Prognostic significance of the neutrophil to lymphocyte ratio in locally advanced breast cancer
- Authors:
- Published online on: July 11, 2024 https://doi.org/10.3892/ol.2024.14562
- Article Number: 429
-
Copyright: © Alshamsan et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Breast cancer (BC) is the most frequently diagnosed malignancy, affecting 2.3 million women and causing nearly half a million deaths in 2022, thus representing one in four cancer cases and one in six cancer-associated deaths among women worldwide (1). Neoadjuvant chemotherapy (NAC) is the standard therapeutic method for locally advanced BC (LABC). NAC confers several advantages, including tumor downstaging to increase the viability of conservative surgery, aiding tumor response assessment and demonstrating comparable effectivity to adjuvant treatment to boost overall survival (OS) (2–4). Achieving a pathological complete response (pCR) following NAC serves as an important prognostic factor, especially when determining improved long-term outcomes (5–8). Conversely, residual disease would indicate the need for adjuvant therapy, particularly in triple-negative BC (TNBC) (9) and human epidermal growth factor receptor 2 (HER2)-positive disease (10).
The neutrophil-to-lymphocyte ratio (NLR) is a measurement that reflects tumor effect and host immune and inflammatory responses to the tumor, which has been shown to yield prognostic significance among various solid cancer types, including BC (11,12). A previous study has suggested that the NLR can be potentially used as an independent prognostic marker for disease-free survival (DFS) and OS in patients with BC, and should be assessed before surgical or medical treatment (13). However, conflicting findings regarding the association between pre-treatment NLR levels and pCR exist. A number of studies have revealed a significant association between lower NLR and higher rates of pCR regardless of molecular subtype (14,15). By contrast, others have identified an association with specific molecular subtypes, e.g., TNBC and HER2-positive disease (16–20), while other studies have found no such association between the NLR and pCR (21–29). Due to the diversity in ethnic backgrounds, the optimal NLR cut-off in the literature spans from 2.1 to 3.3, indicating the lack of a universally applicable threshold for the NLR. Furthermore, specific medical conditions, e.g., chronic inflammatory and autoimmune diseases, can influence NLR levels. Hence, it is vital to employ a receiver operating characteristic (ROC) curve for NLR determination and adherence to eligibility criteria when selecting cases to evaluate the prognostic significance of the NLR (15,30). Since the NLR is a readily measurable parameter that can potentially aid in the management of patients with localized BC, further investigation is warranted.
Therefore, the present study aimed to evaluate the prognostic role of the pre-treatment NLR in the NAC response in patients with LABC. Specifically, the primary objective was to determine whether the NLR serves as a predictive variable for the pCR in patients with LABC. The secondary objective was to determine whether the pre-treatment NLR is associated with DFS and OS in patients with LABC.
Patients and methods
Patients
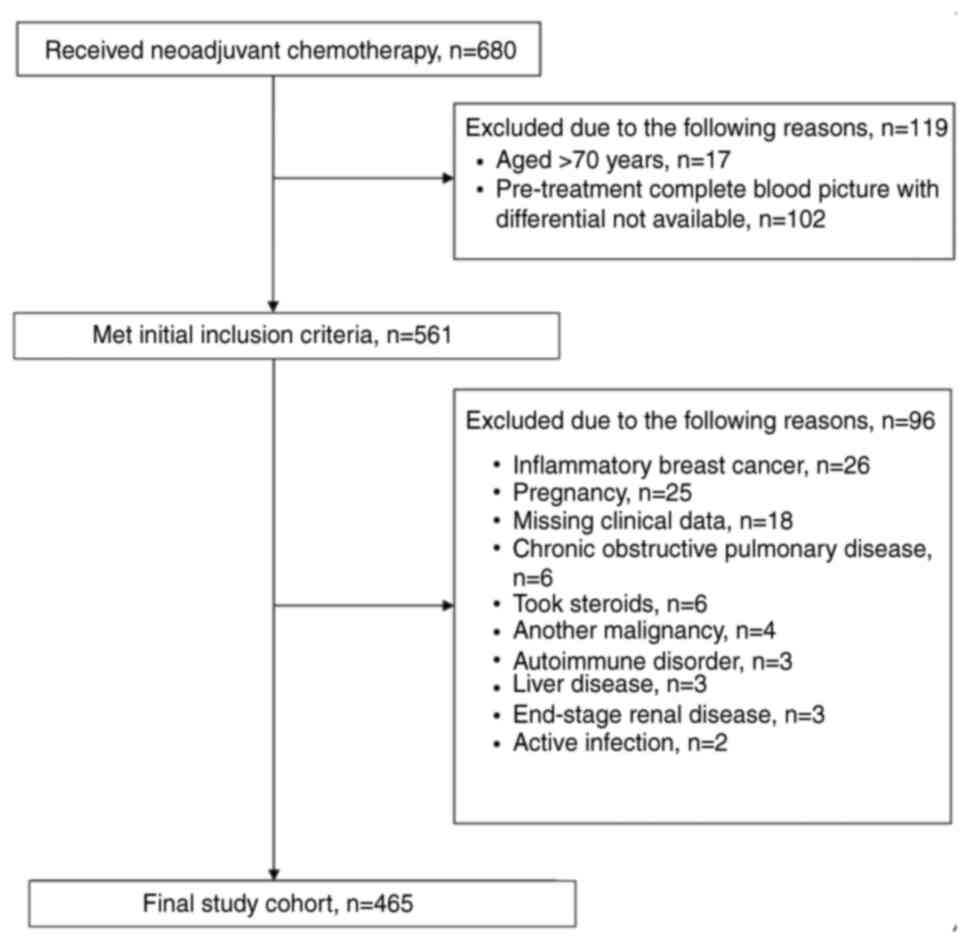
