Serum anti‑KIAA0513 antibody as a common biomarker for mortal atherosclerotic and cancerous diseases
- Authors:
- Published online on: June 19, 2024 https://doi.org/10.3892/mi.2024.169
- Article Number: 45
-
Copyright : © Hiwasa et al. This is an open access article distributed under the terms of Creative Commons Attribution License [CC BY 4.0].
Abstract
Introduction
In recent years, various disease biomarkers have been discovered, and the development of simple blood tests is underway to determine the pathological condition, predict the onset of disease and its prognosis, and identify preventive/therapeutic targets. In terms of biomarker species, studies have reported enzyme, antigen, and, in recent years, nucleotide markers (1-3). However, there are still some reports on antibody markers, which include heat-shock 60-kD protein 1(4), replication protein A2(5), programmed cell death 11(6), metalloproteinase 1, chromobox homolog 1, chromobox homolog 5(7), DnaJ heat shock protein family (Hsp40) member C2(8), adaptor-related protein complex 3 subunit delta 1(9), serpin peptidase inhibitor, clade E member 1(10), death-inducer obliterator 1, cleavage and polyadenylation specificity factor 2, forkhead box J2(11) and thiosulfate sulfurtransferase-like domain-containing 2(12) for acute ischemic stroke (AIS); ATPase, Ca++ transporting, plasma membrane 4(10), bone morphogenetic protein 1 (3,13), deoxyhypusine synthase (14), SH3 domain-binding protein 5(15), prolyl carboxypeptidase (16), low-density lipoprotein receptor-related protein-associated protein 1(17) and additional sex combs-like 2(18) for atherosclerosis; nardilysin (19) for acute cardiac syndrome; and insulin (20), glutamic acid decarboxylase (21), adiponectin (22) and growth arrest and DNA-damage-inducible gene 34 (23,24), and proprotein convertase subtilisin/kexin type 9(25) for diabetes mellitus (DM).
The anti-p53 antibody is a typical antibody biomarker for cancer that has been used in clinical practical for diagnosing, monitoring and predicting the prognosis of esophageal cancer (EC) and head and neck cancer (26,27). Further application of the serological identification of antigens by cDNA expression cloning and the protein array method have identified autoantibodies against tumor-associated calcium signal transducer 2(28), solute carrier family 2/facilitated glucose transporter, member 1(29), tripartite motif-containing 21(30), myomegalin (31), makorin 1(32), esophageal carcinoma SEREX antigen (33), cyclin L2(34), cofilin, β-actin (35) and WD repeat-containing protein 1(36) for EC; FBP-interacting repressor for colorectal cancer (CRC) (37) and gastric cancer (GC) (38); SH3 domain, GRB2-like 1(39) and filamin C (40) for glioma; EP300-interacting inhibitor of differentiation 3 for non-functional pancreatic neuroendocrine tumors (41); wingless-type MMTV integration site family, member 7 for biliary cancer (42); and coatomer protein complex subunit epsilon (43), differential screening-selected gene aberrant in neuroblastoma (44), and sorting nexins 16(45) for obstructive sleep apnea syndrome (OSAS). Here, we report on serum antibodies against KIAA0513 (s-KIAA0513-Ab) as a broad-spectrum biomarker applicable to atherosclerosis-related diseases such as ischemic stroke, cardiovascular disease (CVD), chronic kidney disease (CKD), DM and solid cancers.
Materials and methods
Patients and control sera
The present study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Local Ethical Review Board of the Chiba University, Graduate School of Medicine (Chiba, Japan), as well as by the review boards of the participating hospitals (approval no. 2018-320). The Ethics Committee of Toho University, Graduate School of Medicine, Tokyo, Japan (No. A18103_A17052_A16035_A16001_26095_25024_24038_22047_22047) and Port Square Kashiwado Clinic, Kashiwado Memorial Foundation, China, Japan (approval no. 2012-001) also approved the study protocol. Sera were collected from patients who had provided written informed consent. Each serum sample was centrifuged at 3,000 x g for 10 min at 4˚C, and the supernatant was stored at -80˚C until use.
Serum samples collected from patients with AIS, transient ischemic attack (TIA), asymptomatic ischemic stroke (Asympt-CI), chronic-phase cerebral infarction (cCI), and deep and subcortical white matter hyperintensity (DSWMH) were obtained from Chiba Prefectural Sawara Hospital. The stroke subtypes were determined according to the criteria of the Trial of Org 10172 in the Acute Stroke Treatment classification system (46), and large-artery atherosclerosis and small-artery occlusion (lacune) were included as AIS and ischemic stroke. Samples from patients with DM, CVD and OSAS were obtained from the Chiba University Hospital. CVD included acute myocardial infarction (AMI) and unstable angina. The serum samples of patients with AIS, TIA and AMI were obtained within 2 weeks following disease onset. Samples collected from patients with CKD were obtained from the Kumamoto cohort (47,48), whereas those collected from patients with EC, GC, CRC, lung cancer (LC), and breast cancer (BC) were obtained from the Department of Surgery, Toho University Hospital. Serum samples from healthy donors (HDs) were obtained from Chiba University, Port Square Kashiwado Clinic, the National Hospital Organization, Shimoshizu Hospital (Yotsukaido, Japan), and Chiba Prefectural Sawara Hospital (Katori, Japan). For comparisons with TIA and AIS, serum samples from HDs were selected from patients who exhibited no abnormalities in cranial magnetic resonance imaging.
ProtoArray® screening
The initial screening was performed using ProtoArray® Human Protein Microarrays v4.0 (Thermo Fisher Scientific, Inc.), which were loaded with 9,480 species of proteins, as previously described (11,14,15). In total, 20 serum samples (10 each from the patients and HDs) were employed to detect antigens specifically recognized by IgG antibodies in the patient sera. The complete results from the ProtoArray® screening are presented in Table SI.
Expression and purification of KIAA0513 protein
A sequence encoding cDNA of the isoform c of human KIAA0513 (accession no. NP_001284695.1) was cloned into pGEX-6P (Cytiva). The expression of the cDNA product was induced by treating Escherichia coli (E. coli) KRX (Promega Corp.) cells harboring the pGEX-6P-KIAA0513 and pMINOR with 0.5 mM isopropyl-β-D-thiogalactoside (FUJIFILM Wako Pure Chemical Corporation) at 37˚C for 3 h (49). The cells were lysed by sonication in phosphate-buffered saline (FUJIFILM Wako Pure Chemical Corporation) containing 1% Triton X-100 (FUJIFILM Wako Pure Chemical Corporation) and 2 mM dithiothreitol (FUJIFILM Wako Pure Chemical Corporation). The glutathione S-transferase (GST)-fused proteins were specifically bound to Glutathione-Sepharose 4 Fast Flow medium (Cytiva) and HiPrep 26/10 Desalting column (1 ml) (Cytiva), followed by washing with 30 ml of phosphate-buffered saline. GST-KIAA0513 protein was eluted with 10 mM glutathione (Wako Pure Chemicals) and 2 mM dithiothreitol in phosphate-buffered saline, and concentrated to 3.5 mg/ml in phosphate-buffered saline containing 2 mM dithiothreitol as previously described (49). Protein concentration was determined by Bradford Protein Assay (Bio-Rad Laboratories, Inc.).
Western blot analysis
Purified GST-KIAA0513 and the control GST proteins (0.3 µg/lane) were electrophoresed through sodium dodecyl sulfate-polyacrylamide (11%) gels, followed by directly staining with 0.05% Coomassie Brilliant Blue (Nacalai Tesque) in 50% methanol and 10% acetic acid for 1 h at 25˚C, or blotting onto nitrocellulose membranes (S045A330R, Advantec). The membranes were blocked with 0.1% dry milk (Megmilk Snow Brand Co., Ltd.) in 150 mM NaCl, 20 mM Tris-HCl (pH 7.6) and 0.1% Tween-20 (TBS-T) at 25˚C for 1 h, and then treated with anti-GST (goat, ab6613, Abcam), anti-KIAA0513 (rabbit, HPA012866, Atlas Antibodies) antibodies at a final concentration of 0.2 µg/ml, or the patient sera (1/1,000 fold dilution) at 25˚C overnight. The membranes were washed five times with TBS-T and treated with HRP-conjugated secondary antibodies (HRP-conjugated donkey anti-goat IgG, sc-2020, Santa Cruz Biotechnology, Inc., 1/30,000-fold dilution; HRP-conjugated goat anti-rabbit IgG, 111-035-003, Jackson ImmunoResearch Laboratories, Inc, 1/30,000-fold dilution; HRP-conjugated goat anti-human IgG, A130PD, American Qualex, 1/30,000-fold dilution) at 25˚C for 20 min. Following five washes with TBS-T, the addition of Immobilon (Merck KGaA) produced luminescence, which was detected using LuminoGraph II (Atto Co., Ltd.), as previously described (7,8,11,17).
