baicalein may have a therapeutic impact on brain injury by reducing apoptosis, OS and neuroinflammation. This suggests that baicalein could be a promising treatment option for ICH and related brain injury (30). Another study on ICH supports that baicalein may reduce brain damage by inhibiting ROS and the NLRP3 inflammasome, thus inhibiting NLRP3-mediated inflammatory responses. This research further highlights the potential of baicalein to protect against ICH (32).
Moreover, didymin, a dietary citrus flavonoid, was shown to upregulate RAF kinase inhibitor protein (RKIP) expression in a preclinical model of ICH (33). The research has shown that RKIP can directly bind ASC, thereby disrupting the formation of the NLRP3 inflammasome after ICH (33). In this way, didymin blocks cell pyroptosis and the inflammatory response in the Caspase-1/GSDMD pathway, protecting against injury after ICH (33).
Quercetin, a special subclass of flavonoids, which is a bioactive natural compound established on the structure of flavonoids (34). Based on a recent study, the efficacy of quercetin in a non-clinical model of ICH has been demonstrated. The study reviewed that, the lesion volume and brain water content are significantly reduced in ICH rats after quercetin treatment. These outcomes provided evidence that quercetin has the potential to suppress inflammation and cell death, and is capable of aiding in the restoration of neural function by reducing levels of inflammatory mediators and inhibiting apoptosis mediated by cleaved Caspase-3(35).
Baicalin is an active ingredient of the traditional Chinese drug baicalensis that shows biological activity, including anti-inflammatory properties (36,37). A study found that baicalin displays a dose-dependent suppression of NF-κB expression in the surrounding tissues of the hematoma caused by ICH, and thereby reduces the secretion of IL-1β and IL-6. Additionally, baicalin inhibits the expression of matrix metalloproteinases (MMP)-9, and blocks the degradation of the extracellular matrix (ECM) by MMP-9, thereby helping maintain the integrity of the BBB. It was also suggests that baicalin may have a protective effect against ICH by regulating the expression of protease-activating receptor-1 (PAR-1) to inhibit the cellular apoptosis pathway mediated by PAR-1(38).
A number of other potential flavonoid compounds have demonstrated a protective effect against the OS and inflammation mediated by ICH; for example, a study has highlighted that isoliquiritigenin can inhibit ROS and the activation of the NF-κB-mediated NLRP3 inflammasome through the stimulation of the Nrf2 antioxidant pathway, which in turn alleviates EBI after experimental ICH (39). Breviscapine, a medicinal plant, reportedly possesses a substantial inhibitory effect on the expression of NF-κB pathway-related factors after ICH (40). Another study suggests that pinocembrin lowers the expression of TLR4, myeloid differentiation primary response 88 (MyD88) and TIR-domain-containing adapter-inducing interferon-β and downregulates NF-κB signaling, alleviating brain injury after ICH by inhibiting the inflammatory response (41). Except for pinocembrin, eupatilin has also been confirmed to reduce the inflammatory response triggered by ICH through the TLR4/MyD88 pathway (42). In addition to the aforementioned studies, fisetin, naringin, calycosin and procyanidins have also been demonstrated to protect against the neuroinflammation and/or OS resulting from ICH via various mechanisms, such as inhibiting NF-κB, inhibiting lipid peroxidation and activating the Nrf2 pathway (43-46).
Flavonoids protect ICH by facilitating TGF-β1
The aforementioned materials demonstrate that flavonoids have a wide range of potential as antioxidants and/or anti-inflammatory agents for the prevention and treatment of ICH. However, aside from neuroinflammation and OS, a number of researchers have also identified alternative pathways through which flavonoids can protect ICH. For example, hesperidin, a biologically active flavonoid that can be found in citrus fruits (47), has been highlighted to promote the expression of TGF-β1, while TGF-β1 can promote the production and reconstitution of ECM and protects the BBB by inhibiting the expression of MMP9 and MMP2, to attenuate the damage caused by adverse symptomatic ICH caused by ischemic stroke (48). At present, there are few studies on the protective effect of flavonoid compounds on ICH, which have highlighted the role of TGF-β1; however, the present study provides a reference for future research directions.
Based on the aforementioned studies it is evident that flavonoids have the potential to provide significant protection against ICH through various mechanisms, such as anti-inflammatory, anti-oxidative and anti-pyroptotic actions (Fig. 1; Table I). These findings suggest that flavonoids may be promising candidates for the development of preventive and therapeutic drugs.