A retrospective review was conducted on patients who underwent standard NAC for LABC at King Feisal Specialist Hospital and Research Center (Riyadh, Saudi Arabia) between January 2005 and December 2014. The study protocol was approved by the Medical Ethics Committee and the Research Advisory Council (RAC) of the King Faisal Specialist Hospital and Research Center as part of the Breast Cancer Research Project (approval no. RAC#2051-029). Informed consent was waived by the Medical Ethics Committee owing to the retrospective nature of the study with assurance that all participants remained anonymous, where no identifying information or protected health data were collected. The study adhered to the Declaration of Helsinki and to the REMARK guidelines (31,32). Eligible patients include those with LABC who underwent NAC and breast surgery, were aged 18–70 years and had complete medical records (including surgical pathology assessment data and pre-NAC differential leukocyte count). Exclusion criteria were the presence of inflammatory BC, pregnancy-related BC, autoimmune diseases, hematological disorders, active infection, chronic obstructive pulmonary disease requiring treatment, chronic liver disease, end-stage renal disease, other malignancies, a history of cerebrovascular accidents, use of steroidal medication or missing clinical data.
The data was retrieved from a BC prospective database at King Faisal Specialist Hospital and Research Center. Two investigators independently reviewed the medical records of each patient. Collected data included age at diagnosis, menopausal status, tumor histology, grade, nodal stages, tumor size, immunohistochemistry results for estrogen and progesterone, HER2 upregulation, neoadjuvant chemotherapy protocol, type of surgery and radiation therapy.
Pathology slides were confirmed by a specialized BC pathologist at the institution, and IHC and fluorescence in situ hybridization analyses were performed as previously described. Estrogen or progesterone positivity was defined as ≥1% of the cells having nuclear receptors on immunohistochemical analysis (33). HER2 status was determined by immunohistochemical scoring. HER2 would be considered negative if the score was 0 or 1+ and positive in scores 3+ or 2+, and results were confirmed by fluorescence in situ hybridization. HER2 positivity would be deemed if the HER2-to-chromosome enumeration probe (CEP) 17 ratio was >2 with a HER2 copy number of ≥4 (34,35). The clinical staging of BC followed the Tumor-Node-Metastasis classification system from the American Joint Committee on Cancer and the Union for International Cancer Control, 8th edition (36). White blood cell differential count was obtained post-BC diagnosis and pre-NAC. A pCR was defined in cases of complete absence of viable invasive tumor cells upon pathological examination of both breast and axilla specimens, including surgical margins (ypT0 ypN0). DFS time was defined as the time span between histological diagnosis and disease recurrence or death, whereas OS time was defined as the period between histological diagnosis and the date of death.
Statistical analysis
Continuous variables are presented as median values with interquartile ranges and were compared using the Mann-Whitney U test. Categorical variables are presented as n (%) and were compared using the χ2 test or Fisher's exact test. The NLR cut-off point was determined using the ROC curve. The study population was stratified into low and high NLR groups. Univariate and multivariate logistic regression analyses were used to estimate the associations between various parameters and a pCR. The Kaplan-Meier method was used to estimate probabilities for DFS and OS, and log-rank analysis was used to compare the groups. The hazard ratio (HR) was estimated using Cox regression analysis. P<0.05 was considered to indicate a statistically significant difference. All statistical analyses were performed using SPSS for Mac (v28; IBM Corp.).
