Research progress on long non‑coding RNAs in non‑infectious spinal diseases (Review)
- Authors:
- Published online on: July 9, 2024 https://doi.org/10.3892/mmr.2024.13288
- Article Number: 164
-
Copyright: © Ma et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
The spine is one of the most common sites of lesions in middle-aged and older individuals (>45 years of age) (1). Conditions such as intervertebral disc degeneration (IDD), ankylosing spondylitis (AS) and spinal cord injury (SCI) are common non-infectious spinal diseases that impair the quality of life of patients, and cause a huge economic burden on families and society (2). In addition to causing financial stress, IDD and other spinal disorders are important causes of pain and disability in patients (3). Notably, the incidence of spinal diseases such as IDD has increased, with a trend towards younger individuals (13–20 years of age) (4). Reportedly, ~80% of individuals experience lower back pain due to IDD during their lifetime (5). Unfortunately, the current treatment for these spinal diseases encompasses pain reduction and symptom improvement (6), and does not reverse or cure the diseases (7). Therefore, a more comprehensive understanding of the pathology of these diseases is of great significance for developing new therapeutic drugs, improving and optimising treatment programmes, and curing the diseases.
Although the human genome is expansive, only ~1% of the genome is involved in coding proteins (5), with >98% not directly involved in protein translation (8). RNAs transcribed from these genes not directly involved in protein translation is called non-coding RNAs (ncRNAs). Although ncRNAs are not involved in protein synthesis, they play important roles in regulating gene expression, cell function and development (9). NcRNAs can be divided into numerous categories, including microRNAs (miRNAs/miRs), long non-coding RNAs (lncRNAs), circular RNAs (circRNAs), small interfering RNAs and PIWI-interacting RNA (10). lncRNAs are RNAs with a length of >200 nucleotides that cannot encode proteins and have various biological functions (11). The functions of lncRNAs include gene regulation, RNAs processing and splicing regulation, cell cycle and proliferation, cell signal transduction, chromosome structure and nucleoplasmic transport (12). The present article reviews the functional classification of lncRNAs and their roles in IDD, AS and SCI, and discusses the potential of lncRNAs for treating spinal diseases such as IDD.
Introduction to lncRNAs
Sources and classification of lncRNAs
The majority of lncRNAs are similar in origin to mRNAs and are transcribed by RNA polymerase II. They also have the same structure as mRNAs (13); however, lncRNAs also have certain characteristics, although these are not unique to lncRNAs. lncRNAs account for >60% of all ncRNAs [certain studies report rates of >70% (14) or 70–90% (15)], and most lncRNAs contain >2 exons (16). Some lncRNAs, owing to their reverse splicing or lack of a 5′ cap, are readily connected with their own 3′ poly-A tail, generating circRNA (17). Numerous lncRNA genes have miRNA sequences embedded in their exons or introns, making them a source of miRNAs (13). A number of chemical modification methods exist for lncRNAs, including adenosine methylation, cytosine modification, uridine isomerisation, guanosine methylation and ribose modification (18), resulting in more complex ncRNA biogenesis.
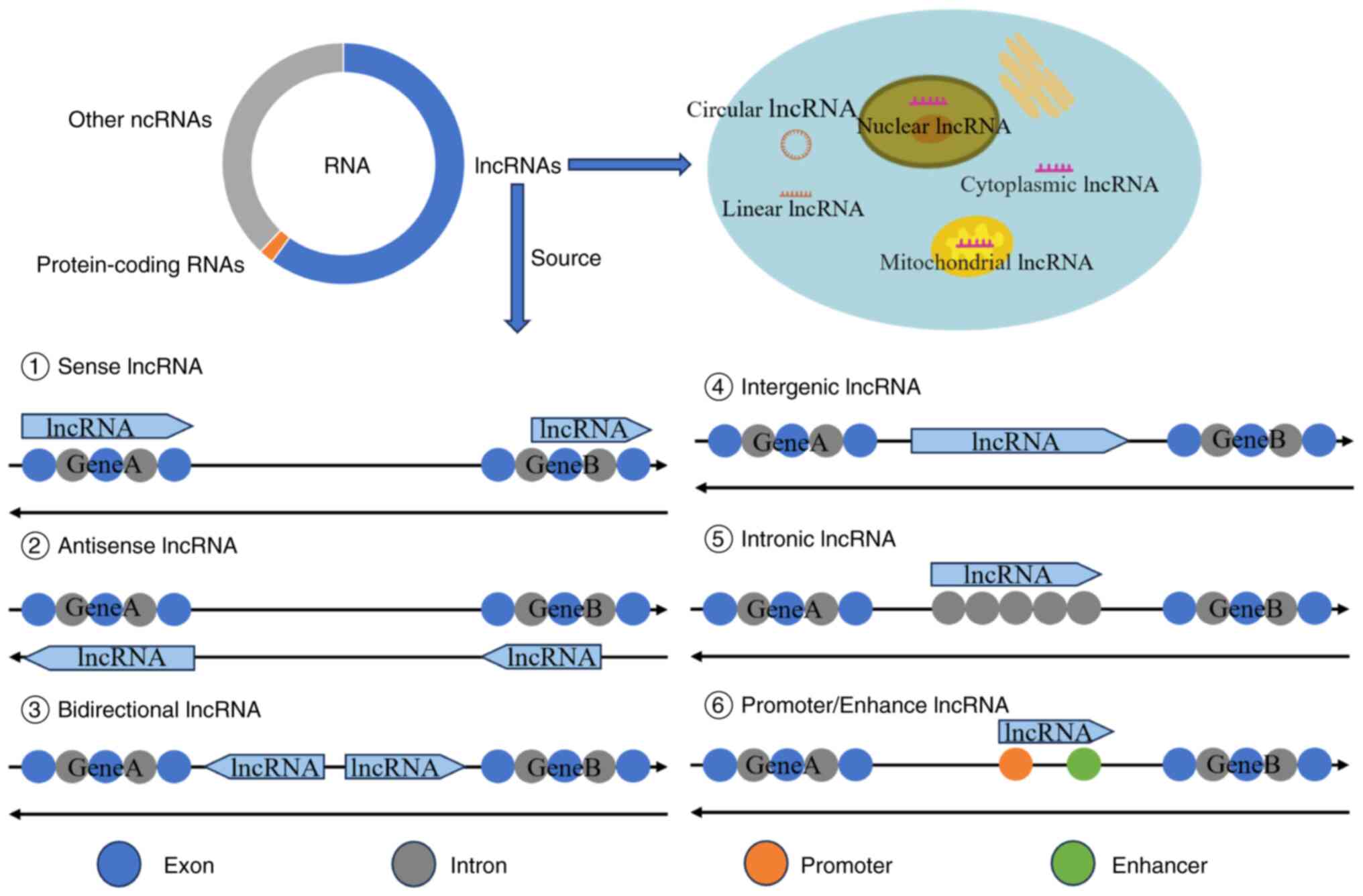
To date >900 lncRNAs have been identified (19), resulting in different methods of classifying lncRNAs (Fig. 1). Based on their location, they can be divided into nuclear, cytoplasmic and mitochondrial lncRNAs (20). According to their shape, lncRNAs can be divided into linear and circular lncRNAs (21). lncRNAs can also be categorised by source as follows: i) Sense transcript; ii) antisense transcript; iii) bidirectional transcript; iv) long intergenic ncRNAs (22); v) intron region; vi) promoter region (14); and vii) enhancer region (23). Finally, lncRNAs can be categorised by function as follows: i) Functional lncRNAs, ii) non-functional lncRNAs; and iii) lncRNAs for which transcription alone is sufficient to exert their effects and those that do not need to rely on transcripts to function (24). In general, the classification of lncRNAs is regarded as imperfect, as functional interactions and overlaps exist among different classifications of lncRNAs (5). In the future, a better lncRNA classification system will be required.