3. Protective effects of flavonoid compounds against SAH
Flavonoids protect SAH by inhibiting inflammation, OS and apoptosis
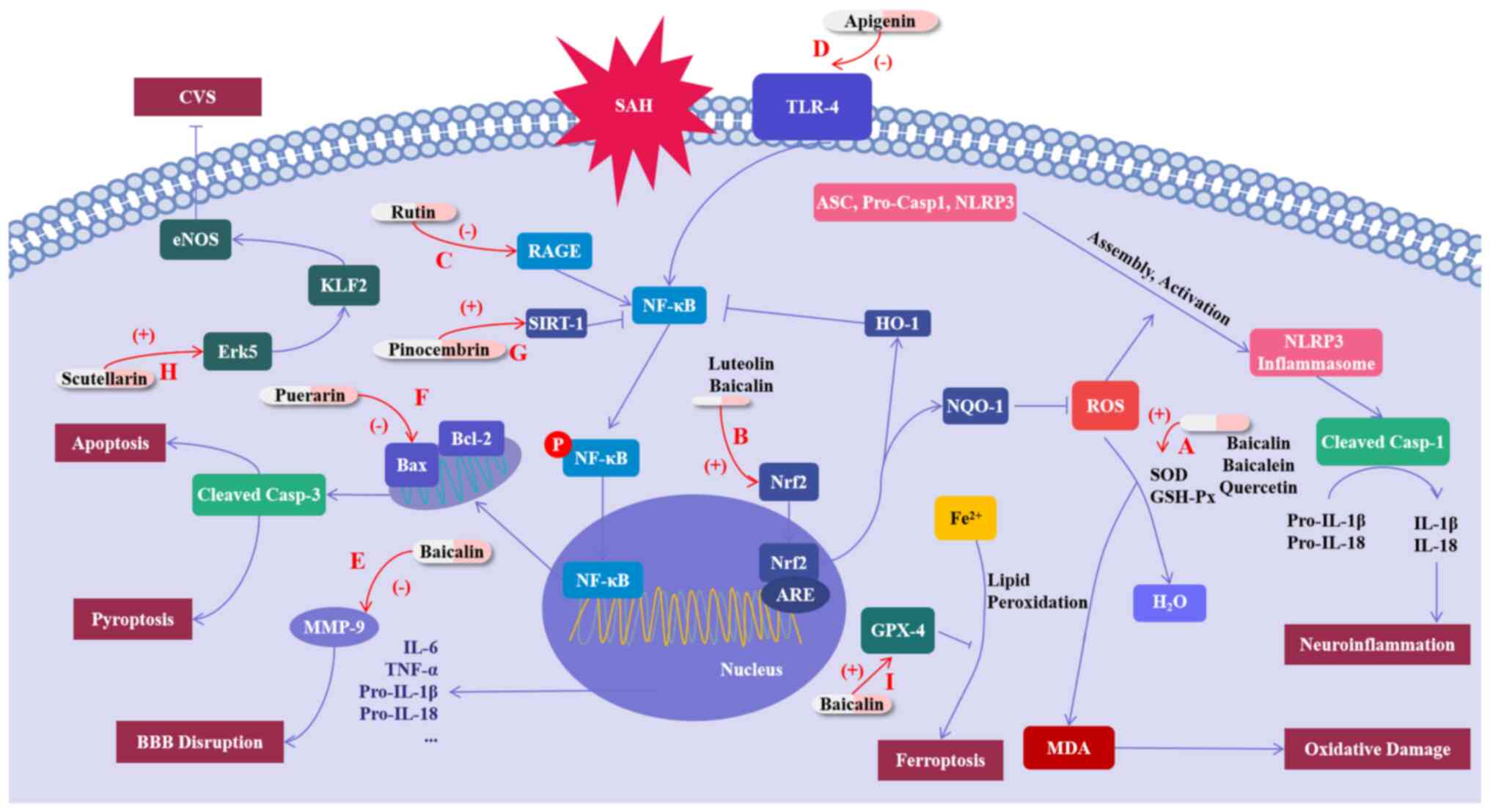
Neuroinflammation and OS are significant factors in the EBI and CVS that occur after SAH (49-51). A number of studies have demonstrated that flavonoids can exert protective effects on SAH by suppressing inflammation and OS. For example, the study by Kuo et al (52) has highlighted the positive effects of early baicalein treatment on rats after SAH. This treatment has been shown to decrease the mortality rate and brain water content in experimental rats, as well as reduce neuronal degeneration by inhibiting CVS (52). In addition, baicalein increases astrocyte activity and retains glutamate transporter-1, thus attenuating OS induced by the glutamate surge after SAH; baicalein also provided resistance against OS by maintaining SOD and catalase activity and reducing MDA levels after SAH (52). Several studies have also emphasized the functions of flavonoid compounds in the similar aspects in SAH, such as luteolin, rutin, apigenin, baicalein, quercetin and proanthocyanidin, which have been shown to have inhibitory effects on inflammation and/or OS after SAH (53-60). Their mechanisms of action include several pathways as described above, such as modulating Nrf2 to inhibit oxidative stress and NLRP3 inflammasome-mediated inflammation, reducing NF-κB activation by inhibiting TLR4 or RAGE, and inhibiting of MDA by increasing SOD and GSH-Px to alleviate oxidative damage (53-60).
Simultaneously, the damage caused by apoptosis after SAH is equally remarkable. Apoptosis is a significant factor in the development of EBI and is also involved in the formation of CVS in patients with SAH. According to the study of Zhang et al (61), puerarin ameliorates the neurological impairment observed in mice, suppresses cerebral edema, decreases BBB destruction and also decreases the apoptosis of cellular neurons. In addition, Zhang et al (61) has shown that puerarin-treated SAH mice have a significantly higher Blc-2/Bax ratio and a reduced level of cleaved Caspase-3 compared with vehicle-treated SAH animals; that is to say, puerarin inhibits apoptosis mediated by cleaved Caspase-3 by inhibiting Bax expression and promoting Blc-2 expression. Meanwhile, puerarin also blocks SAH-induced ROS production, protects the synthesis of Sirt3 after SAH and enhances the function of SOD2 after SAH, thus attenuating oxidative damage after SAH. Overall, the study suggests that puerarin has the potential to reduce neurological dysfunction in mice with SAH by targeting specific pathways involved in apoptosis, including Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2(61).
Flavonoids protect against brain damage after SAH by activating SIRT1
In addition to the aforementioned pathways, a number of other flavonoids have been shown to protect against brain damage after SAH through other routes.
Pinocembrin is a natural compound distributed in propolis. In a study, Zeng et al (62) found that pinocembrin has a significant impact on improving behavior and reducing brain tissue damage after SAH, and that its mechanism of action might be related to Sirtuin-1(62). Sirtuin-1, also known as SIRT-1, is a histone deacetylase that can be found in different parts of the cerebral cortex. Accumulating preclinical evidence has indicated that SIRT1 is a promising molecular candidate for treating SAH. By deacetylating a variety of intracellular targets such as Pgc-1α and ac-NF-κB, SIRT1 provides protection by reducing inflammatory injury, free radical damage and cell death (62). Specifically, inhibition of ac-NF-κB can inhibit its downstream inflammatory pathway, while activation of Pgc-1α can promote the entry of Pgc-1α into the nucleus to promote the expression of antioxidant enzymes, such as SOD, to reduce intracellular ROS levels, thereby protecting normal mitochondrial function and protecting cells from oxidative damage (63). In the study by Zeng et al (62), treatment with pinocembrin significantly increases the levels of SIRT1 and Pgc-1α, and suppresses the expression of ac-NF-κB; therefore, treatment with pinocembrin plays a protective role against brain injury after SAH (62). Although SIRT1 was shown in this aforementioned study to exert a protective effect on SAH by inhibiting inflammation and OS, as a potential target for the treatment of SAH, SIRT1 is less studied in the direction of the protective effects of flavonoids on SAH. Overall, the potential of flavonoids to play a role in the treatment of SAH through SIRT1 still needs to be further explored.
