Metastatic breast cancer with double heterozygosity for the BRCA1 and BRCA2 genes responding to olaparib: A case report
- Authors:
- Published online on: April 9, 2024 https://doi.org/10.3892/ol.2024.14387
- Article Number: 253
-
Copyright: © Shao et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Breast cancer is the most common type of cancer in women; one in 9–12 women will develop breast cancer in their lifetime in developed countries. Up to 10% of all breast cancer cases are inherited, and BRCA1 and BRCA2 gene mutations account for the majority of families with inherited breast cancer (1).
Individuals with a mutation in either BRCA1 or BRCA2 genes have a significantly increased lifetime risk of breast and ovarian cancer (2–4). Olaparib, a poly ADP-ribose polymerase (PARP) inhibitor, was approved by the US Food and Drug Administration in 2014 for the treatment of BRCA-positive advanced ovarian cancer and by the National Medical Products Administration in 2018. There are few reports of germline mutations in both BRCA1 and BRCA2 (5–8). Carriers with inherited deleterious mutations in both BRCA1 and BRCA2 only account for 0.3% of all female BRCA1/2 mutation carriers (5) and for 2.2% of breast cancer BRCA1/2 mutation carriers (6). Previous case reports have described such breast cancer patients but there is a lack of reports on the efficacy of olaparib on them (7,8). Similarly, clinical trials of olaparib have recruited such patients with cancer but lacked specific description of efficacy (9,10). A previous study reported that a patient with BRCA1 and BRCA2 double-germline mutant gastric cancer was resistant to olaparib treatment (11). To the best of our knowledge, however, there are no reports on the efficacy of olaparib in Chinese patients with breast cancer with mutations in both BRCA1 and BRCA2.
Case report
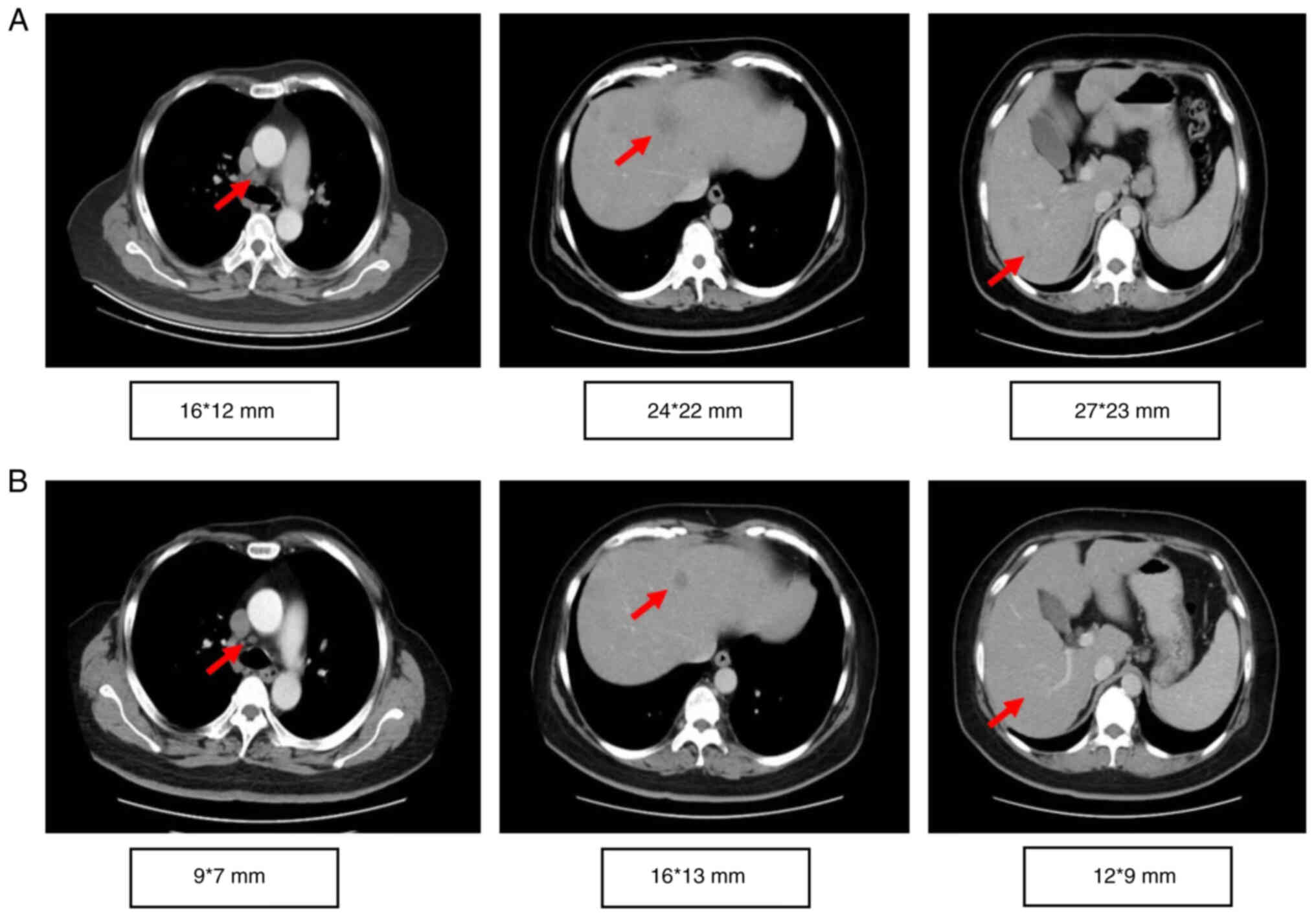