Functions of lncRNAs
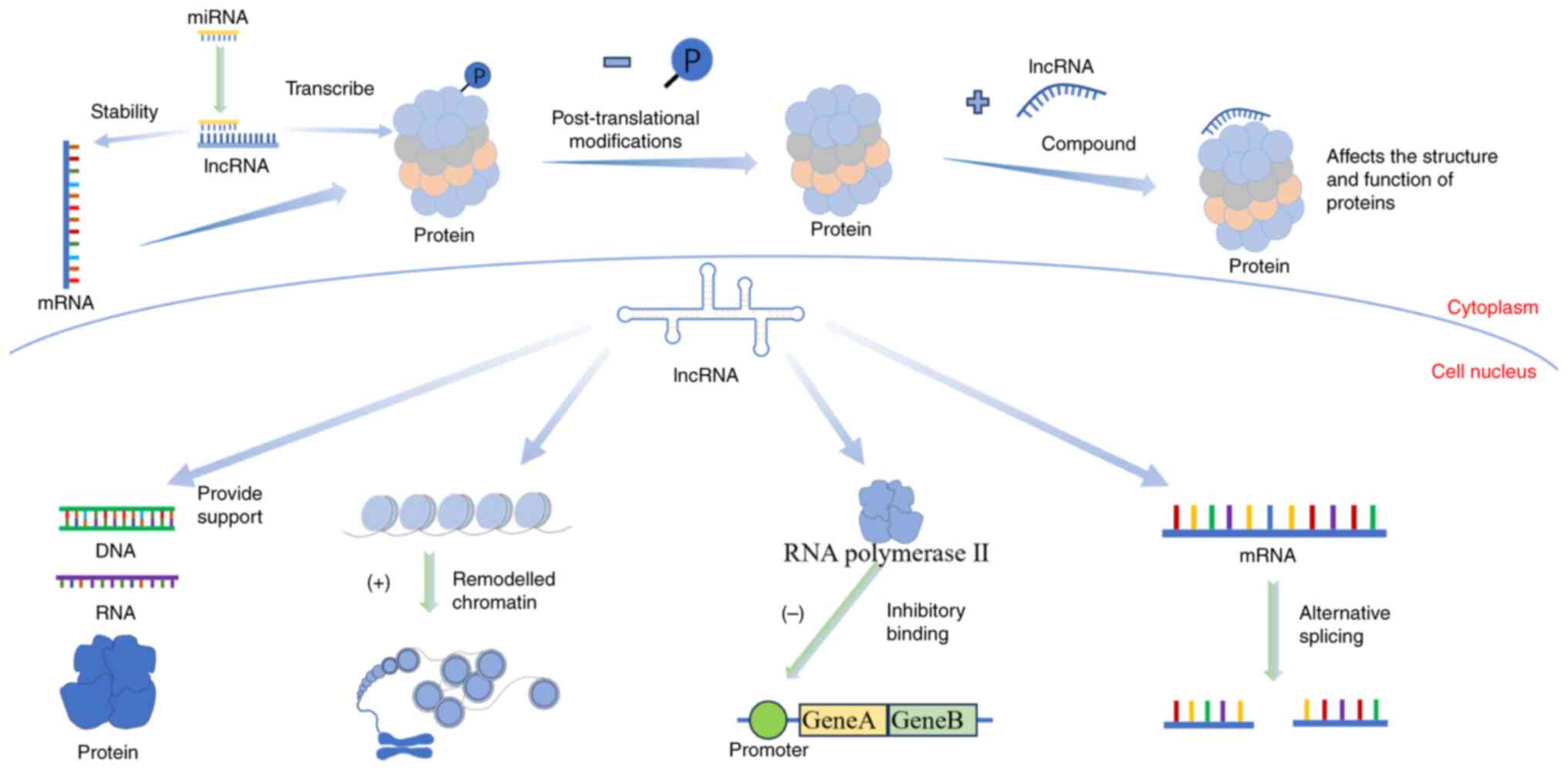
The function of lncRNAs varies depending on their spatial structure and location. lncRNAs have two functional forms including spliced and unspliced (25). The biological genetic information in mRNAs is mainly transmitted through ‘linear’ coding, while the biological information in lncRNAs mainly works through its different ‘spatial’ structural mechanisms (26). The main pathways through which lncRNAs function include epigenetic, transcriptional and post-transcriptional regulation (27). When lncRNAs perform their functions, they usually combine with DNA, RNAs and proteins to form the corresponding complexes. First, lncRNAs bind with DNA and play a role in decompressing chromatin (28). They can also affect chromatin accessibility (29), and the R-loop can make lncRNAs an ideal regulatory centre (30). Next, lncRNAs bind to RNAs. The lncRNAs regulate mRNA expression through competitive endogenous RNA mechanisms (31). Finally, lncRNAs bind with proteins to form complexes, regulating protein structure and function (32). Some lncRNAs directly bind to proteins, affecting cellular signal transduction and gene expression.
The functions of lncRNAs vary depending on their location (Fig. 2). The functions of lncRNAs in the nucleus include: i) Providing scaffolding for DNA (33), RNAs and proteins (34); ii) recruiting chromatin-remodelling complexes to remodel the chromatin (35); iii) preventing RNA polymerase II from attaching to the gene promoter and interfering with transcription (36); and iv) alternative splicing (37). The functions of lncRNAs in the cytoplasm include the following: i) Recruiting proteins that promote mRNA degradation and affect the stability of mRNAs (13); ii) sequestering specific miRNAs through the competing endogenous RNA (ceRNA) mechanism to regulate translation (38); iii) interfering with post-translational modifications; and iv) affecting the structure and function of proteins (39). Although the function of lncRNAs is understood to a certain extent, it cannot be inferred from its primary structure, unlike mRNAs. Therefore, further in-depth exploration of lncRNA functions is required.
Research progress of lncRNAs in IDD
Introduction to IDD
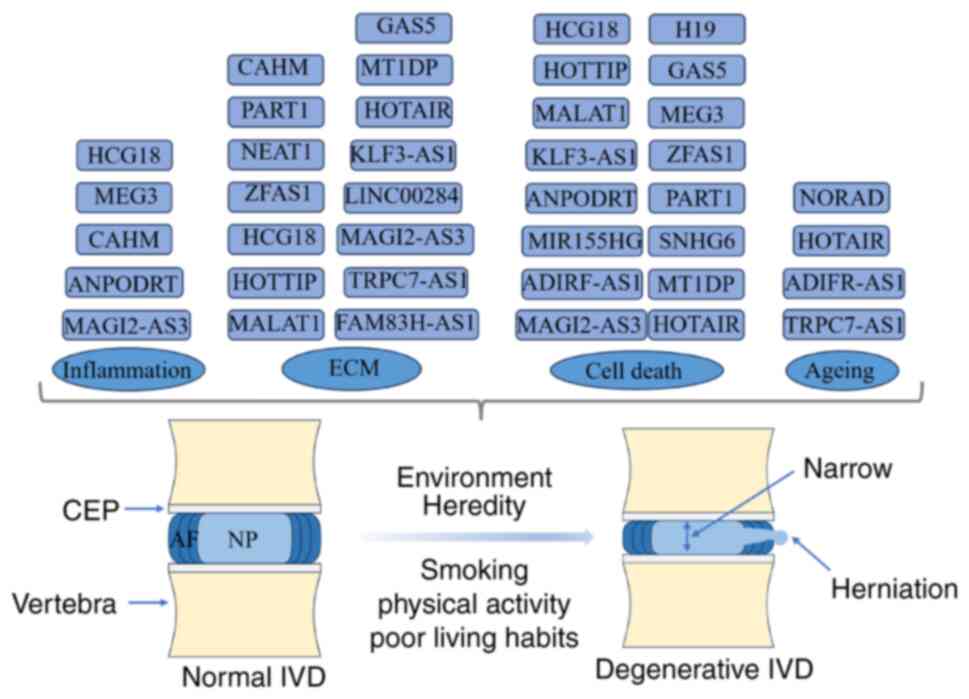
The intervertebral disc (IVD) is the largest non-blood supply tissue in the human body (40). The IVD is composed of three parts: i) The cartilage endplate (CEP) on the upper and lower sides; ii) the surrounding annulus fibrosus (AF); and iii) the nucleus pulposus (NP) in the middle (41). The CEP is ~1-mm thick and contains a number of micropores for material exchange in the NP, but contains no nerve tissue. The AF is divided into outer and inner rings, and the front and sides are twice as thick as the back. Nerve endings are only distributed in the outer ring (42). The AF is very strong and plays a major role in supporting and stabilising the spine (42). The NP is a gel-like tissue with a high water content, containing collagen, proteoglycans, NP cells and water (43). The composition and structure change with age, accounting for 50–60% of the IVD cross-section (44). The toughness and elasticity of the IVD enable it to perform important physiological functions, such as supporting the spine, buffering pressure and providing spine mobility (45).
As the IVD has no blood vessels, its self-repair ability is poor, making it prone to degeneration (40). IDD initiates in NP cells (46), and this is hypothesised to be primarily due to the joint action of genetic and environmental factors. Smoking, physical activity and poor living habits are contributing factors to IDD, although genetic factors are the most important (47). It is estimated that >70% of cases are caused by genetics and the programmed cell death drives the occurrence of IDD (48). After the onset of IDD, the water content in the IVD decreases, and the extracellular matrix (ECM), including type II collagen and proteoglycans, degrades (49). After the CEP and AF are destroyed, the barrier between the IVD, and the circulatory and immune systems is broken, triggering an inflammatory reaction (50). Under the influence of these pathogenic factors and changes in the microenvironment, the proliferative ability of the NP cells decreases, causing cell apoptosis, autophagy and senescence, ultimately resulting in IDD (5). Surgical treatment of IDD also has challenges, including infection (51), recurrence (52) and adjacent spondylosis (53). Therefore, understanding the pathogenesis of IDD and exploring new therapeutic targets are crucial.