Flavonoids protect against brain injury after SAH by promoting the endothelial nitric oxide (NO) pathway
Concerning the pathophysiology of CVS, the endothelial NO pathway is also regarded as one of the primary mechanisms. The production of NO by endothelial nitric oxide synthase (eNOS) in the cerebrovascular endothelium can spread to nearby smooth muscle cells, triggering the activation of soluble guanylyl cyclase. This, in turn, results in the production of cyclic guanosine monophosphate (cGMP). The stimulation of intracellular calcium channels by cGMP facilitates the transport of free Ca2+ to the intracellular zone compartment, resulting in the relaxation of smooth muscle cells, and eventually inhibits CVS (16). Li et al (64) demonstrated that scutellarin can reduce vasospasm and neurological deficits by regulating the Erk5-KLF2-eNOS pathway, confirming the protective effect of scutellarin through the endothelial NO pathway on CVS after SAH (64).
Flavonoids protects against brain injury after SAH by inhibiting ferroptosis
Ferroptosis is a type of programmed cell death characterized by iron-dependent lipid peroxidation. Ferroptosis can trigger some inflammatory mediators, such as TNF-α, IL-1β and IL-6, which can contribute to inflammation by promoting the aggregation and activation of inflammatory cells (65,66). Ferroptosis can also enhance the accumulation of ROS within cells, thereby intensifying the OS damage (67,68). Recently, ferroptosis has been reported in the pathological course of hemorrhagic stroke, including SAH (69). One study has indicated that baicalin may be effective in reducing the damage caused by SAH by preventing ferroptosis. The research highlighted that baicalin attenuates SAH-induced elevation of Fe2+ levels and production of OS markers MDA and ROS in rat brain tissue, and eliminates SAH-induced reduction of GSH levels. In addition to validating Baicalin's inhibition of lipid peroxidation, the study also revealed that baicalin maintains the expression level of glutathione peroxidase 4 (GPX4) protein, which can reduce phospholipid hydroperoxide and inhibit lipoxygenase-mediated lipid peroxidation, thereby exerting a protective effect against ferroptosis. In addition, baicalin also inhibits the protein level of beclin1 and the ratio of LC3-II/I in the brain tissues of SAH rats, suggesting the suppressive effect of baicalin on autophagy in SAH rats. This study demonstrates the ability of baicalin to attenuate brain damage following SAH by inhibiting autophagy-dependent ferroptosis (70).
In conclusion, flavonoids regulate and improve brain damage after SAH in various ways. These are a class of drugs with potential for the treatment of SAH (Fig. 2; Table II). However, further research is still required to elucidate their mode of action.
4. Conclusion
ICH and SAH are harmful brain diseases that cause severe damage to patients globally. Flavonoids, as potent antioxidants and anti-inflammatory agents, have consistently demonstrated their neuroprotective effects in preclinical studies involving ICH and SAH. They have been shown to regulate inflammation, pyroptosis and OS pathways, which in turn involve, for example, the NF-κB-related inflammatory pathways, Nrf2-related antioxidant pathways, lipid peroxidation pathways and caspase-3-mediated pyroptosis. Although their anti-inflammatory, antioxidant and other effects have been demonstrated, flavonoid compounds still need more research to investigate their potential, and they are still constrained by a number of obstructions on the way to the clinic, for example, the mechanism research is not in-depth enough, and the potential toxic and side effects of flavonoids need to be discovered and avoided.
More pathways, targets and more specific mechanisms remain to be discovered and exploited
Other than the researches mentioned in the previous chapters, it is important to highlight that some targets have attracted less attention in the available research. Targets such as TGF-β1, SIRT1, ERK1/2 and RAGE, and pathways such as ferroptosis may potentially be affected by a number of flavonoids. Especially, the activation of the ERK1/2 signaling pathway is implicated in the pathological process of vascular wall proliferation in CVS (71). It has been revealed that cell proliferation in the vessel wall is a crucial factor in the development of CVS in SAH (71), and TLR4, downstream of ERK1/2, can exacerbate the inflammatory response by promoting the secretion of inflammatory factors through c-Fos phosphorylation (72). This, in turn, increases the damage caused by SAH. Moreover, numerous studies have confirmed the inhibitory effects of various flavonoid compounds on ERK1/2-related pathways (73,74). These findings also provide considerable reference value for studies targeting SAH. These targets and pathways may serve as potential sites of damage for different flavonoids, and their research directions are both attractive and immensely valuable.
While studying the effects of flavonoids on new pathways and targets, the studies of pathways and targets that have been demonstrated to be affected by flavonoids should not be stopped. While numerous studies have explored the mechanisms by which flavonoids exert protective effects on ICH and SAH in the studies cited in the present paper, the ways in which flavonoids interact with the specific biomarkers they affect are rarely mentioned. Future research is still necessary to explore this point in depth; for example, to study whether flavonoids play a role as activators or inhibitors of certain enzymes, and to study the binding mode and binding sites of flavonoids to various targets, so as to lay a more reliable theoretical basis for their clinical treatment of ICH and SAH.
Flavonoids may also have negative effects
In a number of studies flavonoid compounds have played a role that can exert protective effects against a variety of diseases. Inevitably, however, there are exceptions to everything and the effects of flavonoid compounds on hemorrhagic stroke are also complex and two-sided. In a certain study, the present study noted that rotenone, a flavonoid compound, has been shown to accelerate ferroptosis after ICH (75). It appears that this type of compound may not be beneficial for treating ICH and SAH. This may be due to the special structure of rotenone, which makes it an inhibitor of mitochondrial respiratory chain complex I, which can enhance the production of mitochondrial ROS (76). The increased production of ROS aggravates lipid peroxidation, which ultimately promotes ferroptosis. This also indicates that some flavonoid compounds may not have a positive impact on hemorrhagic strokes. Upon closer investigation, the present study revealed that there are a number of flavonoid compounds with some potential toxic side effects, including but not limited to: i) Baicalin can induce acetylation of Smad3 through the interaction of Smad3 with the transcriptional coactivator p300 and reduce phosphorylation of AMPK, a metabolic master kinase, leading to increased kidney damage, including glomerular hypertrophy, collagen deposition, inflammatory cell infiltration among the renal tubules and even kidney fibrosis (77); and ii) under the conditions of high dose and long-term use, luteolin induces glutathione depletion and activates the metabolism of CYP450, mediating the formation of o-benzoquinone metabolites, thereby causing cytotoxicity including damage to multiple structures and functions in cells, and even cell apoptosis in primary rat hepatocytes (78).