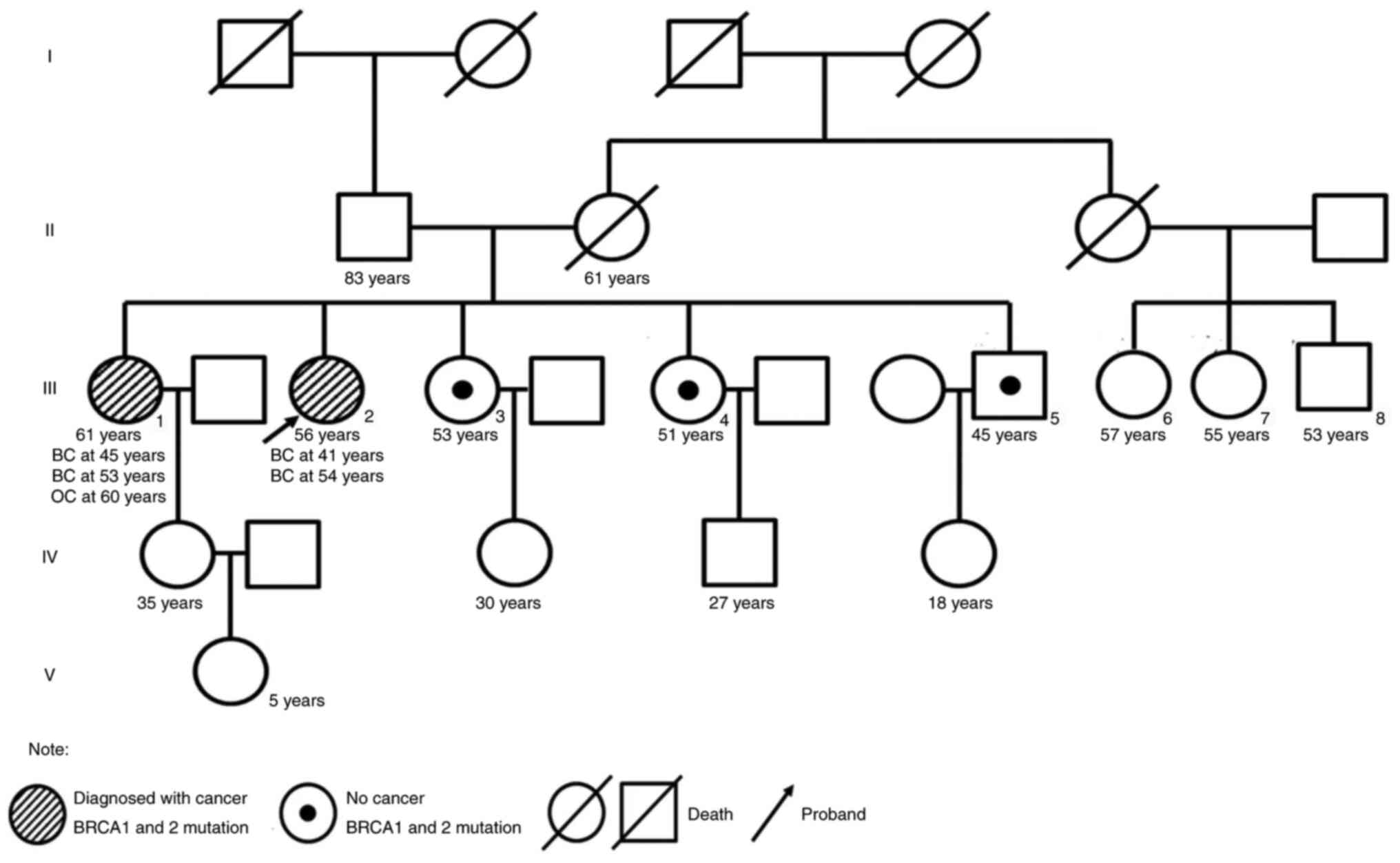
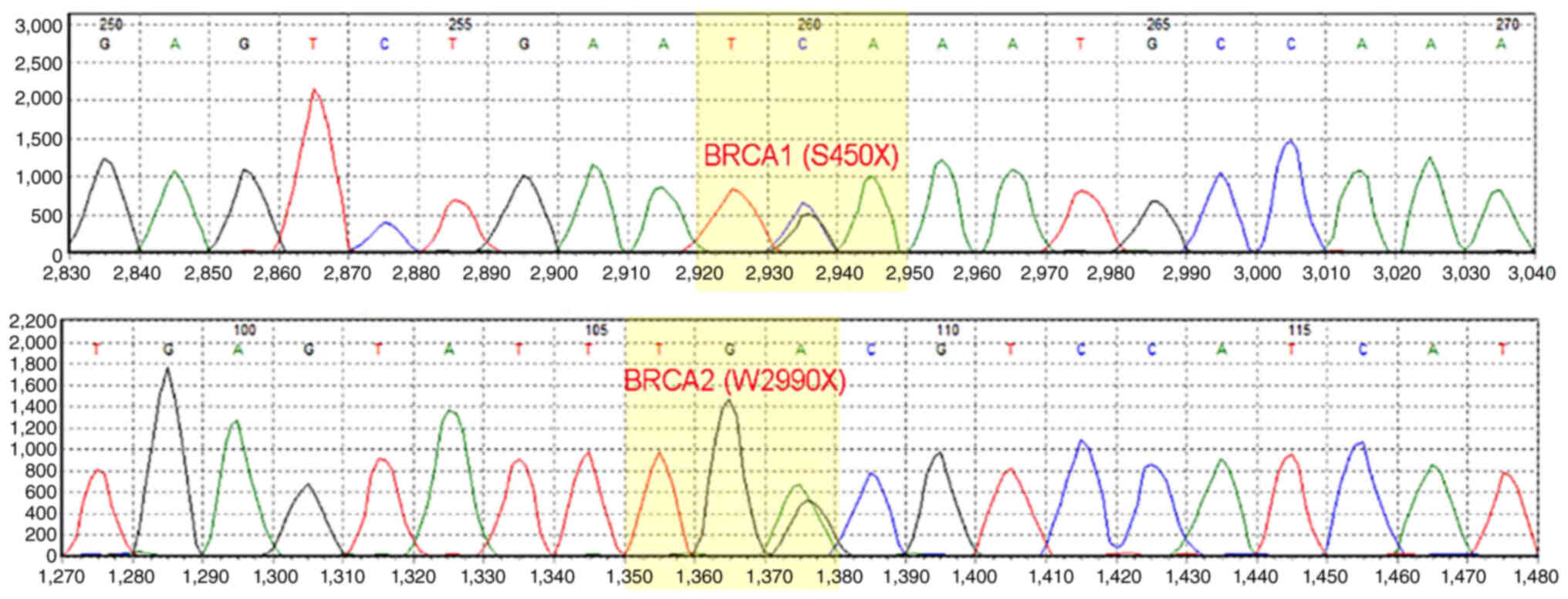
The proband, a 56-year-old woman, underwent a modified radical mastectomy for left breast cancer in May 2004. The pathological diagnosis was medullary carcinoma, pT2N0M0, stage IIA, estrogen receptor (ER)−, progesterone receptor (PR)− and human epidermal growth factor receptor (HER2)1+. The patient received four cycles of adriamycin/cyclophosphamide (dose unknown) regimen. Right breast cancer was diagnosed during the routine follow-up examination and a modified radical mastectomy was performed in May 2016. The pathological diagnosis was invasive ductal carcinoma, pT1N2M0, stage IIIA, ER+++, PR−, HER21+ and Ki-67 (30%). Germline or somatic mutations were not identified at this time. Based on clinical pathology, chemotherapy with epirubicin (90 mg/m2)/cyclophosphamide (600 mg/m2) for four cycles with weekly paclitaxel (80 mg/m2) for 12 weeks was performed from August 2016 to July 2017, followed by 5,000 cGy/25f radiotherapy targeting the chest wall and supraclavicular region. Letrozole (2.5 mg QD) was administered until multiple tumor metastases were detected in bone, liver and lymph nodes (left cervical lymph nodes, clavicular area and internal mammary, right hilar lymph nodes and mediastinal, lung and liver hilar and retroperitoneal lymph nodes) by PET/CT in October 2018. The proband refused biopsy of the metastatic sites. The proband had multiple liver metastases with a maximum metastatic focus 3.1×2.6 cm. The tumor burden was high with multiple liver, lymph node and bone metastases. Due to primary adjuvant endocrine resistance and palbociclib, the only available CDK4/6 inhibitor in China at that time (October 2018), was not affordable for the patient, the patient received combination chemotherapy. Chemotherapy with abraxane (125 mg/m2 d1,d8 q21d) combined with capecitabine (1 