Amplified luminescence proximity homogeneous assay-linked immunosorbent assay (AlphaLISA)
AlphaLISA was performed in 384-well microtiter plates (white opaque OptiPlate™, Revvity) containing either 2.5 µl of 1:100-diluted serum with 2.5 µl of GST or GST-KIAA0513 proteins (10 µg/ml) in AlphaLISA buffer (25 mM N-2-hydroxyethylpiperazine-N-2-ethane sulfonic acid, pH 7.4, 0.1% casein, 0.5% Triton X-100, 1 mg/ml dextran-500 and 0.05% Proclin-300) (Revvity). The reaction mixture was incubated at room temperature for 6-8 h, followed by the addition of anti-human IgG-conjugated acceptor beads (2.5 µl at 40 µg/ml) and glutathione-conjugated donor beads (2.5 µl at 40 µg/ml), and the mixture was incubated at room temperature in the dark for 1-14 days. The chemical emissions were measured using an EnSpire Alpha microplate reader (Revvity), as previously described (7-11). The specific reactions were calculated by subtracting the emission counts of the GST control from the counts of GST-fused KIAA0513 protein.
Immunohistochemical staining
The formalin-fixed paraffin-embedded EC tissues were sectioned into 4-µm-thick slices, which were deparaffinized, blocked with Hyper Peroxide Block and Protein Block (Rabbit Specific HRP/DAB Detection Kit, Abcam), reacted with primary anti-KIAA0513 antibody (rabbit polyclonal antibodies, HPA012866, Atlas Antibodies) at 0.75 µg/ml for 18 h at 4˚C, incubated with biotinylated anti-rabbit IgG (biotin-conjugated goat anti-rabbit IgG, sc-2040, Santa Cruz Biotechinology, Inc.) at 2 µg/ml for 30 min at 25˚C, and reacted with streptavidin conjugated to horseradish peroxidase reagent (ab7403, Abcam) for 30 min at 25˚C. Finally, the reaction was visualized with a chromogen (diaminobenzidine) in DAB substrate (ab64238, Abcam). The sections were then counterstained with hematoxylin (Mfcd00078111, Merck KGaA) for 30 sec at 25˚C, dehydrated, mounted as previously described (11,17). Photomicrographs were obtained using a light microscope (BA210E, Shimazu) at x100 magnification.
Nested case-control study
A nested case-cohort study was conducted using the aforementioned AlphaLISA detection antibody levels. The present study was nested within the Japan Public Health Center-based Prospective Study (50-52), which involved ~30,000 Japanese individuals aged 40-69 years at a baseline period from 1990-1994 whose plasma samples were stored. The plasma samples employed were from 202 cases of incidental AIS in the cohort that occurred between baseline and 2008 and from 202 controls whose age (within 2 years), sex, date of blood sampling (within 3 months), time since last meal (within 4 h), and study location (Public Health Center area) were matched with those of the cases. A conditional logistic regression model was used to estimate the odds ratios (ORs) and 95% confidence intervals (95% CIs). The study participants were informed of the objectives and methods of the study, and those who answered the questionnaire and donated blood were regarded as having given informed consent to participate.
Statistical analysis
The Mann-Whitney U test was employed to determine the significant differences between two groups and the Kruskal-Wallis test (with the Bonferroni correction applied) was used to evaluate the differences among ≥3 groups. Correlations were analyzed using Spearman's correlation analysis and logistic regression analysis. All the statistical analyses were performed using GraphPad Prism 5 (GraphPad Software, Inc.). The predictive values of the putative disease markers were assessed via a receiver operating characteristic (ROC) curve analysis and determined the sensitivity and specificity. Patient survival was evaluated using the Kaplan-Meier method and compared using the log-rank test. X-tile 3.6.1 software (Yale University, New Haven, CT, USA) (53) was used to determine the optimal cut-off values for discrimination of the survival rates between antibody positive and negative groups. All tests were two-tailed, and P-values <0.05 were considered to indicate statistically significant differences.
Results
Recognition of KIAA0513 by serum components from patients with atherosclerosis
The present study employed a ProtoArray loaded with 9,480 protein species to identify the antigens recognized by antibodies in the sera of patients with atherosclerosis. It was found that KIAA0513 isoform c (Accession no. BC030280.1) reacted with 6 of the 10 serum samples from the patients with atherosclerosis, and with only 1 of the 10 samples from the HDs (Table SI). Subsequently, GST-fused full-length KIAA0513 protein was expressed in E. coli and purified by affinity-chromatography.
Presence of autoantibodies against KIAA0513 in the sera of patients with AIS, TIA, DM, EC, or CC
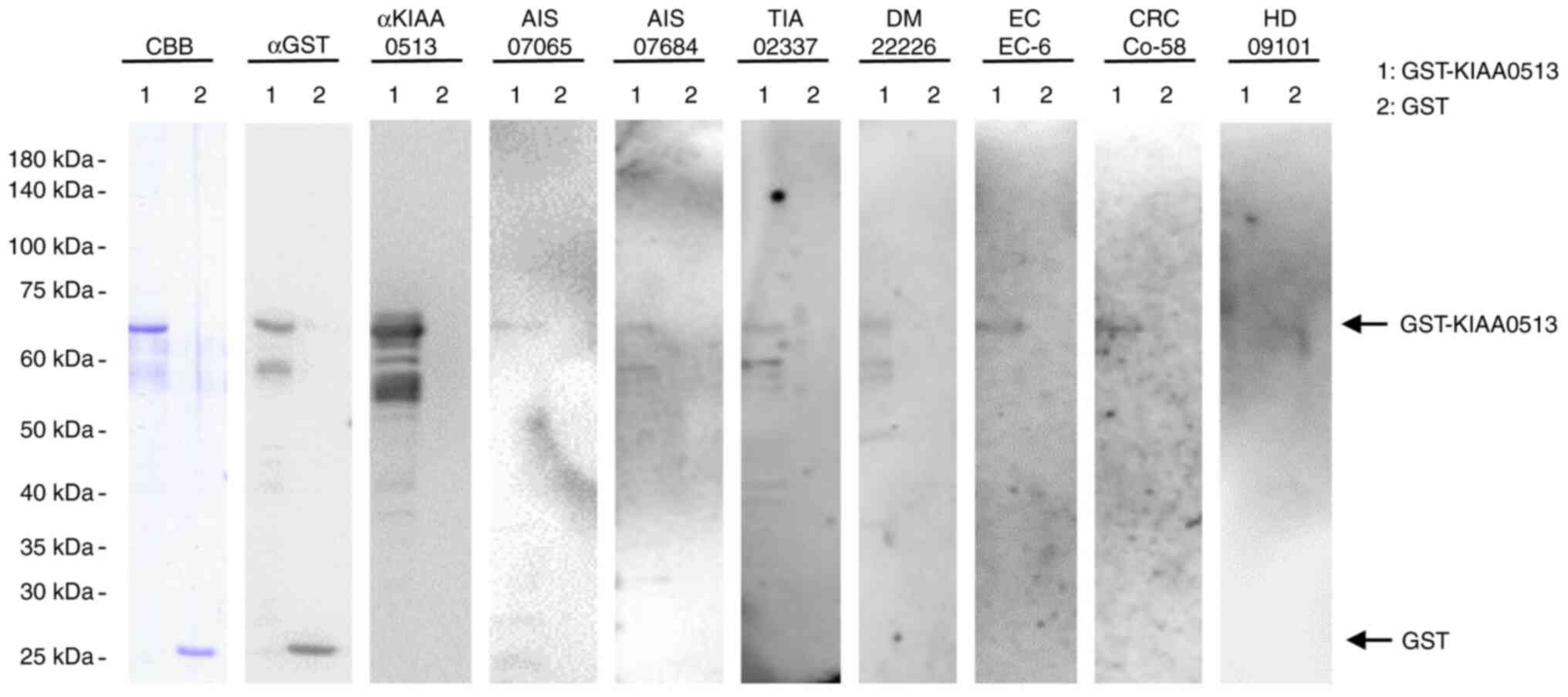
The present study examined the presence of autoantibodies against KIAA0513 in sera using western blot analysis (Fig. 1). GST-KIAA0513 protein reacted with commercial anti-GST and anti-KIAA0513 antibodies, whereas the control, GST, reacted with anti-GST, but not with anti-KIAA0513 antibodies. GST-KIAA0513 protein was also recognized by serum IgG antibodies in the patients with AIS (anonymization nos. #07065 and #070684), TIA (#02337), DM (#22226), EC (#EC-6) and CRC (#Co-58), but not in the HDs (#09101). GST alone exhibited no apparent reaction with any serum from the patients or HDs.