The potential toxicity of flavonoid compounds has become a non-negligible obstacle to their clinical development. Not only that, but the decision on the dosage and the risks involved in the long-term use of the drug should not be ignored. A too-high dosage or long-term use of a drug may have strong toxic side effects and even a risk of death. Nowadays, there is still a lack of more specific and comprehensive clinical studies on the efficacy and toxicity of flavonoids at different doses and the risks of long-term use of flavonoid drugs. Therefore, in future studies, flavonoid compounds with specific structures, toxicity, functions, dose-dependent toxicity and/or side effects and long-term medication risks should be identified and screened and selectively used. This shows a new opportunity for further research, while also raising new challenges. At present, some studies have made attempts to investigate this; for example, Choi et al (79) revealed that long-term combined administration of quercetin and daidzein can inhibit quercetin-induced suppression of glutathione antioxidant defenses (79). Such studies undoubtedly provide evidence to further study the safety of flavonoids and propose treatment options those are safer and more effective.
Issues of bioavailability and biological activities of flavonoid compounds
Another challenge for flavonoids moving towards clinical practice is that the bioavailability of flavonoid compounds varies (80). However, new delivery systems may help different flavonoid compounds play a fuller role in the prevention and treatment of diseases such as ICH and SAH. As research reports, the application of quercetin in nanoemulsion has been found to significantly boost its antioxidant effect (81). This study indicated that the biological activity and pharmacological effects of flavonoids may vary depending on the dosage form of their administration. This finding offers a strong basis and motivation for further research into the potential of these compounds.
Presently, there is a continuous influx of new dosage forms being identified. Several potent flavonoids may be formulated into various dosage forms so that their protective and toxic levels in each formulation can be determined. This could be another interesting point of research. On the other hand, derivatives are also important in enhancing the efficiency of flavonoid compounds. A previous study has shown that the C- and O-glycosides of flavonoids generally show higher radical scavenging activity compared with aglycones; for example, kaempferol C3-O-glycoside (astragalin) shows higher activity compared with kaempferol (82). This study reveals that making flavonoid compounds into different derivatives may reveal unexpected effects. At the same time, in future research, researchers should also pay attention to the selection of patients. Patients with different constitutions may also show considerable differences in drug compliance, which is also a direction worthy of further study.
Considering the aforementioned research progress, flavonoid compounds have shown significant bioactivity and promising clinical potential in ICH and SAH. Nevertheless, the current studies still have some limitations. Firstly, in the studies mentioned in the present review, the experimental animal breeds, animal models, measurement methods and measurement indicators are uneven, making a systematic comparison of their specific efficacy difficult. Therefore, for each compound, it is necessary to compare the protective effects of flavonoids on ICH and SAH in different experimental animal breeds and by different ICH or SAH model construction methods. This not only helps to compare the efficacy of different flavonoids, but also supports the existing research results.
In addition, to the best of our knowledge, there is a lack of large-scale clinical trials and extensive exploration of mechanistic studies. The ‘from the bench to the bedside’ process still contains more unknowns waiting to be explored. Despite this, in our opinion, foods containing flavonoids may play an unexpected role in preventing diseases such as ICH, SAH and other interrelated diseases, and would be readily available as a daily dietary supplement by the concept of ‘homology of medicine and food’ in traditional Chinese medicine. In daily life, individuals can selectively increase their intake of foods that contain beneficial flavonoid compounds, in order to prevent diseases such as ICH and SAH. For example, a study showed that individuals with higher daily intake of flavonoids, such as anthocyanin (predominantly from blueberries and strawberries), had a lower incidence of hypertension (83). Hypertension is an important cause of ICH (84), therefore, increasing the intake of these flavonoids will also probably reduce the risk of ICH accordingly. Flavonoid compounds are widely found in a variety of fruits and vegetables, for example, celery and parsley contain apigenin (85), apples contain rutin (86) and strawberries and blueberries contain anthocyanin (83), and advocating for an appropriate increase in the intake of flavonoids containing those beneficial flavonoids may be of considerable benefit in preventing ICH and SAH and reducing their incidence.
A number of previous reviews have summarized studies on flavonoids. Jäger and Saaby (87) discussed the effects of flavonoids on the central nervous system, including oral bioavailability, BBB permeability and the interaction of flavonoids with several biomarkers. The review by Parrella et al (88) summarized the therapeutic effects of polyphenols, including flavonoids, shown in non-clinical and clinical studies of stroke, while the article by Chen et al (21) summarized the beneficial effects of natural flavonoids on neuroinflammation. However, to the best of our knowledge, there is no review that specifically summarizes the protective effects of flavonoids on ICH and SAH; therefore, the present review summarized the protective effects of flavonoids on ICH and SAH and the mechanisms by which they exert protective effects in detail, which provides a reference for the treatment of ICH and SAH by flavonoids. The current review discussed the challenges of flavonoids towards clinical practice, the limitations of current researches on the protective effects of flavonoids in ICH and SAH and the beneficial directions for future research on flavonoids in ICH and SAH. In our opinion, flavonoids are a class of compounds with potential, and it is hypothesized that their therapeutic potential for ICH and SAH can be more and more fully explored in future studies.
Acknowledgements
Not applicable.
Funding
Funding: This work was partially supported by the National Natural Science Foundation of China (grant no. 81973547).
Availability of data and materials
Not applicable.