g/m2 d1-14 q21d) was initiated but the disease continued to progress in the liver after two cycles (Fig. 1). The timeline of her past medical history is outlined in Table I. The patient had a family history of breast and ovarian cancer (Fig. 2). Targeted next-generation sequencing testing with a panel of 18 cancer genetic susceptibility genes was performed. Genomic DNA (gDNA) was extracted from paraffin-embedded section of the primary tumor with AllPrep DNA/RNA mini Kit (Qiagen 80204). The libraries of gDNA were constructed with a KAPA Hyper Prep kit (Kapa Biosystems, USA) according to the operation manual. gDNA libraries were enriched through a panel of 18 cancer genetic susceptibility genes with its custom-designed capture probes were manufactured by Agilent, USA. P5/P7 primers were adopted to amplify the enriched gDNA libraries. The amplified libraries were qualified by the 2200 Bioanalyzer (Agilent Technologies, Palo Alto, Calif) and quantified by the qBittorrent (version 3). Paired-end sequencing with reads length of 250 bp on the Hiseq X Ten platform (Illumina, San Diego, CA) using MiSeq Reagent Kit v3 (MS-102-3001, Illumina, USA) and the loading concentration was 4 pM. The NGS raw data was initially analyzed with trimmomatic-0.36. Reads were then aligned against human reference genome (version GRCh37/hg19) with bwa (version 0.7.10). Candidate somatic mutations were determined using Samtools (version 1.3.1) and pindel (version 0.2.5b8). Finally, filter alignment and sequencing artifacts were conducted using IGV (Integrative Genomics Viewer). Double deleterious germline mutations in both BRCA1 (S405X) and BRCA2 (W2990X) were identified (Fig. 3). On the basis of these mutations, the proband started olaparib treatment (600 mg BID) in January 2019. The most severe adverse event was anemia (grade 3), which occurred 4 months after the start of the treatment. Other