Expression differences of lncRNAs in IDD
Compared with normal tissues, IDD exhibits numerous differentially expressed ncRNAs, including lncRNAs, which play an important role in the occurrence and development of IDD. Wan et al (54) used lncRNA-mRNA microarrays to study lncRNA expression in NP specimens from human degenerative NP tissue and normal groups. The results revealed that the expression levels of 67 lncRNAs increased and those of 49 decreased. In another study, the same lncRNA-mRNA microarray technology was used to analyse the lncRNA expression in NP specimens of human degenerative NP tissue and normal groups, and 135 significantly upregulated and 170 downregulated lncRNAs were found (55). A similar study found that 2,418 mRNAs and 528 lncRNAs were differentially expressed in the IDD group compared with the control group (56). Shi et al (57) recently constructed a ceRNA network containing 15 lncRNAs, 9 miRNAs and 103 mRNAs, and discovered a lncRNA/miRNA/mRNA axis, which affects the progression of IDD, namely, the small nucleolar RNA host gene 5/miR-299-5p/activating transcription factor 2 axis. However, further experimental verification is still needed. In another study, a ceRNA network was constructed using 15 lncRNAs and 21 miRNAs, and identified the two hub genes MAPK8 and CAPN1 in this regulatory network as key biomarkers of IDD (58). These studies demonstrate that there are obvious differences in lncRNA expression in IDD and that lncRNAs do not function independently but form an extensive ceRNA network system with other ncRNAs.
Association between NP cell phenotypic dysregulation and IDD
As aforementioned, IDD originates primarily from NP cells. As early as 2006, researchers found that transplantation of human NP cells into degenerated IVDs in rabbits could improve the degree of disc degeneration (59). A subsequent similar study showed that transplantation of NP stem cells into degenerated IVDs can more effectively improve IDD than transplantation of NP cells (60). These studies indicate that NP cells play a crucial role in the occurrence and development of IDD. The state of NP cells is influenced by the ECM, inflammation, apoptosis, necrosis and ageing factors. Under a series of negative influences, this ultimately leads to dysregulation of the NP cell phenotype, thereby participating in the occurrence and development of IDD (61). Jiang et al (62) reported that miR-365 can alleviate the development of IDD by regulating the synthesis and degradation of the ECM in NP cells. Propionibacterium acnes has been shown to induce the apoptosis of NP cells through the TLR2/JNK pathway, which can alter the process of IDD (63). Inflammation in NP cells also affects IDD, whereby extracellular lactate can promote activation of the NLR family pyrin domain containing 3 (NLRP3) inflammasome, increasing inflammatory levels in the NP cells and thereby promoting IDD (64). Experimental results from Du et al (65) suggested that activation of cannabinoid receptor type 2 can delay the ageing of NP cells, restore the balance of ECM metabolism and attenuate IDD. Overall, changes in the ECM, cell death, cell inflammation and cell ageing are all part of the complex mechanisms underlying the occurrence and development of IDD. They interact with each other, collectively influencing the process of IDD.
Role of lncRNAs in the ECM
The ECM, type II collagen and proteoglycan (aggrecan) are the main components of the NP. The ECM is produced by NP cells, and the balance between ECM synthesis and degradation contributes to the biomechanical balance and structural stability of the IVD, which is also considered a key indicator for evaluating NP cell function (66). Subsequent studies indicate that lncRNAs can affect the synthesis and degradation of the ECM through a ceRNA mechanism and can directly regulate hub genes or signalling pathways. Gao et al (67) studied degenerated human tissues and cells, and found that prostate androgen-regulated transcript 1 (PART1) was upregulated in degenerated NP tissues. By contrast, PART1 knockdown led to increased ECM synthesis, reduced degradation and enhanced proliferation of NP cells. Growth arrest specific 5 has been shown to be expressed at abnormally high levels in human degenerative NP tissues (68). After downregulation, angiopoietin 2 has been shown to be inhibited in a miR-17-3p-dependent manner, promoting ECM remodelling, inhibiting NP cell apoptosis and improving IDD (68). A recent study showed that the transcription factor FOXO3 may enhance the competitive binding of HOXA transcript at the distal tip (HOTTIP) and miR-615-3p by activating HOTTIP transcription, increasing the expression of the target gene collagen type II α1 of miR-615-3p, inducing NP cell proliferation, and reducing apoptosis to prevent ECM degradation (69). Krüppel-like factor 3 antisense RNA 1 (KLF3-AS1), like HOTTIP, was also shown to be expressed at low levels in degenerative NP tissues. Overexpression of KLF3-AS1 can increase NP cell viability, prevent cell apoptosis and increase ECM synthesis (70).
Shang et al (71) showed that docosahexaenoic acid, which plays a protective role in numerous chronic diseases, can alleviate excessive ECM degradation by reducing nuclear paraspeckle assembly transcript 1 overexpression in degenerative NP tissues. This previous study also revealed that ZNFX1 antisense RNA 1 (ZFAS1) was upregulated in degenerative NP tissues and NP cells treated with IL-1β, and that ZFAS1 competitively bound to miR-4711-5p through a ceRNA mechanism, thereby affecting the expression of adaptor-associated kinase 1, which is the target of miR-4711-5p. Silencing ZFAS1 may inhibit IDD progression by reducing NP apoptosis and ECM degradation (72). Similar to most lncRNAs, long intergenic nonprotein coding RNA 284 was revealed to be upregulated in IDD tissues and IL-1β-induced NP cells, and it activated the Wnt/β-catenin pathway by sponging miR-205-3p, thereby inhibiting NP cell proliferation and ECM synthesis (73). Zhan et al (74) showed that Hox transcript antisense intergenic RNA (HOTAIR) expression was positively associated with the IDD grade. Oe-HOTAIR enhanced NP cell autophagy, and promoted NP cell ECM degradation, apoptosis and senescence (74). A subsequent study found that HOTAIR regulates ECM synthesis and degradation in NP cells (75). In addition, inhibiting HOTAIR in in vivo animal models can effectively reduce IDD symptoms (75). In general, lncRNAs, miRNAs and circRNAs form an intricate regulatory network that jointly affects the synthesis and degradation of the ECM in IDD and IDD progression (Table I and Fig. 3).
Role of lncRNAs in cell death
Owing to different damage factors, NP cells die through a number of the same mechanisms as other cells, such as apoptosis, pyroptosis, autophagy, ferroptosis and necrosis (76). Currently, the most studied process is NP cell apoptosis. As aforementioned, PART1 not only affects the synthesis and degradation of the ECM through the miR-93/matrix metalloproteinase-2 signalling axis, but increased PART1 expression in degenerated tissues can also promote the apoptosis of NP cells and promote IDD (67). Similarly, HOTTIP, while preventing ECM degradation through a ceRNA mechanism, can also inhibit the apoptosis of NP cells and promote proliferation, improving the progression of IDD (69). In degenerated NP cells, small nucleolar RNA host gene 6 (SNHG6) expression has been reported to be upregulated and SNHG6 can induce NP cell apoptosis by targeting miR-101-3p (77). Chen et al (70) illustrated the possible impact of KLF3-AS1 on IDD. Oe-KLF3-AS1 enhanced NP cell viability and prevented apoptosis. The experimental results confirmed that KLF3-AS1 overexpression improved degenerative changes in NP cells through the miR-10a-3p/zinc finger and BTB domain-containing protein 20 (ZBTB20) axis. Yu and Li (78) detected low expression of membrane associated guanylate kinase, WW and PDZ domain containing 2 (MAGI2)-AS3 and IL-10, and high expression of miR-374b-5p in lipopolysaccharide (LPS)-induced NP cells. Notably, miR-374b-5p is a target of MAGI2-AS3 and IL-10. MAGI2-AS3 can increase the expression of IL-10 by competing with miR-374b-5p, thereby reducing the inflammatory response and cell apoptosis. Thus, the regulation of NP cell apoptosis by lncRNAs is mainly based on the ceRNA mechanism.
LncRNAs regulate the apoptosis of NP cells and affect pyroptosis and autophagy. For instance, MIR155 host gene (MIR155HG) has been shown to be upregulated in degenerated human NP tissues, and further experiments revealed that MIR155HG sponges miR-223-3p and promotes NLRP3 expression, thereby inducing pyroptosis in NP cells (79). Furthermore, platelet-rich plasma-derived extracellular vesicles may inhibit tert-butyl hydroperoxide-induced NP cell injury by upregulating metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) expression; MALAT1 regulates the miR-217/silent information regulator sirtuin 1 (SIRT1) signalling axis, and its overexpression can alleviate NP cell pyroptosis (80). As aforementioned, HOTAIR can affect the synthesis and decomposition of the ECM in NP cells and the apoptosis of NP cells. Simultaneously, HOTAIR can also affect autophagy and alter the progression of IDD (75). As the first lncRNA discovered (81), H19 imprinted maternally expressed transcript (H19) is highly conserved and widely expressed in mammals. Sun et al (82) found that H19 was highly expressed in degenerated NP tissues, and experimentally verified that H19 can promote autophagy and apoptosis in NP cells through the miR-139-3p/C-X-C motif chemokine receptor type 4/nuclear factor-κB (NF-κB) signalling axis, thereby affecting IDD (Table I and Fig. 3).