Elevation of s-KIAA0513-Ab levels in the patients with AIS and TIA
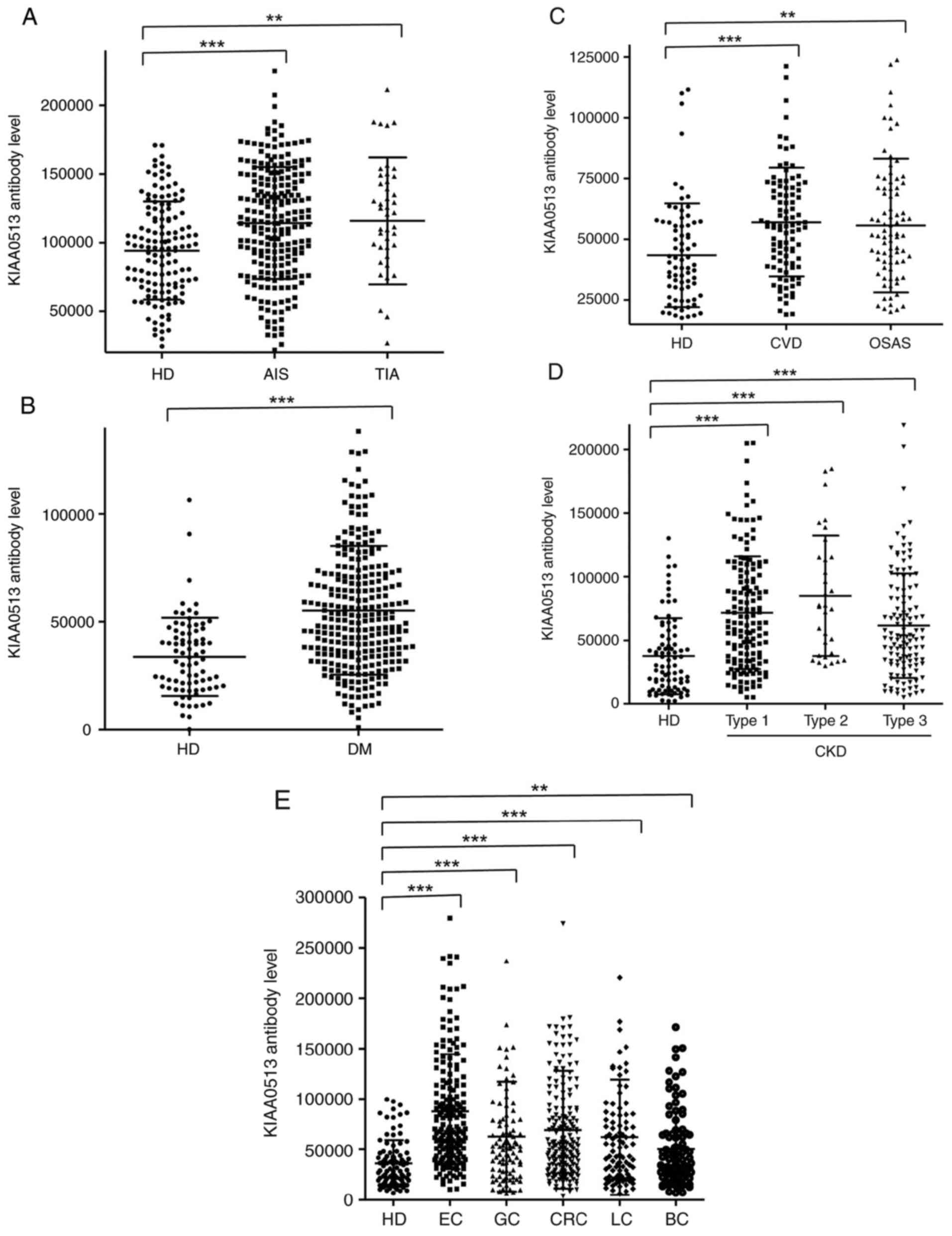
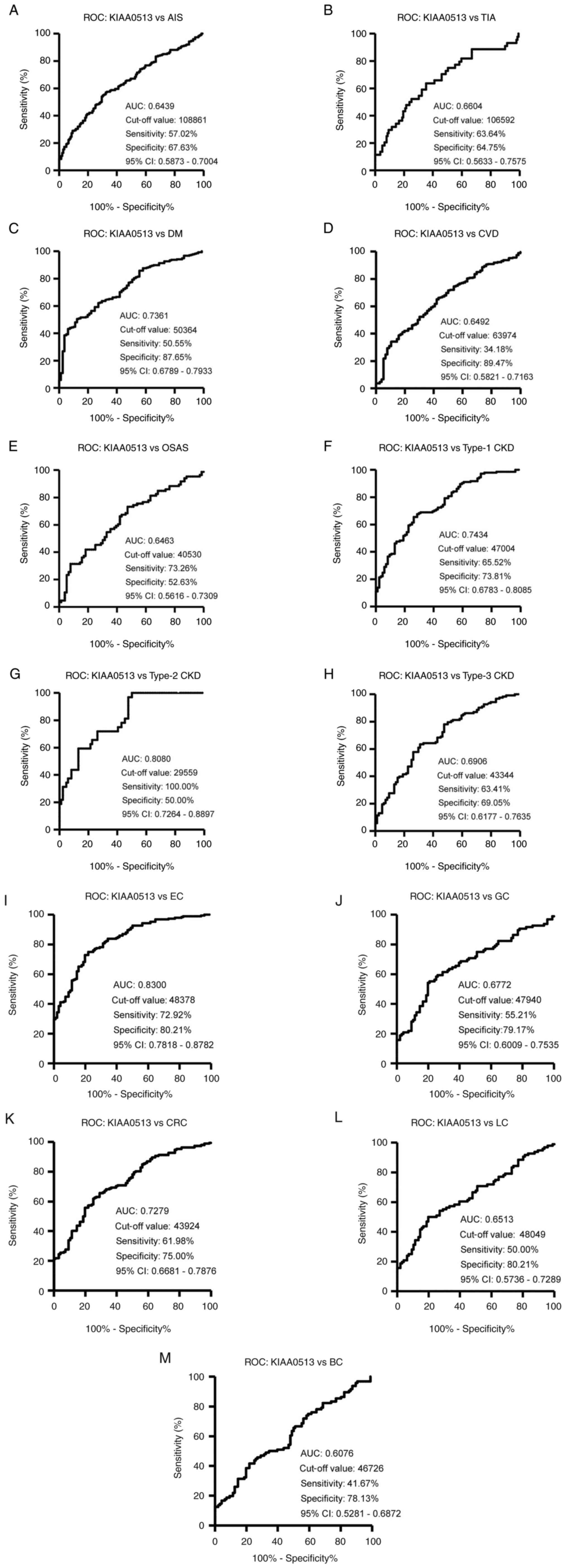
The s-KIAA0513-Ab levels were then examined in the patients with AIS or TIA. Sera from HDs, and patients with AIS and TIA were obtained from the Chiba Prefectural Sawara Hospital. The results of AlphaLISA revealed that the s-KIAA0513-Ab levels were significantly higher in the patients with AIS or TIA than in the HDs (Fig. 2A). Using the cut-off values of the average plus two standard deviations (SDs) of the HD values, the s-KIAA0513-Ab positivity rates for the HDs, patients with AIS, and those with TIA were 0.0, 7.6 and 15.6%, respectively (Table SII). ROC analysis revealed that the areas under the ROC curves (AUCs) of s-KIAA0513-Abs were 0.6439 (95% CI, 0.587-0.700) for AIS (Fig. 3A) and 0.6604 (95% CI, 0.563-0.758) for TIA (Fig. 3B). Thus, TIA (which can be a prodromal stage of AIS) and AIS were equally associated with the s-KIAA0513-Ab marker.
Elevation of serum antibody levels against KIAA0513 in patients with DM
The present study then examined the s-KIAA0513-Ab levels in patients with DM. Serum samples from HDs and patients with DM were obtained from Chiba University and Chiba University Hospital. The s-KIAA0513-Ab levels were significantly higher in the samples from the patients with DM than in those from HDs (Fig. 2B). At a cut-off value equivalent to the average plus two SDs of the HD specimen values, the positive rates of s-KIAA0513-Abs in the HDs and patients with DM were 2.5 and 26.5%, respectively (Table SIII). ROC analysis was performed to evaluate the ability of these antibody markers to indicate the presence of DM. The AUC for s-KIAA0513-Abs was 0.736, yielding a sensitivity and specificity of 50.55 and 87.65%, respectively (Fig. 3C).