side effects, including leukopenia, thrombocytopenia, loss of appetite and nausea were mild and well tolerated. To alleviate symptoms of anemia, olaparib treatment was suspended and the proband received erythropoietin (10,000 IU 3 times/week) and ferrous succinate tablets (0.1 g 3 times/day). Olaparib treatment was resumed at a decreased dosage (450 mg/day) when anemia was reduced to grade 2 after 1 week of the treatment. Grade 3 anemia reoccurred after 2 weeks of decreased dosage olaparib, thus olaparib was suspended, and erythropoietin and ferrous succinate tablets at the aforementioned dosage were administered to relieve the anemia. Subsequently, olaparib treatment was resumed with further decreased dosage (300 mg/day). Grade 3 anemia reoccurred and olaparib treatment was terminated in July 2019. The olaparib treatment intermittently lasted for a total of 6 months and CT imaging showed a partial response (35% reduction; Fig. 1A and B). The proband did not receive chemotherapy or palbociclib because of persistent grade 3 anemia and the prohibitive cost of palbociclib. The proband received oral exemestane (25 mg qd). The patient continued treatment elsewhere and did not disclose her condition during a follow-up telephone conversation 3 months later.
The family members of the proband received genetic counseling and underwent NGS of BRCA genes. The same germline mutations in both BRCA1 and BRCA2 were identified in the proband's elder sister who was diagnosed with bilateral breast cancer at the age of 43 in 2005. Clinical information was not available. The proband's two younger sisters were carriers of the same BRCA1 mutation and her younger brother was a carrier of the same BRCA2 mutation. The offspring of the proband and their siblings declined genetic testing. To the best of our knowledge, no other family member has reported cancer symptoms to date.