Role of lncRNAs in cellular inflammation
Inflammation can accelerate the process of IDD and cause damage and dehydration of the IVD tissue, resulting in loss of its original elasticity and function. Therefore, preventing and controlling inflammation is crucial for treating IDD (83). LncRNAs are important molecules that regulate inflammatory responses. Jiang et al (84) found that IDD tissues showed decreased expression of family with sequence similarity 83 member H (FAM83H)-AS1 compared with normal tissues, and Oe-FAM83H-AS1 could promote NP-cell proliferation, and reduce inflammatory responses and ECM degradation. In addition, miR-22-3p mediated the effect of FAM83H-AS1 on degenerated NP tissue (84). In IL-1β-stimulated NP cells, the expression of HLA complex group 18 (HCG18) and follistatin-like protein 1 (FSTL1) has been shown to be increased. Subsequent overexpression experiments demonstrated the impact of the HCG18/miR-495-3p/FSTL1 signalling axis on NP tissue inflammation and apoptosis (85). Zhang et al (86) showed that melatonin can reduce inflammation and apoptosis in NP cells while upregulating maternally expressed gene 3 (MEG3) expression, whereas MEG3 expression was shown to be downregulated in the degenerated NP. The study revealed that melatonin can reduce NP cell inflammation and apoptosis through the MEG3/miR-15a-5p/peroxisome proliferator-activated receptor γ coactivator 1α/SIRT1 pathway. Similar to most lncRNAs described previously, MAGI2-AS3 also affects IDD progression through a ceRNA mechanism. MAGI2-AS3 has been reported to be poorly expressed in degenerated NP cells, whereas oe-MAGI2-AS3 can downregulate miR-374b-5p and upregulate IL-10 expression to improve inflammation and ECM degradation in NP cells (78). Exosomes derived from bone marrow mesenchymal stem cells (BMSC-Exos) have therapeutic effects on IDD. In co-culture experiments, BMSC-Exos delivered colon adenocarcinoma hypermethylated (CAHM) to inhibit the polarisation of M1 macrophages, thereby reducing the apoptosis of degenerated NP cells, ECM degradation and the expression of inflammatory factors, and improving IDD (87). In addition to the aforementioned lncRNAs, numerous lncRNAs discovered in previous studies also have important effects on the occurrence and development of IDD (56), requiring further verification experiments (Table I and Fig. 3).
Role of lncRNAs in cellular senescence
Senescence of NP cells plays an important role in the development of IDD. During ageing, the regenerative capacity of NP cells weakens, apoptosis and inflammation increase, and cell metabolism becomes abnormal (65), ultimately promoting the occurrence of IDD under the combined action of various factors. In vivo and in vitro experiments have shown that the expression levels of adipogenesis regulatory factor (ADIRF)-AS1 and SERPINA1 are downregulated in high-grade degenerated NP tissues and that both have binding sites for miR-214-3p. Subsequent experiments revealed that ADIRF-AS1 can bind to miR-214-3p, thereby increasing SERPINA1 expression and ultimately delaying NP cell ageing and apoptosis (88). As aforementioned, HOTAIR can promote the apoptosis and ECM degradation of NP cells, and accelerate the ageing of NP cells, which can comprehensively affect the progress of IDD in these numerous aspects (74). Furthermore, transient receptor potential canonical 7 RNA 1-AS1 adsorbs miR-4769-5p through the classical ceRNA mechanism, inhibiting HPN and regulating ageing, vitality and ECM synthesis in NP cells (89). The methylation level of lncRNA activated by DNA damage (NORAD) has been shown to be significantly increased in ageing NP cells, and the expression of WTAP was revealed to be increased in the degenerated NP, significantly promoting the m6 modification of NORAD; the lack of NORAD can promote cellular senescence by affecting the expression of Pumilio-homology domain (90).
In general, research on the mechanisms of lncRNAs in IDD has made great progress since the discovery of the first lncRNAs, H19, and the mechanisms of numerous lncRNAs in IDD have been clarified step-by-step. The same lncRNAs can control IDD through different signalling channels. Different lncRNAs can also function through the same signalling pathway. The impact of lncRNAs on NP cells has also impacted a number of aspects, such as apoptosis, ECM degradation and synthesis, inflammatory response, autophagy and ageing. lncRNAs form a large and complex regulatory network with miRNAs, circRNAs and proteins. To date, most studies have focused on the role of lncRNAs in nucleus pulposus, and there has been little research on CEP and AF. Future research should endeavour to further the understanding of CEP and AF (Table I and Fig. 3).
Research progress on lncRNA expression in AS
AS is a chronic systemic disease caused by autoimmunity (91). The main affected population is adolescents or young adults (20–40 years of age), with a higher prevalence in men (92). AS mainly manifests as pain and stiffness in the lower back, gradually worsening and seriously affecting quality of life (93). At present, the pathogenesis of AS is not completely clear; treatment is mainly controlled by drugs, and the condition cannot be completely cured (94).
Cen et al (95) compared mesenchymal stem cells from patients with AS (ASMSCs) and healthy donors (HDMSCs) and found that HDMSCs had a stronger adipogenic differentiation ability. A total of 263 lncRNAs showed differential expression during adipogenesis in ASMSCs, and reverse transcription quantitative-polymerase chain reaction results showed that the expression of the top 10 lncRNAs was consistent with the high-throughput sequencing results. This is expected to provide new targets for future interventions in adipogenic metaplasia in AS (95). Another study found 200 upregulated and 70 downregulated lncRNAs in a comparison between patients with AS and healthy individuals (96). Wang et al (97) identified lncRNA 122K13.12 and lncRNA 326C3.7 from 205 lncRNAs that were differentially expressed in patients with AS and healthy individuals, and found that 30 miRNAs and 5 mRNAs formed a ceRNA signaling network together with these 2 lncRNAs; this signaling network may regulate the TNF-α signalling pathway in AS (97).
There are numerous differences in lncRNA expression between patients with AS and healthy individuals, thus warranting studies into the specific mechanisms governing this. Fang et al (98) found that NONHSAT227927.1 (a type of lncRNA) and tumour necrosis factor receptor-associated factor 2 (TRAF2) were significantly increased in the peripheral blood cells of patients with AS, and were positively associated with clinical inflammatory indicators. Xinfeng capsules, a traditional Chinese medicine, reduced immune inflammation in AS by inhibiting lncRNA NONHSAT227927.1/TRAF2. Furthermore, NONHSAT227927.1 was shown to activate the nuclear factor-κB-p65 pathway by promoting TRAF2 expression, thereby affecting the inflammatory process of AS (98). Osteoblasts are important regulators of bone formation; however, their specific mechanisms of action in AS remain unclear. Liu et al (99) collected serum and mesenchymal stem cells from patients with AS and healthy donors and found that MEG3 and TNFα-induced protein 3 (TNFAIP3) were downregulated, whereas miR-125a-5p was upregulated. Overexpression and knockdown experiments showed that MEG3 reduced osteogenic differentiation and inhibited AS progression through the miR-125a-5p/TNFAIP3/Wnt/β-catenin axis (99). As the first lncRNA discovered, H19 plays a regulatory role in diseases such as tumours and IDD, and affects the progression of inflammatory diseases such as AS. A total of three molecules, H19, VDR and TGF-β, can bind to miR22-5p and miR675-5p, and experiments have shown that H19 can regulate AS through the miR22-5p/miR675-5p/VDR-IL-17A/IL-23 signalling pathway (100). As a chronic spinal disease, AS causes severe pain, and understanding the role of lncRNAs in AS regulation provides a potential therapeutic strategy for AS (Table II).
Research progress on lncRNAs in SCI
SCI is a severe consequence of trauma to the spinal cord. The spinal cord is affected by external forces or chronic compression, and the corresponding sensory, motor and autonomic nervous functions are impaired owing to factors such as inflammation and apoptosis (101). Nerve cells have poor regenerative ability and cannot be cured after being damaged; therefore, treatment and rehabilitation after SCI are challenging (102). Differentially expressed lncRNAs play a regulatory role in SCI, providing new ideas for its treatment (103). Notably, menstrual blood-derived stem cells (MenSCs) have important therapeutic effects in degenerative and traumatic diseases, such as premature ovarian failure in mice (104) and cutaneous wound (105). Compared with SCI rats, rats treated with MenSCs exhibited significant upregulation of 89 lncRNAs and other RNAs, and significant downregulation of 65 lncRNAs and other RNAs. The lncRNA-miRNA-mRNA and circRNA-miRNA-mRNA ceRNA networks of SCI indicated that the role of lncRNAs in SCI may be mediated by a large interconnected regulatory network (103). Liu et al (106) examined changes in the expression of lncRNAs in the proximal tissue of the T10 layer at different time points during the right hemisection of T10 laminectomy. The expression of 68 lncRNAs first increased and then remained high 3 days after injury. Meanwhile, the expression of 56 lncRNAs decreased initially and remained low 3 days after injury. In a similar study, circulating exosomes were extracted from the blood of rats with SCI and control rats, and ncRNAs in the exosomes were identified, ultimately showing that the expression of ENSRN0T00000067908, XR_590093, XR_591455, XR_360081 and XR_346933 was upregulated, whereas the expression of XR_351404, XR_591426, XR_353833, XR_590076 and XR_590719 was downregulated. These 10 lncRNAs were at the centre of the constructed lncRNA-miRNA-mRNA co-expression network (107). These studies indicated that lncRNAs form important regulatory networks in SCI.