Authors' contributions
HD designed and drafted the manuscript. HD, XG, HL and JG revised the manuscript. LZ conceived and designed the whole project. All authors read and approved the final manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Magid-Bernstein J, Girard R, Polster S, Srinath A, Romanos S, Awad IA and Sansing LH: Cerebral hemorrhage: Pathophysiology, treatment, and future directions. Circ Res. 130:1204–1229. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Feigin VL, Lawes CM, Bennett DA, Barker-Collo SL and Parag V: Worldwide stroke incidence and early case fatality reported in 56 population-based studies: A systematic review. Lancet Neurol. 8:355–369. 2009.PubMed/NCBI View Article : Google Scholar | |
|
van Asch CJ, Luitse MJ, Rinkel GJ, van der Tweel I, Algra A and Klijn CJ: Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: A systematic review and meta-analysis. Lancet Neurol. 9:167–176. 2010.PubMed/NCBI View Article : Google Scholar | |
|
Chen Y, Chen S, Chang J, Wei J, Feng M and Wang R: Perihematomal edema after intracerebral hemorrhage: An update on pathogenesis, risk factors, and therapeutic advances. Front Immunol. 12(740632)2021.PubMed/NCBI View Article : Google Scholar | |
|
Xiao L, Zheng H, Li J, Wang Q and Sun H: Neuroinflammation mediated by NLRP3 inflammasome after intracerebral hemorrhage and potential therapeutic targets. Mol Neurobiol. 57:5130–5149. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Keep RF, Hua Y and Xi G: Intracerebral haemorrhage: Mechanisms of injury and therapeutic targets. Lancet Neurol. 11:720–731. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Wan Y, Holste KG, Hua Y, Keep RF and Xi G: Brain edema formation and therapy after intracerebral hemorrhage. Neurobiol Dis. 176(105948)2023.PubMed/NCBI View Article : Google Scholar | |
|
Muehlschlegel S: Subarachnoid hemorrhage. Continuum (Minneap Minn). 24:1623–1657. 2018.PubMed/NCBI View Article : Google Scholar | |
|
Claassen J and Park S: Spontaneous subarachnoid haemorrhage. Lancet. 400:846–862. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Sveinsson ÓÁ, Ólafsson IH, Kjartansson Ó and Valdimarsson EM: Spontaneous subarachnoid haemorrhage-review. Laeknabladid. 97:355–362. 2011.PubMed/NCBI View Article : Google Scholar : (In Icelandic). | |
|
Lucke-Wold B, Logsdon A, Manoranjan B, Turner RC, McConnell E, Vates GE, Huber JD, Rosen CL and Simard JM: Aneurysmal subarachnoid hemorrhage and neuroinflammation: A comprehensive review. Int J Mol Sci. 17(497)2016.PubMed/NCBI View Article : Google Scholar | |
|
van Gijn J, Kerr RS and Rinkel GJ: Subarachnoid haemorrhage. Lancet. 369:306–318. 2007.PubMed/NCBI View Article : Google Scholar | |
|
Dilli E: Thunderclap Headache. Curr Neurol Neurosci Rep. 14(437)2014.PubMed/NCBI View Article : Google Scholar | |
|
Hao G, Conzen-Dilger C, Schmidt TP, Harder E, Schöps M, Clauser JC, Schubert GA and Lindauer U: Effect of isolated intracranial hypertension on cerebral perfusion within the phase of primary disturbances after subarachnoid hemorrhage in rats. Front Cell Neurosci. 17(1115385)2023.PubMed/NCBI View Article : Google Scholar | |
|
Lynch DG, Shah KA, Powell K, Wadolowski S, Tambo W, Strohl JJ, Unadkat P, Eidelberg D, Huerta PT and Li C: Neurobehavioral impairments predict specific cerebral damage in rat model of subarachnoid hemorrhage. Transl Stroke Res: Jul 26, 2023 (Epub ahead of print). | |
|
Ciurea AV, Palade C, Voinescu D and Nica DA: Subarachnoid hemorrhage and cerebral vasospasm-literature review. J Med Life. 6:120–125. 2013.PubMed/NCBI | |
|
Lauzier DC, Jayaraman K, Yuan JY, Diwan D, Vellimana AK, Osbun JW, Chatterjee AR, Athiraman U, Dhar R and Zipfel GJ: Early brain injury after subarachnoid hemorrhage: Incidence and mechanisms. Stroke. 54:1426–1440. 2023.PubMed/NCBI View Article : Google Scholar | |
|
Yuan B, Zhao XD, Shen JD, Chen SJ, Huang HY, Zhou XM, Han YL, Zhou LJ, Lu XJ and Wu Q: Activation of SIRT1 alleviates ferroptosis in the early brain injury after subarachnoid hemorrhage. Oxid Med Cell Longev. 2022(9069825)2022.PubMed/NCBI View Article : Google Scholar | |
|
Zhang Z, Fang Y, Lenahan C and Chen S: The role of immune inflammation in aneurysmal subarachnoid hemorrhage. Exp Neurol. 336(113535)2021.PubMed/NCBI View Article : Google Scholar | |
|
Clower BR, Yamamoto Y, Cain L, Haines DE and Smith RR: Endothelial injury following experimental subarachnoid hemorrhage in rats: Effects on brain blood flow. Anat Rec. 240:104–114. 1994.PubMed/NCBI View Article : Google Scholar | |
|
Chen Y, Peng F, Xing Z, Chen J, Peng C and Li D: Beneficial effects of natural flavonoids on neuroinflammation. Front Immunol. 13(1006434)2022.PubMed/NCBI View Article : Google Scholar | |
|
Santos-Buelga C and Feliciano AS: Flavonoids: From structure to health issues. Molecules. 22(477)2017.PubMed/NCBI View Article : Google Scholar | |
|
Liu T, Su K, Cai W, Ao H and Li M: Therapeutic potential of puerarin against cerebral diseases: From bench to bedside. Eur J Pharmacol. 953(175695)2023.PubMed/NCBI View Article : Google Scholar | |
|