Discussion
Hereditary breast/ovarian cancer syndrome, accounting for ~10% of breast and 15% of ovarian cancer cases, is often associated with germline mutations in BRCA1 or BRCA2 (3,12), which are present in 0.1–2.17% of the population worldwide (13–18). Due to the high risk of developing breast (up to 87%) and/or ovarian cancer (up to 63%), identification of carriers and surveillance is key to successful clinical management (19).
The frequency of double germline mutations in both BRCA1 and BRCA2 is only 0.3% in female BRCA1/2 mutation carriers (5), which accounts for 0.64–1.80% of BRCA1 (20) and 0.53–0.87% of BRCA2 deleterious mutations (6,21–26). Palmirotta et al (27) summarized worldwide literature on double mutations in BRCA1 and BRCA2 from 1998 to 2017, in which Jewish Ashkenazi patients were deliberately excluded due to high mutation frequency of BRCA1 and BRCA2. Only 34 families with 56 subjects (34 probands and 22 relatives) carrying double mutations of BRCA1 and BRCA2 in 20 articles were reported (27). It is unclear whether double mutations increase cancer risk. In the present study, two individuals (the proband and their elder sister) with the double mutations exhibited cancer symptoms while the single mutation carriers remained healthy, indicating a potential higher risk of cancer in double mutation carriers.
The characteristics of individuals with clinically significant double heterozygosity for BRCA1 and BRCA2 are poorly understood (28). Bilateral breast cancer, a characteristic of familial breast cancer, was present in both the proband and their elder sister in the present case report. Furthermore, previous studies have suggested that patients with both BRCA1 and BRCA2 are more susceptible to gastric cancer, pancreatic cancer, uterine cancer and prostate cancer in addition to breast cancer and ovarian cancer (29,30). Palmirotta et al (27) reported that the phenotype of 56 cases with double heterozygosity varied from unilateral breast cancer at age 26 to asymptomatic at age 72. A total of 42 cases (75%) had a primary tumor, including 35 patients with breast, two each with ovarian and prostate, and one each with cervix, caecum and stomach cancer; 14 of these cases suffered secondary neoplasia (27).
Notably, two pathologically different breast tumors occurred in the present proband (left triple negative medullary carcinoma, and right invasive ductal breast cancer with ER+++, PR− and HER21+). More extensive sequencing on tumor somatic genetic alterations may reveal differences in tumorigenesis as well as additional potential therapeutic targets.
Treatment of BRCA-mutated metastatic breast cancer with olaparib is recommended by the Chinese Society of Clinical Oncology (CSCO) Breast Cancer Guidelines (31) based on the clinical data showing slower spread of hereditary breast cancer with olaparib treatment (32). To the best of our knowledge, however, the response of metastatic breast cancer with double heterozygous BRCA1 and BRCA2 mutations to olaparib has not been described previously. To the best of our knowledge, the present study is the first report demonstrating a partial response to olaparib in such patients. Platinum is also an effective treatment for metastatic breast cancer with BRCA mutations (33). There are also other potential options for patients with hormone receptor-positive, HER2 negative breast cancer including CDK4/6 inhibitors and immunotherapy (34).
Since BRCA1 and BRCA2 genes are located on 13q and 17q respectively, each gene has the same probability (50%) of transmitting to the next generation (35). Genetic counseling is necessary in the medical care of familial breast cancer.
Family history and genetic counseling are key for the clinical management of patients with germline BRCA1/2 mutations (36). With the rapid development of treatment options, genetic screening for both germline and somatic mutations of BRCA is key for making clinical decisions, including type of surgery, the consideration of radiotherapy, and the value of systemic therapy in neoadjuvant and advanced settings (including response to platinum-based chemotherapy and PARP inhibitors) (37,38). The phase III trial OlympiAD has shown that compared with standard therapy, median progression-free survival (PFS) is 2.8 months longer and risk of disease progression or death is 42% lower with olaparib monotherapy in BRCA-mutated metastatic breast cancer (32). Thus, olaparib is a treatment option for metastatic breast cancer with germline mutations of BRCA1 and/or BRCA2 (38).
The present case described a 56-year-old female patient with rare BRCA1 and BRCA2 double germline-mutant metastatic breast cancer. The patient had a family history of breast and ovarian cancer. The patient exhibited primary resistance to both letrozole and albumin-bound paclitaxel combined with capecitabine. NGS showed germline mutations of both BRCA1 and BRCA2, and the patient received olaparib monotherapy. The patient achieved a partial response and PFS was 6 months, although they discontinued olaparib treatment due to continuous grade 3 anemia. Genetic testing is key to determine optimal treatment for breast cancer. In conclusion, olaparib exhibited therapeutic potential for a patient with BRCA1 and BRCA2 double germline-mutant metastatic breast cancer.