Growth-associated protein 43 (GAP43) is important for axonal growth and elongation. Hu et al (108) reported the protective role of vof16 (ischemia-related factor Vof-16) in SCI through the miR-185-5p-GAP43 regulatory network. Tanshinone IIA (TSIIA), a traditional Chinese medicine ingredient, can protect against SCI and tectonic family member 2 (TCTN2) expression has been shown to be low in an LPS-induced cell injury model. By contrast, TSIIA reduced cell damage and increased the expression of TCTN2; follow-up experiments demonstrated that TCTN2 may act as a regulator of dual-specificity phosphatase 1 (DUSP1) expression through miR-125a-5p; that is, TSIIA may regulate SCI through the TCTN2/miR-125a-5p/DUSP1 axis (109). In another previous study (110), researchers injected small extracellular vesicles (sEVs) derived from human umbilical cord mesenchymal stem cells induced by micro-electric field stimulation (EF-sEVs) and normally conditioned human umbilical cord mesenchymal stem cells-derived sEVs. Within the lesions of rats with SCI, MALAT1 expression was elevated in tissues treated with EF-sEVs. In addition, a luciferase reporter gene assay showed that MALAT1 can competitively bind to miR-22-3p and reduce its inhibitory effect on SIRT1, thereby improving SCI (110). A study has indicated that photobiomodulation (PBM) reduces inflammation by inhibiting bone marrow-derived macrophages. Transcriptome sequencing and bioinformatics analyses indicated that taurine upregulated gene 1 (TUG1) may be a target of PBM, and that TUG1 could competitively bind to miR-1192 and reduce miR-1192-induced inhibition of TLR3, leading to increased TLR3 expression and promotion of nerve survival and motor recovery (111) (Table II).
Treatment of spinal diseases based on lncRNAs
The role of lncRNAs in treating spinal diseases is mostly exerted by reducing or increasing the effective expression of the corresponding lncRNAs. Several methods can reduce the expression of lncRNAs. Firstly, silencing lncRNAs, such as siPART1, can enhance the growth of NP cells and increase the synthesis of the ECM, which can be used to treat IDD (67). Secondly, in a previous study, changing the chemical modification of lncRNAs increased WTAP expression in degenerated NP cells and significantly promoted the m6 modification of NORAD, promoting cell senescence. Therefore, therapeutic purposes can be achieved by reducing the m6 modification of NORAD (90). Finally, the formation of a complex with lncRNAs can interfere with its function; for example, some small molecule inhibitors can hide the functional sites of lncRNAs and interfere with their functions. Notably, BRD4 can bind to the HOTAIR promoter, affect HOTAIR function and control glioblastoma (112).
Methods to increase lncRNA expression include lncRNA mimics transfection. A previous study reported that KLF3-AS1 overexpression may improve IDD through the miR-10a-3p/ZBTB20 axis (70). Another method includes exogenously supplementing lncRNAs. Li et al (87) revealed that BMSC-Exo delivered exogenous CAHM can regulate macrophage polarisation and improve IDD (87). LncRNAs have therapeutic potential in spinal diseases, such as IDD, but some problems remain to be solved. When regulating the expression of lncRNAs, their impact cannot be accurately controlled, and their effect is not permanent and may decrease over time. Therefore, treating spinal diseases using lncRNAs requires further research to promote their progress.
Conclusions and outlook
Numerous lncRNAs have complex functions. The present article explains the classification and functions of lncRNAs and their roles in IDD, AS and SCI. In spinal diseases such as IDD, lncRNAs affect different aspects of disease progression, such as the ECM, cell death, inflammation and ageing. Taking IDD as an example, thousands of lncRNAs show differential expression in IDD compared with normal tissues. However, only a handful of lncRNAs have been studied. Increased research is required to fully understand the role of lncRNAs in spinal diseases, and experiments are needed to explore, supplement and improve ceRNA regulatory networks. Application of lncRNA in treating spinal diseases, such as IDD, is still in the preclinical experimental stage. However, lncRNAs have been used in the treatment of some malignant diseases (113) and have shown good efficacy; therefore, lncRNAs show great potential for clinical application in treating spinal diseases such as IDD.
In summary, lncRNAs are important regulatory factors affecting the occurrence and development of spinal diseases, such as IDD. However, current research on lncRNAs in spinal diseases has mainly focused on cells, rats and human degenerative tissues, and studies on more advanced large animals such as monkeys are lacking. Experimental and clinical treatments are meaningful directions for future research.
Acknowledgements
Not applicable.
Funding
This work was supported by the CuiYing Science and Technology Innovation plan project of Lanzhou University Second Hospital (grant no. CY2021-MS-A03).
Availability of data and materials
Not applicable.
Authors' contributions
ZM collected the literature and wrote the article; ZL and JA revised the article; ZM and ZL designed the study; and XL, XZ and SL prepared the figures and tables. ZM, XL, XZ, SL, JA and ZL contributed to data analysis, drafted and critically revised the paper, and agreed to be accountable for all aspects of the work. Data authentication is not applicable. All authors read and approved the final version of the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Wong CK, Mak RY, Kwok TS, Tsang JS, Leung MY, Funabashi M, Macedo LG, Dennett L and Wong AY: Prevalence, incidence, and factors associated with non-specific chronic low back pain in community-dwelling older adults aged 60 years and older: A systematic review and meta-analysis. J Pain. 23:509–534. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Speed C: Low back pain. BMJ. 328:1119–1121. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
GBD 2017, . Disease and Injury Incidence and Prevalence Collaborators: Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the global burden of disease study 2017. Lancet. 392:1789–1858. 2018.PubMed/NCBI | |
|
Samartzis D, Karppinen J, Mok F, Fong DYT, Luk KDK and Cheung KMC: A population-based study of juvenile disc degeneration and its association with overweight and obesity, low back pain, and diminished functional status. J Bone Joint Surg Am. 93:662–670. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Chen WK, Yu XH, Yang W, Wang C, He WS, Yan YG, Zhang J and Wang WJ: lncRNAs: Novel players in intervertebral disc degeneration and osteoarthritis. Cell Prolif. 50:e123132017. View Article : Google Scholar : PubMed/NCBI | |
|
Song C, Hu P, Peng R, Li F, Fang Z and Xu Y: Bioenergetic dysfunction in the pathogenesis of intervertebral disc degeneration. Pharmacol Res. 202:1071192024. View Article : Google Scholar : PubMed/NCBI | |
|
Ohnishi T, Sudo H, Tsujimoto T and Iwasaki N: Age-related spontaneous lumbar intervertebral disc degeneration in a mouse model. J Orthop Res. 36:224–232. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Rinaldi S, Moroni E, Rozza R and Magistrato A: Frontiers and challenges of computing ncRNAs biogenesis, function and modulation. J Chem Theory Comput. 20:993–1018. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Parasramka MA, Maji S, Matsuda A, Yan IK and Patel T: Long non-coding RNAs as novel targets for therapy in hepatocellular carcinoma. Pharmacol Ther. 161:67–78. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang C, Chen Z, Wang X, Zhang Y, Guo X, Xu Z, Yang H and Hao D: The potential mechanisms and application prospects of non-coding RNAs in intervertebral disc degeneration. Front Endocrinol (Lausanne). 13:10811852022. View Article : Google Scholar : PubMed/NCBI | |
|
Mehmandar-Oskuie A, Jahankhani K, Rostamlou A, Mardafkan N, Karamali N, Razavi ZS and Mardi A: Molecular mechanism of lncRNAs in pathogenesis and diagnosis of auto-immune diseases, with a special focus on lncRNA-based therapeutic approaches. Life Sci. 336:1223222024. View Article : Google Scholar : PubMed/NCBI | |
|
Lei HT, Wang JH, Yang HJ, Wu HJ, Nian FH, Jin FM, Yang J, Tian XM and Wang HD: LncRNA-mediated cell autophagy: An emerging field in bone destruction in rheumatoid arthritis. Biomed Pharmacother. 168:1157162023. View Article : Google Scholar : PubMed/NCBI | |
|