Association between s-KIAA0513-Ab levels and CVD and OSAS
Subsequently, the antibody levels in serum samples from patients with CVD obtained from Chiba University Hospital were examined. Given that OSAS is related to atherosclerosis and is associated with a high risk of AIS and CVD (36-39), the present study also examined the sera of patients with OSAS obtained from Chiba University Hospital. Compared with those of the HDs, the s-KIAA0513-Ab levels were significantly higher in the patients with CVD or OSAS (Fig. 2C), although the positive rates in the patients with CVD and those with OSAS were not markedly high (10.3 and 11.6%, respectively) (Table SIV). ROC analysis revealed that the AUCs for CVD and OSAS were 0.649 (95% CI, 0.582-0.716) (Fig. 3D) and 0.646 (95% CI, 0.562-0.731) (Fig. 3E), respectively. Compared with the low P-value (<0.001) of s-KIAA0513-Ab for CVD, the P-value for OSAS was <0.01 (Table SIV), suggesting a weaker association of the s-KIAA0513-Ab marker with OSAS than with CVD.
Elevation of s-KIAA0513-Ab levels in patients with CKD
The present study then examined the antibody levels in the sera of patients with CKD, which is also closely related to atherosclerosis. CKD was divided into three groups as follows: Type 1, diabetic kidney disease; type 2, nephrosclerosis; and type 3, glomerulonephritis. Samples from patients with CKD were obtained from the Kumamoto cohort, and samples from HDs were obtained from Chiba University. Patients from all three CKD groups had significantly higher s-KIAA0513-Ab levels than the HDs (Fig. 2D). The s-KIAA0513-Ab positivity rates in the HDs and patients with types 1, 2 and 3 CKD were 6.1, 29.0, 37.5 and 20.3%, respectively (Table SV), indicating that the highest positive rate was observed in the patients with type 2 CKD. ROC analysis revealed s-KIAA0513-Ab AUCs as high as 0.7434 (95% CI, 0.678-0.809) for type 1 CKD (Fig. 3F), 0.808 (95% CI, 0.726-0.890) for type 2 CKD (Fig. 3G) and 0.691 (95% CI, 0.618-0.764) for type 3 CKD (Fig. 3H).
s-KIAA0513-Ab levels in solid cancer
Given that atherosclerotic diseases are frequently related to cancer with certain common biomarkers being reported (20), the present study examined the serum samples from patients with EC, GC, CRC, LC and BC obtained from Toho University Hospital. The s-KIAA0513-Ab levels were significantly higher in the samples from all patients with cancer than in those from the HDs (Fig. 2E and Table SVI). The highest average value and positive rate of s-KIAA0513-Ab levels were observed for EC. Similarly, the AUC values were highest for EC (0.830), but lowest for BC among the cancers examined (Fig. 3I-M).
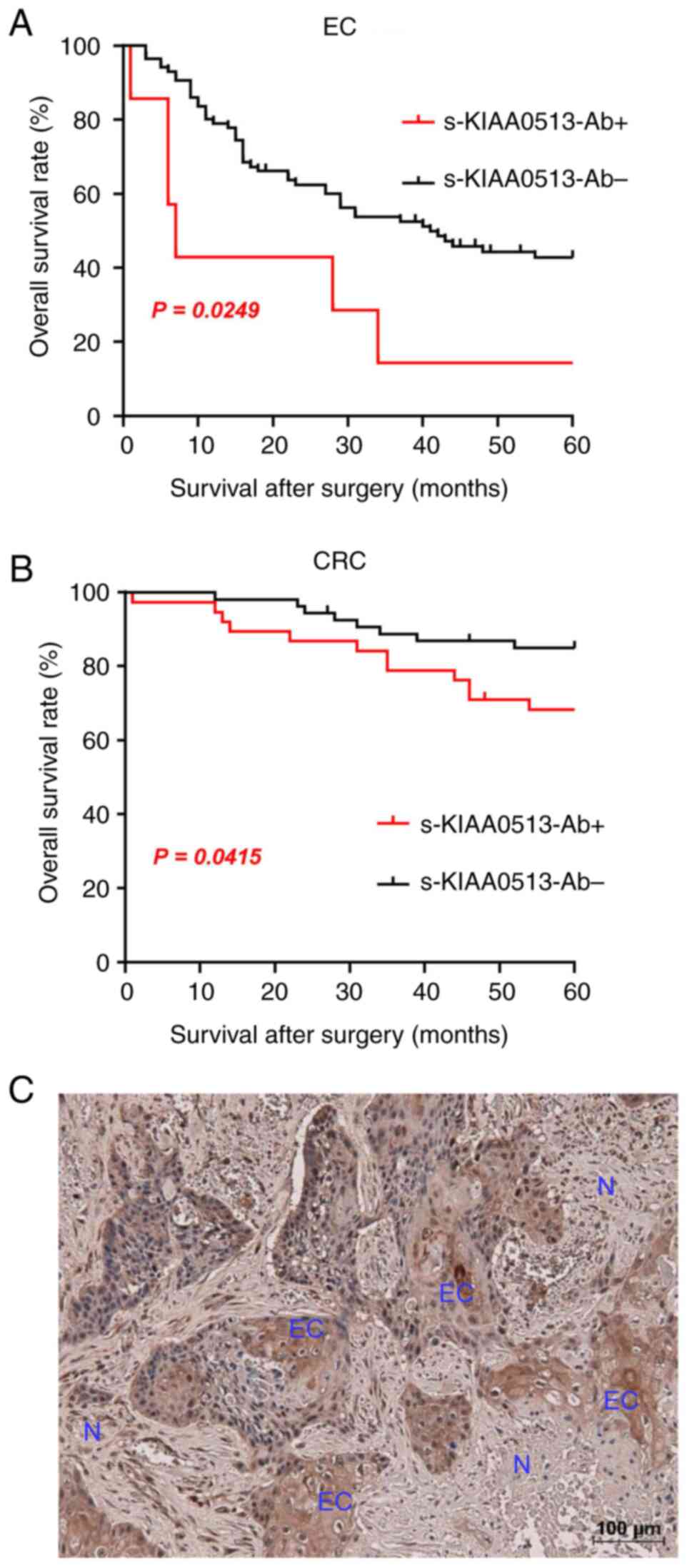
The present study then examined whether the s-KIAA0513-Ab levels are related to the post-operative survival of patients with EC or GC. The s-KIAA0513-Ab levels were divided into the positive and negative groups using the cut-off values obtained using X-tile software (53). The s-KIAA-Ab-positive group presented a more unfavorable prognosis than the negative group in all of EC and CRC (Fig. 4A and B). The X-tile-determined cut-off values are best ones to distinguish the favorable and poor survivals. The cut-off value of EC samples (180,487) was much higher than the average (87,535) (Table SVI), whereas that of CRC (56,879) was lower than the average of CRC (69,308) (Table SVI). Thus, the considerably high levels of s-KIAA-Abs in patients with EC and the moderately high levels in patients with CRC were associated with the prognosis.
The expression levels of KIAA0513 antigenic protein in EC tissues were examined using immunohistochemical staining. A representative example of the staining is illustrated in Fig. 4C. EC tissues were heavily stained by anti-KIAA0513 antibody, whereas surrounding healthy esophageal tissues were not. KIAA0513 protein was localized in the cytoplasm, which is consistent with the findings in a previous study (54). Thus, the high KIAA0513 expression levels may account for some, if not all, of the development of serum KIAA0513-Abs.
Association analysis
An analysis of the association analysis between the s-KIAA0513-Ab levels and participant data was performed using 665 specimens from Chiba Prefectural Sawara Hospital, including 139 specimens from HDs, 225 from patients with AIS, 44 from patients with TIA, 17 from patients with Asympt-CI, 122 from patients with DSWMH, 59 from patients with cCI and 41 from disease controls. The remaining 18 subjects were excluded because they did not have disease information. In this analysis, the Mann-Whitney U test we employed to compare the s-KIAA0513-Ab levels between the male and female participants, with or without DM, hypertension, CVD, dyslipidemia and obesity [body mass index (BMI) ≥25] and with or without smoking and alcohol intake habits. A significant difference in the s-KIAA0513-Ab levels was observed only between the patients with hypertension and those without hypertension (Table I).