Zeng J, Zheng S, Chen Y, Qu Y, Xie J, Hong E, Lv H, Ding R, Feng L and Xie Z: Puerarin attenuates intracerebral hemorrhage-induced early brain injury possibly by PI3K/Akt signal activation-mediated suppression of NF-κB pathway. J Cell Mol Med. 25:7809–7824. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Franza L, Carusi V, Nucera E and Pandolfi F: Luteolin, inflammation and cancer: Special emphasis on gut microbiota. Biofactors. 47:181–189. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Tan X, Yang Y, Xu J, Zhang P, Deng R, Mao Y, He J, Chen Y, Zhang Y, Ding J, et al: Luteolin exerts neuroprotection via modulation of the p62/Keap1/Nrf2 pathway in intracerebral hemorrhage. Front Pharmacol. 10(1551)2020.PubMed/NCBI View Article : Google Scholar | |
|
Sivandzade F, Prasad S, Bhalerao A and Cucullo L: NRF2 and NF-κB interplay in cerebrovascular and neurodegenerative disorders: Molecular mechanisms and possible therapeutic approaches. Redox Biol. 21(101059)2019.PubMed/NCBI View Article : Google Scholar | |
|
Yang Y, Tan X, Xu J, Wang T, Liang T, Xu X, Ma C, Xu Z, Wang W, Li H, et al: Luteolin alleviates neuroinflammation via downregulating the TLR4/TRAF6/NF-κB pathway after intracerebral hemorrhage. Biomed Pharmacother. 126(110044)2020.PubMed/NCBI View Article : Google Scholar | |
|
Yu M, Qi B, Xiaoxiang W, Xu J and Liu X: Baicalein increases cisplatin sensitivity of A549 lung adenocarcinoma cells via PI3K/Akt/NF-κB pathway. Biomed Pharmacother. 90:677–685. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Wei N, Wei Y, Li B and Pang L: Baicalein promotes neuronal and behavioral recovery after intracerebral hemorrhage via suppressing apoptosis, oxidative stress and neuroinflammation. Neurochem Res. 42:1345–1353. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Masomi-Bornwasser J, Kurz E, Frenz C, Schmitt J, Wesp DMA, König J, Lotz J, Ringel F, Kerz T, Krenzlin H and Keric N: The influence of oxidative stress on neurological outcomes in spontaneous intracerebral hemorrhage. Biomolecules. 11(1615)2021.PubMed/NCBI View Article : Google Scholar | |
|
Chen X, Zhou Y, Wang S and Wang W: Mechanism of baicalein in brain injury after intracerebral hemorrhage by inhibiting the ROS/NLRP3 inflammasome pathway. Inflammation. 45:590–602. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Gu L, Sun M, Li R, Zhang X, Tao Y, Yuan Y, Luo X and Xie Z: Didymin suppresses microglia pyroptosis and neuroinflammation through the Asc/caspase-1/GSDMD pathway following experimental intracerebral hemorrhage. Front Immunol. 13(810582)2022.PubMed/NCBI View Article : Google Scholar | |
|
Di Petrillo A, Orrù G, Fais A and Fantini MC: Quercetin and its derivates as antiviral potentials: A comprehensive review. Phytother Res. 36:266–278. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Zhang Y, Yi B, Ma J, Zhang L, Zhang H, Yang Y and Dai Y: Quercetin promotes neuronal and behavioral recovery by suppressing inflammatory response and apoptosis in a rat model of intracerebral hemorrhage. Neurochem Res. 40:195–203. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Fu Y, Xu B, Huang S, Luo X, Deng XL, Luo S, Liu C, Wang Q, Chen JY and Zhou L: Baicalin prevents LPS-induced activation of TLR4/NF-κB p65 pathway and inflammation in mice via inhibiting the expression of CD14. Acta Pharmacol Sin. 42:88–96. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Guo LT, Wang SQ, Su J, Xu LX, Ji ZY, Zhang RY, Zhao QW, Ma ZQ, Deng XY and Ma SP: Baicalin ameliorates neuroinflammation-induced depressive-like behavior through inhibition of toll-like receptor 4 expression via the PI3K/AKT/FoxO1 pathway. J Neuroinflammation. 16(95)2019.PubMed/NCBI View Article : Google Scholar | |
|
Zhou QB, Jia Q, Zhang Y, Li LY, Chi ZF and Liu P: Effects of baicalin on protease-activated receptor-1 expression and brain injury in a rat model of intracerebral hemorrhage. Chin J Physiol. 55:219–226. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Zeng J, Chen Y, Ding R, Feng L, Fu Z, Yang S, Deng X, Xie Z and Zheng S: Isoliquiritigenin alleviates early brain injury after experimental intracerebral hemorrhage via suppressing ROS- and/or NF-κB-mediated NLRP3 inflammasome activation by promoting Nrf2 antioxidant pathway. J Neuroinflammation. 14(119)2017.PubMed/NCBI View Article : Google Scholar | |
|
Chen Z, Wang C, Liu Y, Liang X, Yang C, Zhang X and Li X: Protective effects of medicinal plant breviscapine on postcerebral hemorrhage in rats. J Integr Neurosci. 19:101–109. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Lan X, Han X, Li Q, Li Q, Gao Y, Cheng T, Wan J, Zhu W and Wang J: Pinocembrin protects hemorrhagic brain primarily by inhibiting toll-like receptor 4 and reducing M1 phenotype microglia. Brain Behav Immun. 61:326–339. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Fei X, Chen C, Kai S, Fu X, Man W, Ding B, Wang C and Xu R: Eupatilin attenuates the inflammatory response induced by intracerebral hemorrhage through the TLR4/MyD88 pathway. Int Immunopharmacol. 76(105837)2019.PubMed/NCBI View Article : Google Scholar | |
|
Chen C, Yao L, Cui J and Liu B: Fisetin protects against intracerebral hemorrhage-induced neuroinflammation in aged mice. Cerebrovasc Dis. 45:154–161. 2018.PubMed/NCBI View Article : Google Scholar | |
|
Singh N, Bansal Y, Bhandari R, Marwaha L, Singh R, Chopra K and Kuhad A: Naringin reverses neurobehavioral and biochemical alterations in intracerebroventricular collagenase-induced intracerebral hemorrhage in rats. Pharmacology. 100:172–187. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Chen C, Cui J, Ji X and Yao L: Neuroprotective functions of calycosin against intracerebral hemorrhage-induced oxidative stress and neuroinflammation. Future Med Chem. 12:583–592. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Gao Y and Dong Z: Protective effect of procyanidins on experimental rats with intracerebral hemorrhage. Zhongguo Zhong Yao Za Zhi. 34:3078–3081. 2009.PubMed/NCBI(In Chinese). | |