Jafari-Raddani F, Davoodi-Moghaddam Z, Yousefi AM, Ghaffari SH and Bashash D: An overview of long noncoding RNAs: Biology, functions, therapeutics, analysis methods, and bioinformatics tools. Cell Biochem Funct. 40:800–825. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Khan K, Irfan M, Sattar AA, Faiz MB, Rahman AU, Athar H, Calina D, Sharifi-Rad J and Cho WC: LncRNA SNHG6 role in clinicopathological parameters in cancers. Eur J Med Res. 28:3632023. View Article : Google Scholar : PubMed/NCBI | |
|
Monteiro JP, Bennett M, Rodor J, Caudrillier A, Ulitsky I and Baker AH: Endothelial function and dysfunction in the cardiovascular system: The long non-coding road. Cardiovasc Res. 115:1692–1704. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Dai L, Liang W, Shi Z, Li X, Zhou S, Hu W, Yang Z and Wang X: Systematic characterization and biological functions of non-coding RNAs in glioblastoma. Cell Prolif. 56:e133752023. View Article : Google Scholar : PubMed/NCBI | |
|
Jayasuriya R, Ganesan K, Xu B and Ramkumar KM: Emerging role of long non-coding RNAs in endothelial dysfunction and their molecular mechanisms. Biomed Pharmacother. 145:1124212022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Xu X and Su X: Modifications of noncoding RNAs in cancer and their therapeutic implications. Cell Signal. 108:1107262023. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang JH, Chen JH, Guo B, Fang Y, Xu ZY, Zhan L and Cao YX: Recent insights into noncoding RNAs in primary ovarian insufficiency: Focus on mechanisms and treatments. J Clin Endocrinol Metab. 108:1898–1908. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Liu X, Wang X, Li J, Hu S, Deng Y, Yin H, Bao X, Zhang QC, Wang G, Wang B, et al: Identification of mecciRNAs and their roles in the mitochondrial entry of proteins. Sci China Life Sci. 63:1429–1449. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Ponting CP, Oliver PL and Reik W: Evolution and functions of long noncoding RNAs. Cell. 136:629–641. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Statello L, Guo CJ, Chen LL and Huarte M: Gene regulation by long non-coding RNAs and its biological functions. Nat Rev Mol Cell Biol. 22:96–118. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Areeb Z, Stuart SF, West AJ, Gomez J, Nguyen HPT, Paradiso L, Zulkifli A, Jones J, Kaye AH, Morokoff AP and Luwor RB: Reduced EGFR and increased miR-221 is associated with increased resistance to temozolomide and radiotherapy in glioblastoma. Sci Rep. 10:177682020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang K, Shi ZM, Chang YN, Hu ZM, Qi HX and Hong W: The ways of action of long non-coding RNAs in cytoplasm and nucleus. Gene. 547:1–9. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Lazorthes S, Vallot C, Briois S, Aguirrebengoa M, Thuret JY, St Laurent G, Rougeulle C, Kapranov P, Mann C, Trouche D and Nicolas E: A vlincRNA participates in senescence maintenance by relieving H2AZ-mediated repression at the INK4 locus. Nat Commun. 6:59712015. View Article : Google Scholar : PubMed/NCBI | |
|
Mattick JS: A new paradigm for developmental biology. J Exp Biol. 210:1526–1547. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Fatica A and Bozzoni I: Long non-coding RNAs: New players in cell differentiation and development. Nat Rev Genet. 15:7–21. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Dueva R, Akopyan K, Pederiva C, Trevisan D, Dhanjal S, Lindqvist A and Farnebo M: Neutralization of the positive charges on histone tails by RNA promotes an open chromatin structure. Cell Chem Biol. 26:1436–1449.e5. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Li Y, Syed J and Sugiyama H: RNA-DNA triplex formation by long noncoding RNAs. Cell Chem Biol. 23:1325–1333. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Niehrs C and Luke B: Regulatory R-loops as facilitators of gene expression and genome stability. Nat Rev Mol Cell Biol. 21:167–178. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou X, Lv Y, Xie H, Li Y, Liu C, Zheng M, Wu R, Zhou S, Gu X, Li J and Mi D: RNA sequencing of exosomes secreted by fibroblast and Schwann cells elucidates mechanisms underlying peripheral nerve regeneration. Neural Regen Res. 19:1812–1821. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Wang L, Hu L, Wang X, Geng Z, Wan M, Hao J, Liu H, Fan Y, Xu T and Li Z: Long non-coding RNA LncCplx2 regulates glucose homeostasis and pancreatic β cell function. Mol Metab. 80:1018782024. View Article : Google Scholar : PubMed/NCBI | |
|
Hacisuleyman E, Goff LA, Trapnell C, Williams A, Henao-Mejia J, Sun L, McClanahan P, Hendrickson DG, Sauvageau M, Kelley DR, et al: Topological organization of multichromosomal regions by the long intergenic noncoding RNA Firre. Nat Struct Mol Biol. 21:198–206. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Naganuma T and Hirose T: Paraspeckle formation during the biogenesis of long non-coding RNAs. RNA Biol. 10:456–461. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Kopp F: Molecular functions and biological roles of long non-coding RNAs in human physiology and disease. J Gene Med. 21:e31042019. View Article : Google Scholar : PubMed/NCBI | |
|
Núñez-Martínez HN and Recillas-Targa F: Emerging functions of lncRNA loci beyond the transcript itself. Int J Mol Sci. 23:62582022. View Article : Google Scholar : PubMed/NCBI | |
|
Wang PS, Liu Z, Sweef O, Xie J, Chen J, Zhu H, Zeidler-Erdely PC, Yang C and Wang Z: Long noncoding RNA ABHD11-AS1 interacts with SART3 and regulates CD44 RNA alternative splicing to promote lung carcinogenesis. Environ Int. 185:1084942024. View Article : Google Scholar : PubMed/NCBI | |
|
Rashid F, Shah A and Shan G: Long non-coding RNAs in the cytoplasm. Genomics Proteomics Bioinformatics. 14:73–80. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Huang X, Zhou X, Hu Q, Sun B, Deng M, Qi X and Lü M: Advances in esophageal cancer: A new perspective on pathogenesis associated with long non-coding RNAs. Cancer Lett. 413:94–101. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Han I, Ropper AE, Konya D, Kabatas S, Toktas Z, Aljuboori Z, Zeng X, Chi JH, Zafonte R and Teng YD: Biological approaches to treating intervertebral disk degeneration: Devising stem cell therapies. Cell Transplant. 24:2197–2208. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Hu S, Xiu C, Li M, Zheng Y, Zhang R, Li B and Chen J: Intervertebral disc-intrinsic Hedgehog signaling maintains disc cell phenotypes and prevents disc degeneration through both cell autonomous and non-autonomous mechanisms. Cell Mol Life Sci. 81:742024. View Article : Google Scholar : PubMed/NCBI | |
|
Boubriak OA, Watson N, Sivan SS, Stubbens N and Urban JPG: Factors regulating viable cell density in the intervertebral disc: Blood supply in relation to disc height. J Anat. 222:341–348. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Woods BI, Vo N, Sowa G and Kang JD: Gene therapy for intervertebral disk degeneration. Orthop Clin North Am. 42563–574. (ix)2011. View Article : Google Scholar : PubMed/NCBI | |
|
Pooni JS, Hukins DW, Harris PF, Hilton RC and Davies KE: Comparison of the structure of human intervertebral discs in the cervical, thoracic and lumbar regions of the spine. Surg Radiol Anat. 8:175–182. 1986. View Article : Google Scholar : PubMed/NCBI | |
|
Shapiro IM, Vresilovic EJ and Risbud MV: Is the spinal motion segment a diarthrodial polyaxial joint: What a nice nucleus like you doing in a joint like this? Bone. 50:771–776. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Wang S, Sun J, Yang H, Zou W, Zheng B, Chen Y, Guo Y and Shi J: Profiling and bioinformatics analysis of differentially expressed circular RNAs in human intervertebral disc degeneration. Acta Biochim Biophys Sin (Shanghai). 51:571–579. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Ohnishi T, Iwasaki N and Sudo H: Causes of and molecular targets for the treatment of intervertebral disc degeneration: A review. Cells. 11:3942022. View Article : Google Scholar : PubMed/NCBI | |
|
Battié MC and Videman T: Lumbar disc degeneration: epidemiology and genetics. J Bone Joint Surg Am. 88 (Suppl 2):S3–S9. 2006. View Article : Google Scholar | |
|
Roberts S, Evans H, Trivedi J and Menage J: Histology and pathology of the human intervertebral disc. J Bone Joint Surg Am. 88 (Suppl 2):S10–S14. 2006. View Article : Google Scholar | |
|
Ye F, Lyu FJ, Wang H and Zheng Z: The involvement of immune system in intervertebral disc herniation and degeneration. JOR Spine. 5:e11962022. View Article : Google Scholar : PubMed/NCBI | |
|