Table IAssociation analysis of antibody levels against KIAA0513 protein with data of subjects in the Sawara Hospital cohort. |
Correlation analysis
Spearman's correlation analysis was performed a to determine the correlation between the s-KIAA0513-Ab levels and the continuous variables of participant parameters, including general information such as age, height, weight and BMI; the degree of artery stenosis, such as the maximum intima-media thickness (max-IMT); lifestyle factors such as smoking duration (years) and alcohol intake frequency (times/week); and blood test data. The average values of these parameters are listed in Table SVII. There was a significant correlation between the s-KIAA013-Ab levels and age, max-IMT, alkaline phosphatase, potassium, C-reactive protein (CRP), blood sugar and smoking duration (Table II), and an inverse correlation with height and weight. The correlation with max-IMT suggests that the s-KIAA0513-Ab levels are associated with stenosis and atherosclerosis, which was further confirmed by using other cohorts. Spearman's correlation analysis of the CKD cohort (300 participants) revealed a significant correlation with plaque score, max-IMT (55,56) and cardio-ankle vascular index (CAVI) (right) (57) (Table SVIII), which are indices of atherosclerosis. CRP was also associated with s-KIAA0513-Abs in the CKD cohort, suggesting the involvement of inflammation. By contrast, age, height, weight, BMI and potassium levels exhibited no significant correlation with s-KIAA0513-Abs in the CKD cohort. AIS is closely related to age, which may be indirectly associated with s-KIAA0513-Abs.
Table IICorrelation analysis of serum antibody levels against KIAA0513 with data on subjects in the Sawara Hospital cohort. |
Japan Public Health Center (JPHC) cohort analysis
A case-control study nested within the Japan Public Health Center-based Prospective Study was then conducted, which involved ~30,000 plasma samples (50-52). The level of antibodies against the KIAA0513 protein was positively associated with the risk of AIS. The ORs (95% CIs) were 2.11 (1.17-3.81) and 2.23 (1.18-4.21) for those in the third and highest quartiles of antibody levels, respectively, compared with those in the lowest quartile (Table III). These results indicate that the antibody markers against the KIAA0513 protein are useful for predicting the onset of AIS.
Discussion
In the present study, the initial ProtoArray screening identified KIAA0513 as an antigen, as recognized by serum IgG in patients with atherosclerosis, and subsequently recombinant GST-tagged KIAA0513 protein of 301 amino acids was purified. Western blot analysis confirmed the presence of autoantibodies against KIAA0513 (Fig. 1). Using the KIAA0513 protein as an antigen, the serum antibody levels were examined using AlphaLISA. The results revealed significantly higher s-KIAA0513-Ab levels in the patients with AIS, TIA, DM, CVD, OSAS, CKD, EC, GC, CC, LC and MC than in the HDs (Fig. 2A-E and Table SII, Table SIII, Table SIV, Table SV and Table VI). Among these diseases, the highest AUC values were observed for EC, type 2 CKD and DM (Fig. 3A-M). The close association between s-KIAA0513-Ab levels and hypertension (Table I) could account for the association with OSAS, which is frequently accompanied by hypertension (58). Spearman's correlation analysis revealed a significant correlation between s-KIAA0513-Ab and max-IMT, plaque score and CAVI, all of which are indices of atherosclerosis-related lesions (Tables II and SVIII) (55-57). By contrast, the s-KIAA0513-Ab levels were weakly correlated with blood sugar (P=0.023), but were completely unrelated to HbA1c, a typical DM marker (Table II). Thus, although the patients with DM exhibited high s-KIAA0513-Ab levels compared with the HDs, this antibody marker may not primarily reflect DM lesions, but rather atherosclerotic lesions caused by DM. Given that angiogenesis is essential for the development of cancer, vascular malformation may be accompanied by the typical alterations in atherosclerosis. In fact, DM and arteriosclerotic diseases are cancer risk factors (59-61).
There are three known splicing variants of KIAA0513: Isoform a (411 amino acids, NP_055547.1), isoform b (301 amino acids, NP_001273495.1) and isoform c (301 amino acids, NP_001284695.1). The full-length 301 amino acids of KIAA0513 isoforms c and b are exactly the same as the first 301 amino acids of KIAA0513 isoform a. The present study also purified GST-fused KIAA0513 isoform a and examined the serum antibodies using sera from HDs and patients with AIS and CVD. Both isoforms a and c of KIAA0513 exhibited higher antibody levels in the sera from patients with AIS or CVD than in the sera from HDs (Fig. S1A and B). The reactivity of KIAA0513 isoform c against serum antibodies was closely associated with that of KIAA0513 isoform a, although the former was higher than the latter (Fig. S1C), implying that the major epitope sites of serum autoantibodies are located in the 301 amino acids of isoform c.
KIAA0513 mRNA expression has been observed predominantly in the neurons and glial cells of the brain, with low-level expression in most human tissues, whereas the KIAA0513 protein was exclusively found in the brain (54). Among brain regions, the highest expression was in the cerebellum, cortex, hippocampus, pons, putamen and amygdala. Using a yeast 2-hybrid analysis of a fetal brain cDNA library, Lauriat et al (54) found that the N-terminal portion of KIAA0513 interacted with KIBRA, HAX1 and INTS4. A coimmunoprecipitation analysis revealed a physical association between KIAA0513 and KIBRA. Given that KIBRA, HAX1 and INTS4 are involved in synaptic and apoptotic signaling, KIAA0513 can also participate in these signaling pathways.
In addition to the KIAA0513-Abs employed in the present study, autoantibodies against ATPase, Ca++ transporting, plasma membrane 4, bone morphogenetic protein 1, deoxyhypusine synthase, low-density lipoprotein receptor-related protein-associated protein 1 and additional sex combs-like 2, which are markers of atherosclerosis, were also elevated in the sera of patients with EC (13,14,17,18), indicating that arterial abnormalities can also affect the carcinogenic process. In fact, angiogenesis is essential for the development of solid tumors (62), and diabetes and obesity, which induce arteriosclerosis, are risk factors for CRC and EC (63-65). Given that all tissues and organs require oxygen and nutrition provided by arteries, the alteration of arterial structure and/or function can affect numerous tissues and organs. All tissues and organs present in a body can affect each other to a certain degree (66). In other words, the AIS, CVD and CKD caused by atherosclerosis, the atherosclerosis induced by DM, and the solid cancer caused by arterial lesions can be interrelated with each other via arterial abnormalities. Markers associated with such abnormalities could therefore detect all of the above disorders.
Cancer, heart disease, cerebrovascular disease and renal failure are the first, second, fourth and eighth leading causes of mortality in Japan, respectively (Ministry of Health, Labor and Welfare 2018 vital statistics; https://www.mhlw.go.jp/toukei/saikin/hw/jinkou/kakutei18/dl/10_h6.pdf). The majority of the other causes of death are unavoidable, such as senility and accidents. In other words, the onset and progression of cancer, heart disease, cerebrovascular disease and renal failure (as well as their risk factor DM) can be suppressed by proper health management, such as early diagnosis and intervention. Notably, cancer, heart disease, cerebrovascular disease, renal failure and DM can be detected by the s-KIAA0513-Ab marker, making it applicable for diagnostic purposes and providing appropriate treatment, lifestyle guidance, etc., leading to improved quality of life.
As of 2020, numerous reports have shown that the presence of underlying diseases, such as DM, heart disease, cerebrovascular disease, cancer, OSAS and kidney disease aggravate the coronavirus disease 2019 (COVID-19) (67-70). The s-KIAA0513-Ab marker is therefore a highly useful tool for detecting patients with COVID-19 who are at a higher risk of mortality. Antibody markers are generally more sensitive than antigen markers. Given that the KIAA0513 protein has particularly high antigenicity, this KIAA0513-Ab marker is extremely sensitive. Given the major life-threatening diseases can be detected by this marker, the KIAA0513-Ab marker could be referred to as a ‘supermarker’.
The present study has certain limitations, which should be mentioned. First, although the increase in s-KIAA0513-Ab levels could be attributable to the high KIAA0513 expression levels (Fig. 4C) as suggested above, the association between the expression of the antigen and the antibody has not been completely verified. The antigen levels can be examined by immunohistochemistry, western blot analysis and mass spectrometry. However, accurately quantifying protein amounts across many specimens using these methods is still challenging. The introduction of the AlphaLISA method for the quantification of antigenic proteins may be another approach. Second, the significant differences of the prognosis between the s-KIAA-Ab-positive and -negative groups were observed in EC and CRC (Fig. 4A and B) but not GC, LC, or BC. Further accumulation of the latter specimens may clarify the association between s-KIAA0513-Ab levels and their prognosis.
In conclusion, the serum anti-KIAA0513 antibody marker appears to be useful for diagnosing the progress of atherosclerosis, which can lead to the onset of life-threatening AIS, CVD and cancer.
Supplementary Material
Comparison of reactivity between isoform c (amino acids 2-302) and isoform a (amino acids 1-411) of KIAA0513 as antigens for evaluation of serum antibody levels. The s-KIAA0513-Ab levels of HDs and patients with AIS or CVD were examined by AlphaLISA using GST-KIAA05132-302 (A) and GST-KIAA05131-411 (B) proteins as the antigens. A scatter dot plot of the antibody levels is shown. Results are presented as described in the legend of Fig. 2. ***P<0.001 vs. HD specimens. The bars represent the average ± SD. (C) Correlation plot of antibody levels against KIAA0513 isoform a vs. isoform c.