|
Pyrzynska K: Hesperidin: A review on extraction methods, stability and biological activities. Nutrients. 14(2387)2022.PubMed/NCBI View Article : Google Scholar | |
|
Qin Z, Chen L, Liu M, Tan H and Zheng L: Hesperidin reduces adverse symptomatic intracerebral hemorrhage by promoting TGF-β1 for treating ischemic stroke using tissue plasminogen activator. Neurol Sci. 41:139–147. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Dumont AS, Dumont RJ, Chow MM, Lin CL, Calisaneller T, Ley KF, Kassell NF and Lee KS: Cerebral vasospasm after subarachnoid hemorrhage: Putative role of inflammation. Neurosurgery. 53:123–135. 2003.PubMed/NCBI View Article : Google Scholar | |
|
Erdi F, Keskin F, Esen H, Kaya B, Feyzioglu B, Kilinc I, Karatas Y, Cuce G and Kalkan E: Telmisartan ameliorates oxidative stress and subarachnoid haemorrhage-induced cerebral vasospasm. Neurol Res. 38:224–231. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Xu W, Li T, Gao L, Zheng J, Yan J, Zhang J and Shao A: Apelin-13/APJ system attenuates early brain injury via suppression of endoplasmic reticulum stress-associated TXNIP/NLRP3 inflammasome activation and oxidative stress in a AMPK-dependent manner after subarachnoid hemorrhage in rats. J Neuroinflammation. 16(247)2019.PubMed/NCBI View Article : Google Scholar | |
|
Kuo CP, Wen LL, Chen CM, Huh B, Cherng CH, Wong CS, Liaw WJ, Yeh CC, Lin BF and Wu CT: Attenuation of neurological injury with early baicalein treatment following subarachnoid hemorrhage in rats. J Neurosurg. 119:1028–1037. 2013.PubMed/NCBI View Article : Google Scholar | |
|
Hao G, Dong Y, Huo R, Wen K, Zhang Y and Liang G: Rutin Inhibits neuroinflammation and provides neuroprotection in an experimental rat model of subarachnoid hemorrhage, possibly through suppressing the RAGE-NF-κB inflammatory signaling pathway. Neurochem Res. 41:1496–1504. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Zhang T, Su J, Guo B, Wang K, Li X and Liang G: Apigenin protects blood-brain barrier and ameliorates early brain injury by inhibiting TLR4-mediated inflammatory pathway in subarachnoid hemorrhage rats. Int Immunopharmacol. 28:79–87. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Shi X, Fu Y, Zhang S, Ding H and Chen J: Baicalin attenuates subarachnoid hemorrhagic brain injury by modulating blood-brain barrier disruption, inflammation, and oxidative damage in mice. Oxid Med Cell Longev. 2017(1401790)2017.PubMed/NCBI View Article : Google Scholar | |
|
Zhang H, Tu X, Song S, Liang R and Shi S: Baicalin reduces early brain injury after subarachnoid hemorrhage in rats. Chin J Integr Med. 26:510–518. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Gül Ş, Aydoğmuş E, Bahadir B, Büyükuysal MÇ and Güven B: Neuroprotective effects of quercetin on cerebral vasospasm following experimental subarachnoid haemorrhage in rats. Turk J Med Sci. 50:1106–1110. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Dong YS, Wang JL, Feng DY, Qin HZ, Wen H, Yin ZM, Gao GD and Li C: Protective effect of quercetin against oxidative stress and brain edema in an experimental rat model of subarachnoid hemorrhage. Int J Med Sci. 11:282–290. 2014.PubMed/NCBI View Article : Google Scholar | |
|
Tekiner A, Yilmaz MB, Bolat E, Goker T, Sargon MF and Arat A: The therapeutic value of proanthocyanidin in experimental cerebral vasospasm following subarachnoid hemorrhage. Turk Neurosurg. 24:885–890. 2014.PubMed/NCBI View Article : Google Scholar | |
|
Zhang ZH, Liu JQ, Hu CD, Zhao XT, Qin FY, Zhuang Z and Zhang XS: Luteolin confers cerebroprotection after subarachnoid hemorrhage by suppression of NLPR3 inflammasome activation through Nrf2-dependent pathway. Oxid Med Cell Longev. 2021(5838101)2021.PubMed/NCBI View Article : Google Scholar | |
|
Zhang Y, Yang X, Ge X and Zhang F: Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice. Biomed Pharmacother. 109:726–733. 2019.PubMed/NCBI View Article : Google Scholar | |
|
Zeng Y, Fang Z, Lai J, Wu Z, Lin W, Yao H, Hu W, Chen J, Guo X and Chen X: Activation of sirtuin-1 by pinocembrin treatment contributes to reduced early brain injury after subarachnoid hemorrhage. Oxid Med Cell Longev. 2022(2242833)2022.PubMed/NCBI View Article : Google Scholar | |
|
Xu W, Yan J, Ocak U, Lenahan C, Shao A, Tang J, Zhang J and Zhang JH: Melanocortin 1 receptor attenuates early brain injury following subarachnoid hemorrhage by controlling mitochondrial metabolism via AMPK/SIRT1/PGC-1α pathway in rats. Theranostics. 11:522–539. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Li Q, Chen Y, Zhang X, Zuo S, Ge H, Chen Y, Liu X, Zhang JH, Ruan H and Feng H: Scutellarin attenuates vasospasm through the Erk5-KLF2-eNOS pathway after subarachnoid hemorrhage in rats. J Clin Neurosci. 34:264–270. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Sun Y, Chen P, Zhai B, Zhang M, Xiang Y, Fang J, Xu S, Gao Y, Chen X, Sui X and Li G: The emerging role of ferroptosis in inflammation. Biomed Pharmacother. 127(110108)2020.PubMed/NCBI View Article : Google Scholar | |
|