Results
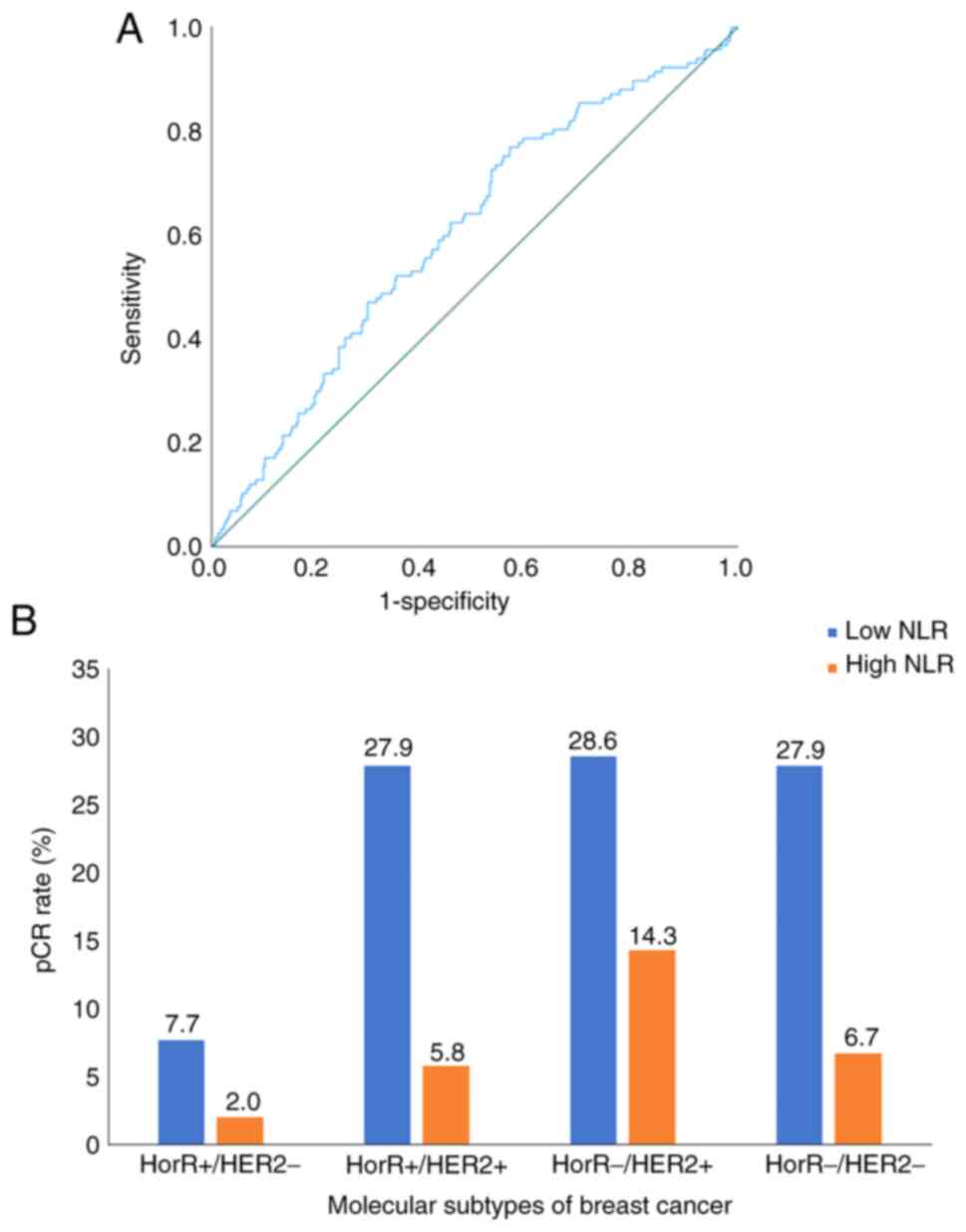
The present study included 465 consecutive patients who underwent NAC between January 2005 and December 2014. The flow diagram for the study population is illustrated in Fig. 1. The median age at diagnosis was 44 years (interquartile range, 38–50 years), where the median NLR was 1.91 (interquartile range, 1.27–2.65). The pre-treatment NLR was found to be significantly associated with pCR as a continuous variable (P<0.001), with a cut-off point of 2.2, 77% sensitivity and 57% specificity (Fig. 2A). The patients were thereafter stratified into low NLR (≤2.2; 291 patients; 62.6%) and high NLR (>2.2; 174 patients; 37.4%) groups. No significant differences were observed between the two groups in terms of patient demographics, disease and management characteristics (Table I). However, the pCR rate was higher in the low NLR group (30.9%) compared with that in the high NLR group (15.5%) (P<0.001).