ProtoArray® screening for autoantibodies in atherosclerosis.
Comparison of the serum antibody levels of HDs vs. those of patients with AIS or TIA.
Comparison of the serum antibody levels of HDs vs. those of patients with DM.
Comparison of the serum antibody levels of HDs vs. those of patients with CVD or OSAS.
Comparison of s-KIAA0513-Ab levels of HDs vs. those of patients with CKD.
Comparison of s-KIAA0513-Ab levels between HDs and patients with cancer.
Information of subjects in the Sawara Hospital cohort used for correlation analysis.
Correlation analysis between serum KIAA0513-Ab levels and the data of CKD cohort.
Acknowledgements
The authors would like to thank Professor Masaki Takiguchi (Chiba University), Professor Hao Wang (Jinan University), Professor Kenichiro Kitamura (Yamanashi University), Dr Xiao-Meng Zhang (Chiba University), Dr Kazushige Katsura (RIKEN), Dr Hideyuki Kuroda (Fujikura Kasei Co.), Dr Rika Nakamura (Fujikura Kasei Co.), Dr Natsuko Shinmen (Fujikura Kasei, Co.) and Dr Noboru Ohsawa (RIKEN) for supporting this research, as well as Ms. Seiko Otsuka, Masae Suzuki, Chiho Kusaka, Satoko Ishibashi, Chiemi Mishima-Tsumagari, Risa Kimura, Akiko Kimura, Ryo Fukushima, Yuko Ohta, and Aki Furuya for providing technical assistance.
Funding
Funding: The present study was supported, in part, by research grants from the Japan Science and Technology Agency (JST: Exploratory Research No. 14657335), and JSPS KAKENHI Grant no. 20K17953, 22K07273, 20K07810, 21K19437 and 21K08695. The Japan Public Health Center-based Prospective Study was supported by National Cancer Center Research and Development Fund (since 2011) and a Grant-in-Aid for Cancer Research from the Ministry of Health, Labour and Welfare of Japan (from 1989 to 2010).
Availability of data and materials
All data of the ProtoArray v4.0 human protein microarray system are available in the public Figshare database (https://figshare.com/articles/dataset/Results_of_protein_array_for_atherosclerosis/25906330). The other datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
TH, MT, KYo, KM, KT, HS and YH conceived and designed the study. YY, MI, SYo, MT, YK and HT collected the blood samples and the clinicopathological data. MK, SYL, BSZ and GT performed the experiments and acquired the data. SM, TMac, MSh, SYa and GT contributed the reagents, materials, analysis tools or patient data. BSZ, TMat, TMac, MSa and HI analyzed and interpreted the data. MK, SYL, MI, MSa, KYa, NS, ST and AH performed the statistical analyses. TH, YY, KYa, NS and MSh drafted the manuscript. TH, TMatsutani, HI and ST confirm the authenticity of all the raw data. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
The present study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Local Ethical Review Board of the Chiba University, Graduate School of Medicine (Chiba, Japan), as well as by the review boards of the participating hospitals (approval no. 2018-320). The Ethics Committee of Toho University, Graduate School of Medicine (No. A18103_A17052_A16035_A16001_26095_25024_24038_22047_22047) and Port Square Kashiwado Clinic, Kashiwado Memorial Foundation (approval no. 2012-001) also approved the study protocol. Sera were collected from patients who had provided written informed consent. Serum samples were collected from patients who had provided written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The present study was performed in collaboration with Fujikura Kasei Co., Ltd. GT is an employee of Fujikura Kasei Co., Ltd. The other authors have no competing interests.
References
|
Goo YA and Goodlett DR: Advances in proteomic prostate cancer biomarker discovery. J Proteomics. 73:1839–1850. 2010.PubMed/NCBI View Article : Google Scholar | |
|
Imafuku Y, Omenn GS and Hanash S: Proteomics approaches to identify tumor antigen directed autoantibodies as cancer biomarkers. Dis Markers. 20:149–153. 2004.PubMed/NCBI View Article : Google Scholar | |
|
Ho PTB, Clark IM and Le LTT: MicroRNA-based diagnosis and therapy. Int J Mol Sci. 23(7167)2022.PubMed/NCBI View Article : Google Scholar | |
|
Kramer J, Harcos P, Prohászka Z, Horváth L, Karádi I, Singh M, Császár A, Romics L and Füst G: Frequencies of certain complement protein alleles and serum levels of anti-heat-shock protein antibodies in cerebrovascular diseases. Stroke. 31:2648–2652. 2000.PubMed/NCBI View Article : Google Scholar | |
|
Machida T, Kubota M, Kobayashi E, Iwadate Y, Saeki N, Yamaura A, Nomura F, Takiguchi M and Hiwasa T: Identification of stroke-associated-antigens via screening of recombinant proteins from the human expression cDNA library (SEREX). J Translat Med. 13(71)2015.PubMed/NCBI View Article : Google Scholar | |
|
Yoshida Y, Wang H, Hiwasa T, Machida T, Kobayashi E, Mine S, Tomiyoshi G, Nakamura R, Shinmen N, Kuroda H, et al: Elevation of autoantibody level against PDCD11 in patients with transient ischemic attack. Oncotarget. 9:8836–8848. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Wang H, Zhang XM, Tomiyoshi G, Nakamura R, Shinmen N, Kuroda H, Kimura R, Mine S, Kamitsukasa I, Wada T, et al: Association of serum levels of antibodies against MMP1, CBX1, and CBX5 with transient ischemic attack and cerebral infarction. Oncotarget. 9:5600–5613. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Yoshida Y, Zhang XM, Wang H, Machida T, Mine S, Kobayashi E, Adachi A, Matsutani T, Kamitsukasa I, Wada T, et al: Elevated levels of autoantibodies against DNAJC2 in sera of patients with atherosclerotic diseases. Heliyon. 6(e04661)2020.PubMed/NCBI View Article : Google Scholar | |
|
Li SY, Yoshida Y, Kobayashi E, Kubota M, Matsutani T, Mine S, Machida T, Maezawa Y, Takemoto M, Yokote K, et al: Serum anti-AP3D1 antibodies are risk factors for acute ischemic stroke related with atherosclerosis. Sci Rep. 11(13450)2021.PubMed/NCBI View Article : Google Scholar | |
|
Kubota M, Yoshida Y, Kobayashi E, Matsutani T, Li SY, Zhang BS, Mine S, Machida T, Takizawa H, Hiwasa T and Iwadate Y: Serum anti-SERPINE1 antibody as a potential biomarker of acute cerebral infarction. Sci Rep. 11(21772)2021.PubMed/NCBI View Article : Google Scholar | |
|
Hiwasa T, Wang H, Goto KI, Mine S, Machida T, Kobayashi E, Yoshida Y, Adachi A, Matsutani T, Sata M, et al: Serum anti-DIDO1, anti-CPSF2, and anti-FOXJ2 antibodies as predictive risk markers for acute ischemic stroke. BMC Med. 19(131)2021.PubMed/NCBI View Article : Google Scholar | |
|
Kubota M, Zhang BS, Li SY, Yoshida Y, Wang H, Adachi A, Matsutani T, Mine S, Machida T, Kamitsukasa I, et al: Serum anti-TSTD2 antibody as a biomarker for atherosclerosis-induced ischemic stroke and chronic kidney disease. Med Int (Lond). 3(4)2022.PubMed/NCBI View Article : Google Scholar | |
|
Hiwasa T, Machida T, Zhang XM, Kimura R, Wang H, Iwase K, Ashino H, Taira A, Arita E, Mine S, et al: Elevated levels of autoantibodies against ATP2B4 and BMP-1 in sera of patients with atherosclerosis-related diseases. Immunome Res. 11(097)2015. | |
|
Nakamura R, Tomiyoshi G, Shinmen N, Kuroda H, Kudo T, Doi H, Mine S, Machida T, Kamitsukasa I, Wada T, et al: An anti-deoxyhypusine synthase antibody as a marker of atherosclerosis-related cerebral infarction, myocardial infarction, diabetes mellitus, and chronic kidney disease. SM Atheroscler J. 1(1001)2017. | |
|
Hiwasa T, Tomiyoshi G, Nakamura R, Shinmen N, Kuroda H, Kunimatsu M, Mine S, Machida T, Sato E, Takemoto M, et al: Serum SH3BP5-specific antibody level is a biomarker of atherosclerosis. Immunome Res. 13(132)2017. | |