Wang F, He J, Xing R, Sha T and Sun B: Molecular mechanisms of ferroptosis and their role in inflammation. Int Rev Immunol. 42:71–81. 2023.PubMed/NCBI View Article : Google Scholar | |
|
Li J, Cao F, Yin HL, Huang ZJ, Lin ZT, Mao N, Sun B and Wang G: Ferroptosis: Past, present and future. Cell Death Dis. 11(88)2020.PubMed/NCBI View Article : Google Scholar | |
|
Yu Y, Yan Y, Niu F, Wang Y, Chen X, Su G, Liu Y, Zhao X, Qian L, Liu P and Xiong Y: Ferroptosis: A cell death connecting oxidative stress, inflammation and cardiovascular diseases. Cell Death Discov. 7(193)2021.PubMed/NCBI View Article : Google Scholar | |
|
Gao S, Zhou L, Lu J, Fang Y, Wu H, Xu W, Pan Y, Wang J, Wang X, Zhang J and Shao A: Cepharanthine attenuates early brain injury after subarachnoid hemorrhage in mice via inhibiting 15-lipoxygenase-1-mediated microglia and endothelial cell ferroptosis. Oxid Med Cell Longev. 2022(4295208)2022.PubMed/NCBI View Article : Google Scholar | |
|
Zheng B, Zhou X, Pang L, Che Y and Qi X: Baicalin suppresses autophagy-dependent ferroptosis in early brain injury after subarachnoid hemorrhage. Bioengineered. 12:7794–7804. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Chen D, Chen JJ, Yin Q, Guan JH and Liu YH: Role of ERK1/2 and vascular cell proliferation in cerebral vasospasm after experimental subarachnoid hemorrhage. Acta Neurochir (Wien). 151:1127–1134. 2009.PubMed/NCBI View Article : Google Scholar | |
|
Curson JEB, Liu L, Luo L, Muusse TW, Lucas RM, Gunther KS, Vajjhala PR, Abrol R, Jones A, Kapetanovic R, et al: TLR4 phosphorylation at tyrosine 672 activates the ERK/c-FOS signaling module for LPS-induced cytokine responses in macrophages. Eur J Immunol. 53(e2250056)2023.PubMed/NCBI View Article : Google Scholar | |
|
Lin CW, Chen PN, Chen MK, Yang WE, Tang CH, Yang SF and Hsieh YS: Kaempferol reduces matrix metalloproteinase-2 expression by down-regulating ERK1/2 and the activator protein-1 signaling pathways in oral cancer cells. PLoS One. 8(e80883)2013.PubMed/NCBI View Article : Google Scholar | |
|
Zong J, Zhang DP, Zhou H, Bian ZY, Deng W, Dai J, Yuan Y, Gan HW, Guo HP and Tang QZ: Baicalein protects against cardiac hypertrophy through blocking MEK-ERK1/2 signaling. J Cell Biochem. 114:1058–1065. 2013.PubMed/NCBI View Article : Google Scholar | |
|
Cheng Y, Zhang Z, Tang H, Chen B, Cai Y, Wei Y, Zhao W, Wu ZB and Shang H: Mitochondrial inhibitor rotenone triggers and enhances neuronal ferroptosis following intracerebral hemorrhage. ACS Chem Neurosci. 14:1071–1079. 2023.PubMed/NCBI View Article : Google Scholar | |
|
Li N, Ragheb K, Lawler G, Sturgis J, Rajwa B, Melendez JA and Robinson JP: Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J Biol Chem. 278:8516–8525. 2003.PubMed/NCBI View Article : Google Scholar | |
|
Li K, Liang Y, Cheng A, Wang Q, Li Y, Wei H, Zhou C and Wan X: Antiviral properties of baicalin: A concise review. Rev Bras Farmacogn. 31:408–419. 2021.PubMed/NCBI View Article : Google Scholar | |
|
Yao C, Dai S, Wang C, Fu K, Wu R, Zhao X, Yao Y and Li Y: Luteolin as a potential hepatoprotective drug: Molecular mechanisms and treatment strategies. Biomed Pharmacother. 167(115464)2023.PubMed/NCBI View Article : Google Scholar | |
|
Choi EJ, Lee BH, Lee K and Chee KM: Long-term combined administration of quercetin and daidzein inhibits quercetin-induced suppression of glutathione antioxidant defenses. Food Chem Toxicol. 43:793–798. 2005.PubMed/NCBI View Article : Google Scholar | |
|
Di Lorenzo C, Colombo F, Biella S, Stockley C and Restani P: Polyphenols and human health: The role of bioavailability. Nutrients. 13(273)2021.PubMed/NCBI View Article : Google Scholar | |
|
Galho AR, Cordeiro MF, Ribeiro SA, Marques MS, Antunes MF, Luz DC, Hädrich G, Muccillo-Baisch AL, Barros DM, Lima JV, et al: Protective role of free and quercetin-loaded nanoemulsion against damage induced by intracerebral haemorrhage in rats. Nanotechnology. 27(175101)2016.PubMed/NCBI View Article : Google Scholar | |
|
Waki T, Nakanishi I, Matsumoto K, Kitajima J, Chikuma T and Kobayashi S: Key role of chemical hardness to compare 2,2-diphenyl-1-picrylhydrazyl radical scavenging power of flavone and flavonol O-glycoside and C-glycoside derivatives. Chem Pharm Bull (Tokyo). 60:37–44. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Cassidy A, O'Reilly ÉJ, Kay C, Sampson L, Franz M, Forman JP, Curhan G and Rimm EB: Habitual intake of flavonoid subclasses and incident hypertension in adults. Am J Clin Nutr. 93:338–347. 2011.PubMed/NCBI View Article : Google Scholar | |
|
Elijovich L, Patel PV and Hemphill JC III: Intracerebral hemorrhage. Semin Neurol. 28:657–667. 2008.PubMed/NCBI View Article : Google Scholar | |
|
Sung B, Chung HY and Kim ND: Role of apigenin in cancer prevention via the induction of apoptosis and autophagy. J Cancer Prev. 21:216–226. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Ganeshpurkar A and Saluja AK: The pharmacological potential of rutin. Saudi Pharm J. 25:149–164. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Jäger A and Saaby L: Flavonoids and the CNS. Molecules. 16:1471–1485. 2011.PubMed/NCBI View Article : Google Scholar | |
|
Parrella E, Gussago C, Porrini V, Benarese M and Pizzi M: From preclinical stroke models to humans: Polyphenols in the prevention and treatment of stroke. Nutrients. 13(85)2020.PubMed/NCBI View Article : Google Scholar |