Takahashi J, Shono Y, Hirabayashi H, Kamimura M, Nakagawa H, Ebara S and Kato H: Usefulness of white blood cell differential for early diagnosis of surgical wound infection following spinal instrumentation surgery. Spine (Phila Pa 1976). 31:1020–1025. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Tsujimoto T, Sudo H, Todoh M, Yamada K, Iwasaki K, Ohnishi T, Hirohama N, Nonoyama T, Ukeba D, Ura K, et al: An acellular bioresorbable ultra-purified alginate gel promotes intervertebral disc repair: A preclinical proof-of-concept study. EBioMedicine. 37:521–534. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Kuo CC, Soliman MAR, Baig RA, Aguirre AO, Ruggiero N, Donnelly BM, Siddiqi M, Khan A, Quiceno E, Mullin JP and Pollina J: Vertebral bone quality score as a predictor of adjacent segment disease after lumbar interbody fusion. Neurosurgery. Feb 9–2024.(Epub ahead of print). View Article : Google Scholar | |
|
Wan ZY, Song F, Sun Z, Chen YF, Zhang WL, Samartzis D, Ma CJ, Che L, Liu X, Ali MA, et al: Aberrantly expressed long noncoding RNAs in human intervertebral disc degeneration: A microarray related study. Arthritis Res Ther. 16:4652014. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, Ni H, Zhao Y, Chen K, Li M, Li C, Zhu X and Fu Q: Potential role of lncRNAs in contributing to pathogenesis of intervertebral disc degeneration based on microarray data. Med Sci Monit. 21:3449–3458. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Wu Y, Li S, Shen J, Wang Z and Liu H: Nucleus pulposus related lncRNA and mRNA expression profiles in intervertebral disc degeneration. Genomics. 115:1105702023. View Article : Google Scholar : PubMed/NCBI | |
|
Shi Y, Guo R, Zeng Y, Fang Q, Wang X, Liu W, Huang G and Wu W: SNHG5/miR-299-5p/ATF2 axis as a biomarker in immune microenvironment of intervertebral disc degeneration. Mediators Inflamm. 2022:25582752022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang Y, Zhang J, Sun Z, Wang H, Ning R, Xu L, Zhao Y, Yang K, Xi X and Tian J: MAPK8 and CAPN1 as potential biomarkers of intervertebral disc degeneration overlapping immune infiltration, autophagy, and ceRNA. Front Immunol. 14:11887742023. View Article : Google Scholar : PubMed/NCBI | |
|
Iwashina T, Mochida J, Sakai D, Yamamoto Y, Miyazaki T, Ando K and Hotta T: Feasibility of using a human nucleus pulposus cell line as a cell source in cell transplantation therapy for intervertebral disc degeneration. Spine (Phila Pa 1976). 31:1177–1186. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Chen X, Zhu L, Wu G, Liang Z, Yang L and Du Z: A comparison between nucleus pulposus-derived stem cell transplantation and nucleus pulposus cell transplantation for the treatment of intervertebral disc degeneration in a rabbit model. Int J Surg. 28:77–82. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Li Z, Li X, Chen C, Li S, Shen J, Tse G, Chan MTV and Wu WKK: Long non-coding RNAs in nucleus pulposus cell function and intervertebral disc degeneration. Cell Prolif. 51:e124832018. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang C, Liu Y, Zhao W, Yang Y, Ren Z, Wang X, Hao D, Du H and Yin S: microRNA-365 attenuated intervertebral disc degeneration through modulating nucleus pulposus cell apoptosis and extracellular matrix degradation by targeting EFNA3. J Cell Mol Med. 28:e180542024. View Article : Google Scholar : PubMed/NCBI | |
|
Lin Y, Jiao Y, Yuan Y, Zhou Z, Zheng Y, Xiao J, Li C, Chen Z and Cao P: Propionibacterium acnes induces intervertebral disc degeneration by promoting nucleus pulposus cell apoptosis via the TLR2/JNK/mitochondrial-mediated pathway. Emerg Microbes Infect. 7:12018. View Article : Google Scholar : PubMed/NCBI | |
|
Zhao K, An R, Xiang Q, Li G, Wang K, Song Y, Liao Z, Li S, Hua W, Feng X, et al: Acid-sensing ion channels regulate nucleus pulposus cell inflammation and pyroptosis via the NLRP3 inflammasome in intervertebral disc degeneration. Cell Prolif. 54:e129412021. View Article : Google Scholar : PubMed/NCBI | |
|
Du J, Xu M, Kong F, Zhu P, Mao Y, Liu Y, Zhou H, Dong Z, Yu Z, Du T, et al: CB2R attenuates intervertebral disc degeneration by delaying nucleus pulposus cell senescence through AMPK/GSK3β pathway. Aging Dis. 13:552–567. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang W, Zhao P and Zhang X: Apelin promotes ECM synthesis by enhancing autophagy flux via TFEB in human degenerative NP cells under oxidative stress. Biomed Res Int. 2020:48971702020.PubMed/NCBI | |
|
Gao D, Hao L and Zhao Z: Long non-coding RNA PART1 promotes intervertebral disc degeneration through regulating the miR-93/MMP2 pathway in nucleus pulposus cells. Int J Mol Med. 46:289–299. 2020.PubMed/NCBI | |
|
Yu X, Liu Q, Wang Y, Bao Y, Jiang Y, Li M, Li Z, Wang B, Yu L, Wang S, et al: Depleted long noncoding RNA GAS5 relieves intervertebral disc degeneration via microRNA-17-3p/Ang-2. Oxid Med Cell Longev. 2022:17924122022. View Article : Google Scholar : PubMed/NCBI | |
|
Hao Y, Zhu G, Yu L, Ren Z, Zhou W, Zhang P and Lian X: FOXO3-activated HOTTIP sequesters miR-615-3p away from COL2A1 to mitigate intervertebral disc degeneration. Am J Pathol. 194:280–295. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Chen S, Zhuang Q, Li P, Zeng J, Peng Y, Ding Z, Cao H, Zheng R and Wang W: The long non-coding RNA KLF3-AS1/miR-10a-3p/ZBTB20 axis improves the degenerative changes in human nucleus pulposus cells. Cell Tissue Res. 393:97–109. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Shang L, Ma H, Zhang X, Mao R, Ma C and Ruan Z: Docosahexaenoic acid alleviates the excessive degradation of extracellular matrix in the nucleus pulposus by reducing the content of lncRNA NEAT1 to prevent the progression of intervertebral disc degeneration. Clin Exp Pharmacol Physiol. 50:403–414. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Wang Z, Liu B, Ma X, Wang Y, Han W and Xiang L: lncRNA ZFAS1 promotes intervertebral disc degeneration by upregulating AAK1. Open Med (Wars). 17:1973–1986. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhu M, Yan X, Zhao Y, Xue H, Wang Z, Wu B, Li X and Shen Y: lncRNA LINC00284 promotes nucleus pulposus cell proliferation and ECM synthesis via regulation of the miR-205-3p/Wnt/β-catenin axis. Mol Med Rep. 25:1792022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhan S, Wang K, Xiang Q, Song Y, Li S, Liang H, Luo R, Wang B, Liao Z, Zhang Y and Yang C: lncRNA HOTAIR upregulates autophagy to promote apoptosis and senescence of nucleus pulposus cells. J Cell Physiol. 235:2195–2208. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhan S, Wang K, Song Y, Li S, Yin H, Luo R, Liao Z, Wu X, Zhang Y and Yang C: Long non-coding RNA HOTAIR modulates intervertebral disc degenerative changes via Wnt/β-catenin pathway. Arthritis Res Ther. 21:2012019. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou D, Mei Y, Song C, Cheng K, Cai W, Guo D, Gao S, Lv J, Liu T, Zhou Y, et al: Exploration of the mode of death and potential death mechanisms of nucleus pulposus cells. Eur J Clin Invest. e142262024.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI | |
|
Gao ZX, Lin YC, Wu ZP, Zhang P, Cheng QH, Ye LH, Wu FH, Chen YJ, Fu MH, Cheng CG and Gao YC: LncRNA SNHG6 can regulate the proliferation and apoptosis of rat degenerate nucleus pulposus cells via regulating the expression of miR-101-3p. Eur Rev Med Pharmacol Sci. 24:8251–8262. 2020.PubMed/NCBI | |
|
Yu J and Li C: Role of lncRNA MAGI2-AS3 in lipopolysaccharide-induced nucleus pulposus cells injury by regulating miR-374b-5p/interleukin-10 axis. Immun Inflamm Dis. 11:e7722023. View Article : Google Scholar : PubMed/NCBI | |
|
Yang W, Huang XD, Zhang T, Zhou YB, Zou YC and Zhang J: LncRNA MIR155HG functions as a ceRNA of miR-223-3p to promote cell pyroptosis in human degenerative NP cells. Clin Exp Immunol. 207:241–252. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Tao X, Xue F, Xu J and Wang W: Platelet-rich plasma-derived extracellular vesicles inhibit NF-κB/NLRP3 pathway-mediated pyroptosis in intervertebral disc degeneration via the MALAT1/microRNA-217/SIRT1 axis. Cell Signal. 117:1111062024. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Zhou Y, Huang T, Cheng ASL, Yu J, Kang W and To KF: The interplay of LncRNA-H19 and its binding partners in physiological process and gastric carcinogenesis. Int J Mol Sci. 18:4502017. View Article : Google Scholar : PubMed/NCBI | |
|
Sun Z, Tang X, Wang H, Sun H, Chu P, Sun L and Tian J: LncRNA H19 aggravates intervertebral disc degeneration by promoting the autophagy and apoptosis of nucleus pulposus cells through the miR-139/CXCR4/NF-κB axis. Stem Cells Dev. 30:736–748. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Guo C, Liu Y, Zhao Z, Wu Y, Kong Q and Wang Y: Regulating inflammation and apoptosis: A smart microgel gene delivery system for repairing degenerative nucleus pulposus. J Control Release. 365:1004–1018. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang X and Chen D: LncRNA FAM83H-AS1 maintains intervertebral disc tissue homeostasis and attenuates inflammation-related pain via promoting nucleus pulposus cell growth through miR-22-3p inhibition. Ann Transl Med. 8:15182020. View Article : Google Scholar : PubMed/NCBI | |