Acknowledgements
The authors would like thank Dr Yue Zhang (Genetron Health Beijing, China) for their constructive suggestions regarding the writing and revision of the manuscript.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available in the GSA-human repository, https://ngdc.cncb.ac.cn/gsa-human/s/30mKWWj6.
Authors' contributions
BS and LD confirm the authenticity of all the raw data. BS and LD conceived and designed the study. BS collected cases, analyzed data analysis. BS wrote the manuscript. BS and LD revised the content. LD was responsible for treating the patient. Both authors have read and approved the final manuscript.
Ethics approval and consent to participate
The patient and her relatives took part in the program ‘Genetic test of solid tumor, circulating tumor DNA and genetic diseases’, which was approved by the ethics committee of Beijing Cancer Hospital (Beijing, China; approval no. 2016XJS01-ZY01). All participants provided written informed consent to participate.
Patient consent for publication
Written informed consent was obtained from the patient and her family members for publication of this case report and any accompanying images.
Competing interests
The authors declare that they have no competing interests.
Glossary
Abbreviations
Abbreviations:
|
ER |
estrogen receptor |
|
PR |
progesterone receptor |
|
HER2 |
human epidermal growth factor receptor 2 |
|
PARP |
poly ADP-ribose polymerase |
|
PFS |
progression-free survival |
References
|
Stenehjem DD, Telford C, Unni SK, Bauer H, Sainski A, Deka R, Schauerhamer MB, Ye X, Tak CR, Ma J, et al: BRCA testing and outcomes in women with breast cancer. Breast Cancer Res Treat. 186:839–850. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Ford D, Easton DF, Bishop DT, Narod SA and Goldgar DE: Risks of cancer in BRCA1-mutation carriers. Breast cancer linkage consortium. Lancet. 343:692–695. 1994. View Article : Google Scholar : PubMed/NCBI | |
|
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, Liu Q, Cochran C, Bennett LM, Ding W, et al: A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 266:66–71. 1994. View Article : Google Scholar : PubMed/NCBI | |
|
Wooster R, Neuhausen SL, Mangion J, Quirk Y, Ford D, Collins N, Nguyen K, Seal S, Tran T, Averill D, et al: Localization of a breast cancer susceptibility gene, BRCA2, to chromosome 13q12-13. Science. 265:2088–2090. 1994. View Article : Google Scholar : PubMed/NCBI | |
|
Rebbeck TR, Friebel TM, Mitra N, Wan F, Chen S, Andrulis IL, Apostolou P, Arnold N, Arun BK, Barrowdale D, et al: Inheritance of deleterious mutations at both BRCA1 and BRCA2 in an international sample of 32,295 women. Breast Cancer Res. 18:1122016. View Article : Google Scholar : PubMed/NCBI | |
|
Lavie O, Narod S, Lejbkowicz F, Dishon S, Goldberg Y, Gemer O and Rennert G: Double heterozygosity in the BRCA1 and BRCA2 genes in the Jewish population. Ann Oncol. 22:964–966. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Meynard G, Mansi L, Lebahar P, Villanueva C, Klajer E, Calcagno F, Vivalta A, Chaix M, Collonge-Rame MA, Populaire C, et al: First description of a double heterozygosity for BRCA1 and BRCA2 pathogenic variants in a French metastatic breast cancer patient: A case report. Oncol Rep. 37:1573–1578. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Friedman E, Bar-Sade Bruchim R, Kruglikova A, Risel S, Levy-Lahad E, Halle D, Bar-On E, Gershoni-Baruch R, Dagan E, Kepten I, et al: Double heterozygotes for the Ashkenazi founder mutations in BRCA1 and BRCA2 genes. Am J Hum Genet. 63:1224–1227. 1998. View Article : Google Scholar : PubMed/NCBI | |
|
No authors listed. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med. 377:17002017. View Article : Google Scholar | |
|