|
Zhang XM, Wang H, Mine S, Takemoto M, Yokote K, Kitamura K, Kobayashi Y, Machida T, Kobayashi E, Yoshida Y, et al: Association of serum anti-prolylcarboxypeptidase antibody marker with atherosclerotic diseases accompanied by hypertension. J Mol Biomark Diagn. 8(361)2017. | |
|
Sumazaki M, Shimada H, Ito M, Shiratori F, Kobayashi E, Yoshida Y, Adachi A, Matsutani T, Iwadate Y, Mine S, et al: Serum anti-LRPAP1 is a common biomarker for digestive organ cancers and atherosclerotic diseases. Cancer Sci. 111:4453–4464. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Li SY, Yoshida Y, Kobayashi E, Adachi A, Hirono S, Matsutani T, Mine S, Machida T, Ohno M, Nishi E, et al: Association between serum anti-ASXL2 antibody levels and acute ischemic stroke, acute myocardial infarction, diabetes mellitus, chronic kidney disease and digestive organ cancer, and their possible association with atherosclerosis and hypertension. Int J Mol Med. 46:1274–1288. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Chen PM, Ohno M, Hiwasa T, Nishi K, Saijo S, Sakamoto J, Morita Y, Matsuda S, Watanabe S, Kuwabara Y, et al: Nardilysin is a promising biomarker for the early diagnosis of acute coronary syndrome. Int J Cardiol. 243:1–8. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Palmer JP, Asplin CM, Clemons P, Lyen K, Tatpati O, Raghu PK and Paquette TL: Insulin antibodies in insulin-dependent diabetics before insulin treatment. Science. 222:1337–1339. 1983.PubMed/NCBI View Article : Google Scholar | |
|
Baekkeskov S, Aanstoot HJ, Christgau S, Reetz A, Solimena M, Cascalho M, Folli F, Richter-Olesen H and De Camilli P: Identification of the 64K autoantigen in insulin dependent diabetes as the GABA-synthesizing enzyme glutamic acid decarboxylase. Nature. 347:151–156. 1990.PubMed/NCBI View Article : Google Scholar | |
|
Hiwasa T, Zhang XM, Kimura R, Ohno M, Chen PM, Nishi E, Ono K, Kimura T, Kamitsukasa I, Wada T, et al: Elevated adiponectin antibody levels in sera of patients with atherosclerosis-related coronary artery disease, cerebral infarction, and diabetes mellitus. J Circ Biomark. 5(8)2016.PubMed/NCBI View Article : Google Scholar | |
|
Sugimoto K, Tomiyoshi G, Mori M, Kuwabara S, Hirano S, Sawai S, Beppu M, Muto M, Uzawa A, Kitamura K, et al: Identification of serum anti-GADD34 antibody as a common marker of diabetes mellitus and parkinson disease. J Alzheimers Dis Parkinsonism. 7(358)2017. | |
|
Tomiyoshi G, Nakamura R, Shinmen N, Yoshida Y, Mine S, Machida T, Iwase K, Iwadate Y, Hiwasa T and Kuroda H: GADD34 activates p53 and may have utility as a marker of atherosclerosis. Front Med (Lausanne). 10(1128921)2023.PubMed/NCBI View Article : Google Scholar | |
|
Yamagata H, Hayashi A, Yoshida Y, Koshizaka M, Onishi S, Yoshida T, Hiwasa T and Takemoto M: Association of high proprotein convertase subtilisin/kexin type 9 antibody level with poor prognosis in patients with diabetes: A prospective study. Sci Rep. 13(5391)2023.PubMed/NCBI View Article : Google Scholar | |
|
Shimada H, Takeda A, Arima M, Okazumi S, Matsubara H, Nabeya Y, Funami Y, Hayashi H, Gunji Y, Suzuki T, et al: Serum p53 antibody is a useful tumor marker in superficial esophageal squamous cell carcinoma. Cancer. 89:1677–1683. 2000.PubMed/NCBI | |
|
Shimada H, Ochiai T and Nomura F: Japan p53 Antibody Research Group. Titration of serum p53 antibodies in 1,085 patients with various types of malignant tumors: A multiinstitutional analysis by the Japan p53 antibody research group. Cancer. 97:682–689. 2003.PubMed/NCBI View Article : Google Scholar | |
|
Nakashima K, Shimada H, Ochiai T, Kuboshima M, Kuroiwa N, Okazumi S, Matsubara H, Nomura F, Takiguchi M and Hiwasa T: Serological identification of TROP2 by recombinant cDNA expression cloning using sera of patients with esophageal squamous cell carcinoma. Int J Cancer. 112:1029–1035. 2004.PubMed/NCBI View Article : Google Scholar | |
|
Kuboshima M, Shimada H, Liu TL, Nakashima K, Nomura F, Takiguchi M, Hiwasa T and Ochiai T: Identification of a novel SEREX antigen, SLC2A1/GLUT1, in esophageal squamous cell carcinoma. Int J Oncol. 28:463–468. 2006.PubMed/NCBI | |
|
Kuboshima M, Shimada H, Liu TL, Nomura F, Takiguchi M, Hiwasa T and Ochiai T: Presence of serum tripartite motif-containing 21 antibodies in patients with esophageal squamous cell carcinoma. Cancer Sci. 97:380–386. 2006.PubMed/NCBI View Article : Google Scholar | |
|
Shimada H, Kuboshima M, Shiratori T, Nabeya Y, Takeuchi A, Takagi H, Nomura F, Takiguchi M, Ochiai T and Hiwasa T: Serum anti-myomegalin antibodies in patients with esophageal squamous cell carcinoma. Int J Oncol. 30:97–103. 2007.PubMed/NCBI | |
|
Shimada H, Shiratori T, Yasuraok M, Kagaya A, Kuboshima M, Nomura F, Takiguchi M, Ochiai T, Matsubara H and Hiwasa T: Identification of makorin 1 as a novel SEREX antigen of esophageal squamous cell carcinoma. BMC Cancer. 9(232)2009.PubMed/NCBI View Article : Google Scholar | |
|
Kagaya A, Shimada H, Shiratori T, Kuboshima M, Nakashima-Fujita K, Yasuraoka M, Nishimori T, Kurei S, Hachiya T, Murakami A, et al: Identification of a novel SEREX antigen family, ECSA, in esophageal squamous cell carcinoma. Proteome Sci. 9(31)2011.PubMed/NCBI View Article : Google Scholar | |
|
Shimada H, Ito M, Kagaya A, Shiratori T, Kuboshima M, Suzuki M, Liu TL, Nabeya Y, Matsubara H, Matsushita K, et al: Elevated serum antibody levels against cyclin L2 in patients with esophageal squamous cell carcinoma. J Cancer Sci Ther. 7:60–66. 2015. | |
|
Ito M, Hiwasa T, Yajima S, Suzuki T, Oshima Y, Nanami T, Sumazaki M, Shiratori F, Li SY, Iwadate Y, et al: Low anti-CFL1 antibody with high anti-ACTB antibody is a poor prognostic factor in esophageal squamous cell carcinoma. Esophagus. 19:617–625. 2022.PubMed/NCBI View Article : Google Scholar | |
|
Ito M, Yajima S, Suzuki T, Oshima Y, Nanami T, Sumazaki M, Shiratori F, Wang H, Hu L, Takizawa H, et al: The combination of positive anti-WDR1 antibodies with negative anti-CFL1 antibodies is a poor prognostic factor for patients with esophageal carcinoma. Med Int (Lond). 3(11)2023.PubMed/NCBI View Article : Google Scholar | |
|
Kobayashi S, Hoshino T, Hiwasa T, Satoh M, Rahmutulla B, Tsuchida S, Komukai Y, Tanaka T, Matsubara H, Shimada H, et al: Anti-FIRs (PUF60) auto-antibodies are detected in the sera of early-stage colon cancer patients. Oncotarget. 7:82493–82503. 2016.PubMed/NCBI View Article : Google Scholar | |
|
Kobayashi S, Hiwasa T, Ishige T, Rahmutulla B, Kano M, Hoshino T, Minamoto T, Shimada H, Nomura F, Matsubara H and Matsushita K: Anti-FIRΔexon2, a splicing variant form of PUF60, auto-antibody is detected in the sera of esophageal squamous cell carcinoma. Cancer Sci. 110:2004–2013. 2019.PubMed/NCBI View Article : Google Scholar | |
|
Matsutani T, Hiwasa T, Takiguchi M, Oide T, Kunimatsu M, Saeki N and Iwadate Y: Autologous antibody to src-homology 3-domain GRB2-like 1 specifically increases in the sera of patients with low-grade gliomas. J Exp Clin Cancer Res. 31(85)2012.PubMed/NCBI View Article : Google Scholar | |
|
Adachi-Hayama M, Adachi A, Shinozaki N, Matsutani T, Hiwasa T, Takiguchi M, Saeki N and Iwadate Y: Circulating anti-filamin C antibody as a potential serum biomarker for low-grade gliomas. BMC Cancer. 14(452)2014.PubMed/NCBI View Article : Google Scholar | |
|