|
Luo Y, He Y, Wang Y, Xu Y and Yang L: LncRNA HCG18 promotes inflammation and apoptosis in intervertebral disc degeneration via the miR-495-3p/FSTL1 axis. Mol Cell Biochem. 479:171–181. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang C, Qiu Y and Yuan F: The long non-coding RNA maternally expressed 3-micorRNA-15a-5p axis is modulated by melatonin and prevents nucleus pulposus cell inflammation and apoptosis. Basic Clin Pharmacol Toxicol. 133:603–619. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Li W, Xu Y and Chen W: Bone mesenchymal stem cells deliver exogenous lncRNA CAHM via exosomes to regulate macrophage polarization and ameliorate intervertebral disc degeneration. Exp Cell Res. 421:1134082022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhong H, Zhou Z, Guo L, Liu FS, Wang X, Li J, Lv GH and Zou MX: SERPINA1 is a hub gene associated with intervertebral disc degeneration grade and affects the nucleus pulposus cell phenotype through the ADIRF-AS1/miR-214-3p axis. Transl Res. 245:99–116. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Wang X, Li D, Wu H, Liu F, Liu F, Zhang Q and Li J: LncRNA TRPC7-AS1 regulates nucleus pulposus cellular senescence and ECM synthesis via competing with HPN for miR-4769-5p binding. Mech Ageing Dev. 190:1112932020. View Article : Google Scholar : PubMed/NCBI | |
|
Li G, Ma L, He S, Luo R, Wang B, Zhang W, Song Y, Liao Z, Ke W, Xiang Q, et al: WTAP-mediated m6A modification of lncRNA NORAD promotes intervertebral disc degeneration. Nat Commun. 13:14692022. View Article : Google Scholar : PubMed/NCBI | |
|
Mauro D, Thomas R, Guggino G, Lories R, Brown MA and Ciccia F: Ankylosing spondylitis: An autoimmune or autoinflammatory disease? Nat Rev Rheumatol. 17:387–404. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Kenar G, Yarkan-Tuğsal H, Çetin-Özmen P, Solmaz D, Can G and Önen F: A lower frequency of inflammatory back pain in male patients with ankylosing spondylitis compared with female patients. Rheumatol Int. 44:477–482. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Braun J and Sieper J: Ankylosing spondylitis. Lancet. 369:1379–1390. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Ward MM, Deodhar A, Akl EA, Lui A, Ermann J, Gensler LS, Smith JA, Borenstein D, Hiratzka J, Weiss PF, et al: American college of rheumatology/spondylitis association of america/spondyloarthritis research and treatment network 2015 recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Rheumatol. 68:282–298. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Cen S, Cai M, Wang Y, Lu X, Chen Z, Chen H, Fang Y, Wu C, Qiu S and Liu Z: Aberrant lncRNA-mRNA expression profile and function networks during the adipogenesis of mesenchymal stem cells from patients with ankylosing spondylitis. Front Genet. 13:9918752022. View Article : Google Scholar : PubMed/NCBI | |
|
Huang D, Liu J, Wan L, Fang Y, Long Y, Zhang Y and Bao B: Identification of lncRNAs associated with the pathogenesis of ankylosing spondylitis. BMC Musculoskelet Disord. 22:2722021. View Article : Google Scholar : PubMed/NCBI | |
|
Wang JX, Jing FY, Xu YC, Zong HX, Chu YR, Wang C, Chen KM, Tong WQ, Wang XL and Xu SQ: The potential regulatory mechanism of lncRNA 122K13.12 and lncRNA 326C3.7 in ankylosing spondylitis. Front Mol Biosci. 8:7454412021. View Article : Google Scholar : PubMed/NCBI | |
|
Fang Y, Liu J, Xin L, Jiang H, Wen J, Li X, Wang F, He M and Han Q: Xinfeng capsule inhibits lncRNA NONHSAT227927.1/TRAF2 to alleviate NF-κB-p65-induced immuno-inflammation in ankylosing spondylitis. J Ethnopharmacol. 323:1176772024. View Article : Google Scholar : PubMed/NCBI | |
|
Liu C, Liang T, Zhang Z, Chen J, Xue J, Zhan X and Ren L: MEG3 alleviates ankylosing spondylitis by suppressing osteogenic differentiation of mesenchymal stem cells through regulating microRNA-125a-5p-mediated TNFAIP3. Apoptosis. 28:498–513. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang X, Ji S, Cai G, Pan Z, Han R, Yuan Y, Xu S, Yang J, Hu X, Chen M, et al: H19 increases IL-17A/IL-23 releases via regulating VDR by interacting with miR675-5p/miR22-5p in ankylosing spondylitis. Mol Ther Nucleic Acids. 19:393–404. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Baskozos G, Dawes JM, Austin JS, Antunes-Martins A, McDermott L, Clark AJ, Trendafilova T, Lees JG, McMahon SB, Mogil JS, et al: Comprehensive analysis of long noncoding RNA expression in dorsal root ganglion reveals cell-type specificity and dysregulation after nerve injury. Pain. 160:463–485. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Yang Y, Fan R, Li H, Chen H, Gong H and Guo G: Polysaccharides as a promising platform for the treatment of spinal cord injury: A review. Carbohydr Polym. 327:1216722024. View Article : Google Scholar : PubMed/NCBI | |
|
Qi L, Jiang W, He W, Li X, Wu J, Chen S, Liao Z, Yu S, Liu J, Sun Y, et al: Transcriptome profile analysis in spinal cord injury rats with transplantation of menstrual blood-derived stem cells. Front Mol Neurosci. 17:13354042024. View Article : Google Scholar : PubMed/NCBI | |
|
Wang Z, Wang Y, Yang T, Li J and Yang X: Study of the reparative effects of menstrual-derived stem cells on premature ovarian failure in mice. Stem Cell Res Ther. 8:112017. View Article : Google Scholar : PubMed/NCBI | |
|
Chen L, Qu J, Mei Q, Chen X, Fang Y, Chen L, Li Y and Xiang C: Small extracellular vesicles from menstrual blood-derived mesenchymal stem cells (MenSCs) as a novel therapeutic impetus in regenerative medicine. Stem Cell Res Ther. 12:4332021. View Article : Google Scholar : PubMed/NCBI | |
|
Liu W, Tao JC, Zhu SZ, Dai CL, Wang YX, Yu B, Yao C and Sun YY: Expression and regulatory network of long noncoding RNA in rats after spinal cord hemisection injury. Neural Regen Res. 17:2300–2304. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Li JA, Shi MP, Cong L, Gu MY, Chen YH, Wang SY, Li ZH, Zan CF and Wei WF: Circulating exosomal lncRNA contributes to the pathogenesis of spinal cord injury in rats. Neural Regen Res. 18:889–894. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Hu Y, Sun YF, Yuan H, Liu J, Chen L, Liu DH, Xu Y, Zhou XF, Ding L, Zhang ZT, et al: Vof16-miR-185-5p-GAP43 network improves the outcomes following spinal cord injury via enhancing self-repair and promoting axonal growth. CNS Neurosci Ther. 30:e145352024. View Article : Google Scholar : PubMed/NCBI | |
|
Yan Q, Xun Y, Lei D and Zhai H: Tanshinone IIA protects motor neuron-like NSC-34 cells against lipopolysaccharide-induced cell injury by the regulation of the lncRNA TCTN2/miR-125a-5/DUSP1 axis. Regen Ther. 24:417–425. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Li K, Liu Z, Wu P, Chen S, Wang M, Liu W, Zhang L, Guo S, Liu Y, Liu P, et al: Micro electrical fields induced MSC-sEVs attenuate neuronal cell apoptosis by activating autophagy via lncRNA MALAT1/miR-22-3p/SIRT1/AMPK axis in spinal cord injury. J Nanobiotechnology. 21:4512023. View Article : Google Scholar : PubMed/NCBI | |
|
Ju C, Ma Y, Zuo X, Wang X, Song Z, Zhang Z, Zhu Z, Li X, Liang Z, Ding T, et al: Photobiomodulation promotes spinal cord injury repair by inhibiting macrophage polarization through lncRNA TUG1-miR-1192/TLR3 axis. Cell Mol Biol Lett. 28:52023. View Article : Google Scholar : PubMed/NCBI | |
|
Pastori C, Kapranov P, Penas C, Peschansky V, Volmar CH, Sarkaria JN, Bregy A, Komotar R, St Laurent G, Ayad NG and Wahlestedt C: The bromodomain protein BRD4 controls HOTAIR, a long noncoding RNA essential for glioblastoma proliferation. Proc Natl Acad Sci USA. 112:8326–8331. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Li L, Gao Y, Yu B, Zhang J, Ma G and Jin X: Role of LncRNA H19 in tumor progression and treatment. Mol Cell Probes. 75:1019612024. View Article : Google Scholar : PubMed/NCBI | |
|
Cao S, Ma Y, Yang H, Luo G, Cheng H, Jin X and Sun T: Long noncoding RNA HCG18 promotes extracellular matrix degradation of nucleus pulposus cells in intervertebral disc degeneration by regulating the miR-4306/EPAS1 axis. World Neurosurg. 172:e52–e61. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Chen WK, Zhang HJ, Zou MX, Wang C, Yan YG, Zhan XL, Li XL and Wang WJ: LncRNA HOTAIR influences cell proliferation via miR-130b/PTEN/AKT axis in IDD. Cell Cycle. 21:323–339. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Chen W, Wang F, Wang J, Chen F and Chen T: The molecular mechanism of long non-coding RNA MALAT1-mediated regulation of chondrocyte pyroptosis in ankylosing spondylitis. Mol Cells. 45:365–375. 2022. View Article : Google Scholar : PubMed/NCBI |