Moore K, Colombo N, Scambia G, Kim BG, Oaknin A, Friedlander M, Lisyanskaya A, Floquet A, Leary A, Sonke GS, et al: Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 379:2495–2505. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Wen L, Li X, Shi J, Zhang S, Wang R, Yao M and Guo J: Allele-specific expression mediates primary resistance to poly (ADP-ribose) polymerase inhibitor therapy in a case of BRCA1/2 double-germline mutant gastric cancer. J Int Med Res. 48:3000605198862262020. View Article : Google Scholar : PubMed/NCBI | |
|
Wooster R, Bignell G, Lancaster J, Swift S, Seal S, Mangion J, Collins N, Gregory S, Gumbs C and Micklem G: Identification of the breast cancer susceptibility gene BRCA2. Nature. 378:789–792. 1995. View Article : Google Scholar : PubMed/NCBI | |
|
Lei H, Zhang M, Zhang L, Hemminki K, Wang XJ and Chen T: Overview on population screening for carriers with germline BRCA mutation in China. Front Oncol. 12:10023602022. View Article : Google Scholar : PubMed/NCBI | |
|
Fernández-Lopez JC, Romero-Córdoba S, Rebollar-Vega R, Alfaro-Ruiz LA, Jiménez-Morales S, Beltrán-Anaya F, Arellano-Llamas R, Cedro-Tanda A, Rios-Romero M, Ramirez-Florencio M, et al: Population and breast cancer patients' analysis reveals the diversity of genomic variation of the BRCA genes in the Mexican population. Hum Genomics. 13:32019. View Article : Google Scholar : PubMed/NCBI | |
|
Gabai-Kapara E, Lahad A, Kaufman B, Friedman E, Segev S, Renbaum P, Beeri R, Gal M, Grinshpun-Cohen J, Djemal K, et al: Population-based screening for breast and ovarian cancer risk due to BRCA1 and BRCA2. Proc Natl Acad Sci USA. 111:14205–14210. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Manickam K, Buchanan AH, Schwartz MLB, Hallquist MLG, Williams JL, Rahm AK, Rocha H, Savatt JM, Evans AE, Butry LM, et al: Exome sequencing-based screening for BRCA1/2 expected pathogenic variants among adult biobank participants. JAMA Netw Open. 1:e1821402018. View Article : Google Scholar : PubMed/NCBI | |
|
Momozawa Y, Iwasaki Y, Parsons MT, Kamatani Y, Takahashi A, Tamura C, Katagiri T, Yoshida T, Nakamura S, Sugano K, et al: Germline pathogenic variants of 11 breast cancer genes in 7,051 Japanese patients and 11,241 controls. Nat Commun. 9:40832018. View Article : Google Scholar : PubMed/NCBI | |
|
Maxwell KN, Domchek SM, Nathanson KL and Robson ME: Population frequency of germline BRCA1/2 mutations. J Clin Oncol. 34:4183–4185. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Smith A, Moran A, Boyd MC, Bulman M, Shenton A, Smith L, Iddenden R, Woodward ER, Lalloo F, Maher ER and Evans DGR: Phenocopies in BRCA1 and BRCA2 families: Evidence for modifier genes and implications for screening. J Med Genet. 44:10–15. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Frank TS, Deffenbaugh AM, Reid JE, Hulick M, Ward BE, Lingenfelter B, Gumpper KL, Scholl T, Tavtigian SV, Pruss DR and Critchfield GC: Clinical characteristics of individuals with germline mutations in BRCA1 and BRCA2: Analysis of 10,000 individuals. J Clin Oncol. 20:1480–1490. 2022. View Article : Google Scholar | |
|
Ramus SJ, Friedman LS, Gayther SA, Ponder BA, Bobrow L, van der Looji M, Papp J and Olah E: A breast/ovarian cancer patient with germline mutations in both BRCA1 and BRCA2. Nat Genet. 15:14–15. 1997. View Article : Google Scholar : PubMed/NCBI | |
|
Liede A, Rehal P, Vesprini D, Jack E, Abrahamson J and Narod SA: A breast cancer patient of Scottish descent with germ-line mutations in BRCA1 and BRCA2. Am J Hum Genet. 62:1543–1544. 1998. View Article : Google Scholar : PubMed/NCBI | |
|
Caldes T, de la Hoya M, Tosar A, Sulleiro S, Godino J, Ibañez D, Martin M, Perez-Segura P and Diaz-Rubio E: A breast cancer family from Spain with germline mutations in both the BRCA1 and BRCA2 genes. J Med Genet. 39:e442002. View Article : Google Scholar : PubMed/NCBI | |
|
Bell DW, Erban J, Sgroi DC and Haber DA: Selective loss of heterozygosity in multiple breast cancers from a carrier of mutations in both BRCA1 and BRCA2. Cancer Res. 62:2741–2743. 2002.PubMed/NCBI | |
|