Hontani K, Tsuchikawa T, Hiwasa T, Nakamura T, Ueno T, Kushibiki T, Takahashi M, Inoko K, Takano H, Takeuchi S, et al: Identification of novel serum autoantibodies against EID3 in non-functional pancreatic neuroendocrine tumors. Oncotarget. 8:106206–106221. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Takahashi M, Tsuchikawa T, Hiwasa T, Nakamura T, Hontani K, Kushibiki T, Inoko K, Takano H, Hatanaka Y, Matsushita K, et al: Identification of antibody against wingless-type MMTV integration site family member 7B as a biliary cancer tumor marker. Oncol Rep. 49(34)2023.PubMed/NCBI View Article : Google Scholar | |
|
Matsumura T, Terada J, Kinoshita T, Sakurai Y, Yahaba M, Ema R, Amata A, Sakao S, Nagashima K, Tatsumi K and Hiwasa T: Circulating anti-coatomer protein complex subunit epsilon (COPE) autoantibodies as a potential biomarker for cardio- and cerebro-vascular events in patients with obstructive sleep apnea. J Clin Sleep Med. 13:393–400. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Matsumura T, Terada J, Kinoshita T, Sakurai Y, Yahaba M, Tsushima K, Sakao S, Nagashima K, Iwata Y, Ozaki T, et al: Autoantibody against NBL1 in obstructive sleep apnea patients with cardiovascular disease. PLoS One. 13(e0195015)2018.PubMed/NCBI View Article : Google Scholar | |
|
Katsumata Y, Terada J, Matsumura T, Koshikawa K, Sakao S, Tomiyoshi G, Shinmen N, Nakamura R, Kuroda H, Nagashima K, et al: Circulating anti-sorting nexins 16 antibodies as an emerging biomarker of coronary artery disease in patients with obstructive sleep apnea. Diagnostics (Basel). 10(71)2020.PubMed/NCBI View Article : Google Scholar | |
|
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL and Marsh EE III: Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in acute stroke treatment. Stroke. 24:35–41. 1993.PubMed/NCBI View Article : Google Scholar | |
|
Nishiura R, Fujimoto S, Sato Y, Yamada K, Hisanaga S, Hara S, Nakao H and Kitamura K: Elevated osteoprotegerin levels predict cardiovascular events in new hemodialysis patients. Am J Nephrol. 29:257–263. 2009.PubMed/NCBI View Article : Google Scholar | |
|
Komatsu H, Fujimoto S, Hara S, Fukuda A, Fukudome K, Yamada K, Sato Y and Kitamura K: Recent therapeutic strategies improve renal outcome in patients with IgA nephropathy. Am J Nephrol. 30:19–25. 2009.PubMed/NCBI View Article : Google Scholar | |
|
Chumpolkulwong N, Sakamoto K, Hayashi A, Iraha F, Shinya N, Matsuda N, Kiga D, Urushibata A, Shirouzu M, Oki K, et al: Translation of ‘rare’ codons in a cell-free protein synthesis system from escherichia coli. J Struct Funct Genomics. 7:31–36. 2006.PubMed/NCBI View Article : Google Scholar | |
|
Tsugane S and Sawada N: The JPHC study: Design and some findings on the typical Japanese diet. Jpn J Clin Oncol. 44:777–782. 2014.PubMed/NCBI View Article : Google Scholar | |
|
Ikeda A, Iso H, Sasazuki S, Inoue M and Tsugane S: JPHC Study Group. The combination of Helicobacter pylori- and cytotoxin-associated gene-A seropositivity in relation to the risk of myocardial infarction in middle-aged Japanese: The Japan public health center-based study. Atherosclerosis. 230:67–72. 2013.PubMed/NCBI View Article : Google Scholar | |
|
Iso H, Noda H, Ikeda A, Yamagishi K, Inoue M, Iwasaki M and Tsugane S: The impact of C-reactive protein on risk of stroke, stroke subtypes, and ischemic heart disease in middle-aged Japanese: The Japan public health center-based study. J Atheroscler Thromb. 19:756–766. 2012.PubMed/NCBI | |
|
Camp RL, Dolled-Filhart M and Rimm DL: X-tile: A new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res. 10:7252–7259. 2004.PubMed/NCBI View Article : Google Scholar | |
|
Lauriat TL, Dracheva S, Kremerskothen J, Duning K, Haroutunian V, Buxbaum JD, Hyde TM, Kleinman JE and McInnes LA: Characterization of KIAA0513, a novel signaling molecule that interacts with modulators of neuroplasticity, apoptosis, and the cytoskeleton. Brain Res. 1121:1–11. 2006.PubMed/NCBI View Article : Google Scholar | |
|
Tran LT, Park HJ and Kim HD: Is the carotid intima-media thickness really a good surrogate marker of atherosclerosis? J Atheroscler Thromb. 19:680–690. 2012.PubMed/NCBI View Article : Google Scholar | |
|
Zureik M, Ducimetière P, Touboul PJ, Courbon D, Bonithon-Kopp C, Berr C and Magne C: Common carotid intima-media thickness predicts occurrence of carotid atherosclerotic plaques: Longitudinal results from the aging vascular study (EVA) study. Arterioscler Thromb Vasc Biol. 20:1622–1629. 2000.PubMed/NCBI View Article : Google Scholar | |
|
Shirai K, Utino J, Otsuka K and Takata M: A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb. 13:101–107. 2006.PubMed/NCBI View Article : Google Scholar | |
|
Floras JS: Hypertension and sleep apnea. Can J Cardiol. 31:889–897. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Gallagher EJ and LeRoith D: Obesity and diabetes: The increased risk of cancer and cancer-related mortality. Physiol Rev. 95:727–748. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Goto A, Yamaji T, Sawada N, Momozawa Y, Kamatani Y, Kubo M, Shimazu T, Inoue M, Noda M, Tsugane S and Iwasaki M: Diabetes and cancer risk: A mendelian randomization study. Int J Cancer. 146:712–719. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Tapia-Vieyra JV, Delgado-Coello B and Mas-Oliva J: Atherosclerosis and cancer; A resemblance with far-reaching implications. Arch Med Res. 48:12–26. 2017.PubMed/NCBI View Article : Google Scholar | |
|
Makrilia N, Lappa T, Xyla V, Nikolaidis I and Syrigos K: The role of angiogenesis in solid tumours: An overview. Eur J Intern Med. 20:663–671. 2009.PubMed/NCBI View Article : Google Scholar | |
|
Jarvandi S, Davidson NO and Schootman M: Increased risk of colorectal cancer in type 2 diabetes is independent of diet quality. PLoS One. 8(e74616)2013.PubMed/NCBI View Article : Google Scholar | |
|
Fujihara S, Kato K, Morishita A, Iwama H, Nishioka T, Chiyo T, Nishiyama N, Miyoshi H, Kobayashi M, Kobara H, et al: Antidiabetic drug metformin inhibits esophageal adenocarcinoma cell proliferation in vitro and in vivo. Int J Oncol. 46:2172–2180. 2015.PubMed/NCBI View Article : Google Scholar | |
|
Berger NA: Young adult cancer: Influence of the obesity pandemic. Obesity (Silver Spring). 26:641–650. 2018.PubMed/NCBI View Article : Google Scholar | |
|
Hiwasa T and Shimada H: Autoantibody in cancer. In: Biomarkers in Cancer Therapy. Shimada H (ed). Springer Nature, Singapore, 25-40, 2018. | |
|
Zhou Y, Yang Q, Chi J, Dong B, Lv W, Shen L and Wang Y: Comorbidities and the risk of severe or fatal outcomes associated with coronavirus disease 2019: A systematic review and meta-analysis. Int J Infect Dis. 99:47–56. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Wu T, Zuo Z, Kang S, Jiang L, Luo X, Xia Z, Liu J, Xiao X, Ye M and Deng M: Multi-organ dysfunction in patients with COVID-19: A systematic review and meta-analysis. Aging Dis. 11:874–894. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Ye Q, Lu D, Shang S, Fu J, Gong F, Shu Q and Mao J: Crosstalk between coronavirus disease 2019 and cardiovascular disease and its treatment. ESC Heart Fail. 7:3464–3472. 2020.PubMed/NCBI View Article : Google Scholar | |
|
Maas MB, Kim M, Malkani RG, Abbott SM and Zee PC: Obstructive sleep apnea and risk of COVID-19 infection, hospitalization and respiratory failure. Sleep Breath. 25:1155–1157. 2021.PubMed/NCBI View Article : Google Scholar |