Claus EB, Petruzella S, Matloff E and Carter D: Prevalence of BRCA1 and BRCA2 mutations in women diagnosed with ductal carcinoma in situ. JAMA. 293:964–969. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Zuradelli M, Peissel B, Manoukian S, Zaffaroni D, Barile M, Pensotti V, Cavallari U, Masci G, Mariette F, Benski AC, et al: Four new cases of double heterozygosity for BRCA1 and BRCA2 gene mutations: Clinical, pathological, and family characteristics. Breast Cancer Res Treat. 124:251–258. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Palmirotta R, Lovero D, Stucci LS, Silvestris E, Quaresmini D, Cardascia A and Silvestris F: Double heterozygosity for BRCA1 pathogenic variant and BRCA2 polymorphic stop codon K3326X: A case report in a Southern Italian family. Int J Mol Sci. 19:2852018. View Article : Google Scholar : PubMed/NCBI | |
|
Sugano K, Nakamura S, Ando J, Takayama S, Kamata H, Sekiguchi I, Ubukata M, Kodama T, Arai M, Kasumi F, et al: Cross-sectional analysis of germline BRCA1 and BRCA2 mutations in Japanese patients suspected to have hereditary breast/ovarian cancer. Cancer Sci. 99:1967–1976. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Nomizu T, Sakuma T, Yamada M, Matsuzaki M, Katagata N, Watanabe F, Nihei M, Ishioka C, Takenoshita S and Abe R: Three cases of kindred with familial breast cancer in which carrier detection by BRCA gene testing was performed on family members. Breast Cancer. 19:270–274. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Tobalina L, Armenia J, Irving E, O'Connor MJ and Forment JV: A meta-analysis of reversion mutations in BRCA genes identifies signatures of DNA end-joining repair mechanisms driving therapy resistance. Ann Oncol. 32:103–112. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang Z, Li J, Chen J, Liu Y, Wang K, Nie J, Wang X, Hao C, Yin Y, Wang S, et al: Chinese society of clinical oncology (CSCO) breast cancer guidelines 2022. Transl Breast Cancer Res. 3:132022. View Article : Google Scholar | |
|
Robson ME, Tung N, Conte P, Im SA, Senkus E, Xu B, Masuda N, Delaloge S, Li W, Armstrong A, et al: OlympiAD final overall survival and tolerability results: Olaparib versus chemotherapy treatment of physician's choice in patients with a germline BRCA mutation and HER2-negative metastatic breast cancer. Ann Oncol. 30:558–566. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Tutt A, Tovey H, Cheang MCU, Kernaghan S, Kilburn L, Gazinska P, Owen J, Abraham J, Barrett S, Barrett-Lee P, et al: Carboplatin in BRCA1/2-mutated and triple-negative breast cancer BRCAness subgroups: The TNT trial. Nat Med. 24:628–637. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Spring LM, Wander SA, Andre F, Moy B, Turner NC and Bardia A: Cyclin-dependent kinase 4 and 6 inhibitors for hormone receptor-positive breast cancer: past, present, and future. Lancet. 395:817–827. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Heidemann S, Fischer C, Engel C, Fischer B, Harder L, Schlegelberger B, Niederacher D, Goecke TO, Doelken SC, Dikow N, et al: Double heterozygosity for mutations in BRCA1 and BRCA2 in German breast cancer patients: Implications on test strategies and clinical management. Breast Cancer Res Treat. 134:1229–1239. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Samadder NJ, Giridhar KV, Baffy N, Riegert-Johnson D and Couch FJ: Hereditary cancer syndromes-A primer on diagnosis and management: Part 1: Breast-ovarian cancer syndromes. Mayo Clin Proc. 94:1084–1098. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Tung NM and Garber JE: BRCA1/2 testing: Therapeutic implications for breast cancer management. Br J Cancer. 119:141–152. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Daly MB, Pilarski R, Berry M, Buys SS, Farmer M, Friedman S, Garber JE, Kauff ND, Khan S, Klein C, et al: NCCN guidelines insights: Genetic/familial high-risk assessment: breast and ovarian, version 2.2017. J Natl Compr Canc Netw. 15:9–20.38. 2017.Robert M, Frenel JS, Gourmelon C, Patsouris A, Augereau P and Campone M: Olaparib for the treatment of breast cancer. Expert Opin Investig Drugs 26: 751–759, 2017. View Article : Google Scholar : PubMed/NCBI |