Overall, the pCR rate was 25.2%, varying among molecular subtypes as follows: Hormone receptor (HorR)-/HER2+ disease (42.9%), HorR+/HER2+ (33.7%), HorR-/HER2-(34.6%) and HorR+/HER2-(9.7%). The pCR rate of molecular subtypes as stratified by NLR groups is presented in Fig. 2B. Multivariate analysis showed that pCR was independently associated with the NLR (OR, 2.59; 95% CI, 1.52–4.38; P<0.001), along with molecular subtype (HorR+/HER2+: OR, 5.64; 95% CI, 2.77–11.48; P<0.001; HorR-/HER2+: OR, 9.58; 95% CI, 4.58–20.01; P<0.001; HorR-/HER2-: OR, 6.25; 95% CI, 3.10–12.60; P<0.001) and N stage (OR, 0.51; 95% CI, 0.29–0.88; P=0.01) (Table II).
Table II.Univariate and multivariate binary logistic regression of variables associated with pathological complete response in patients with locally advanced breast cancer. |
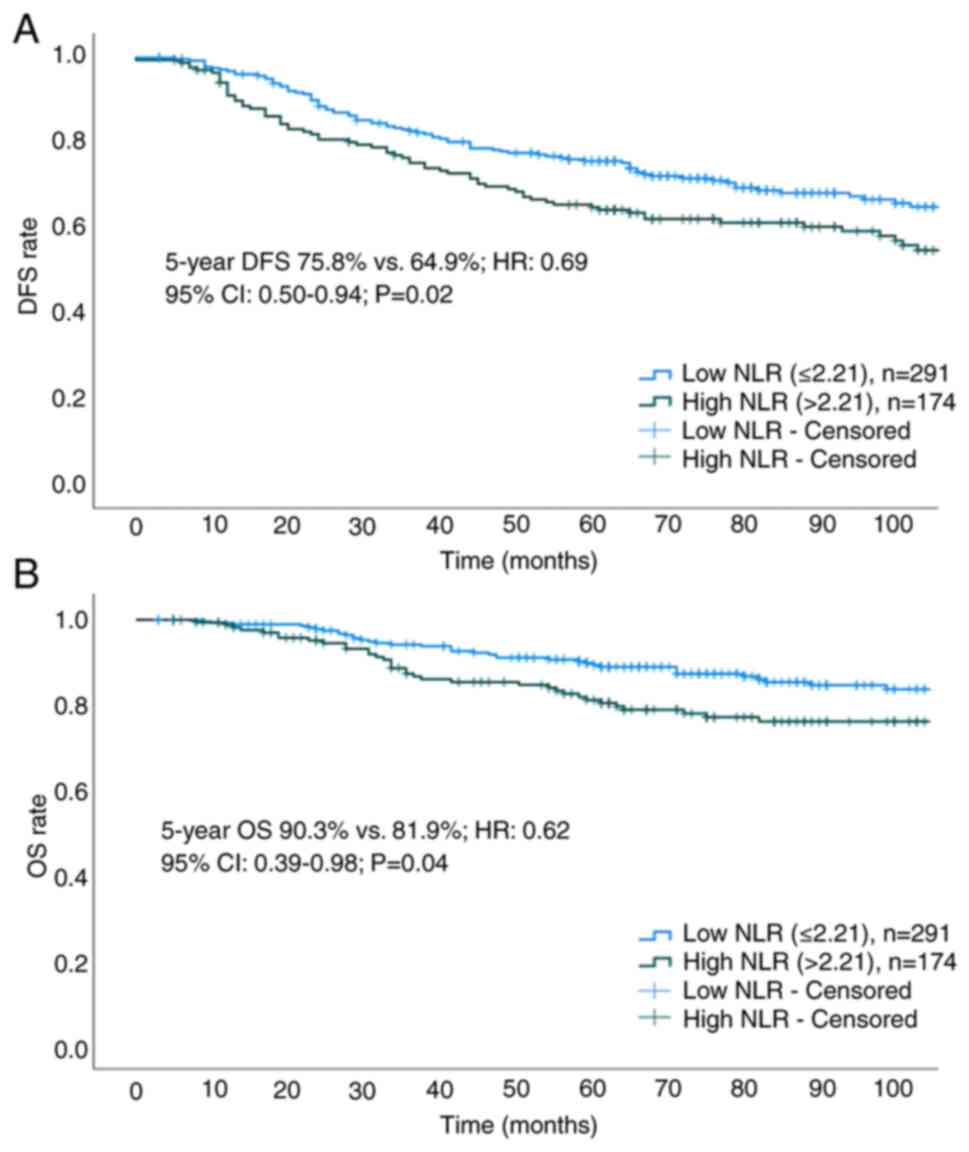
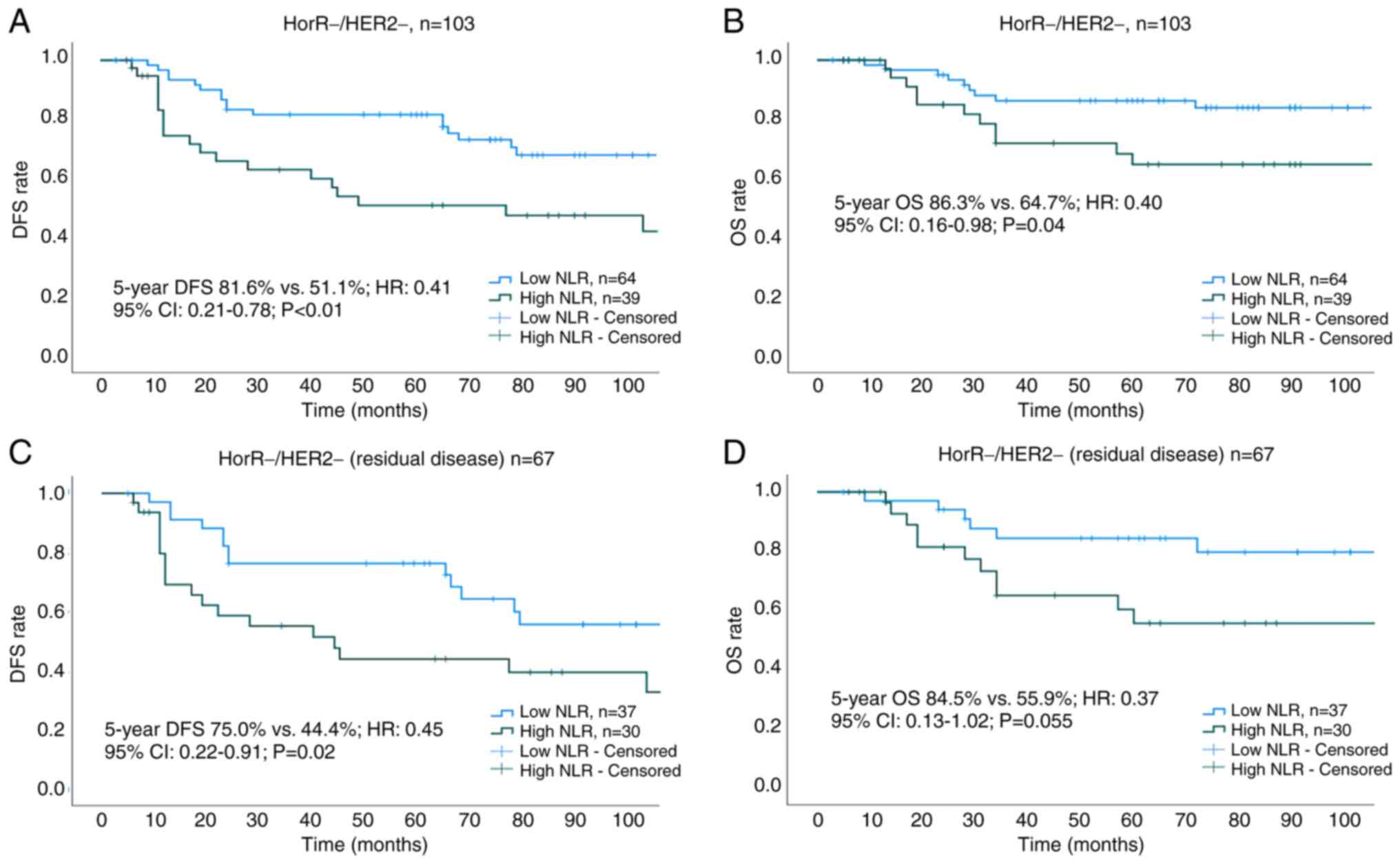
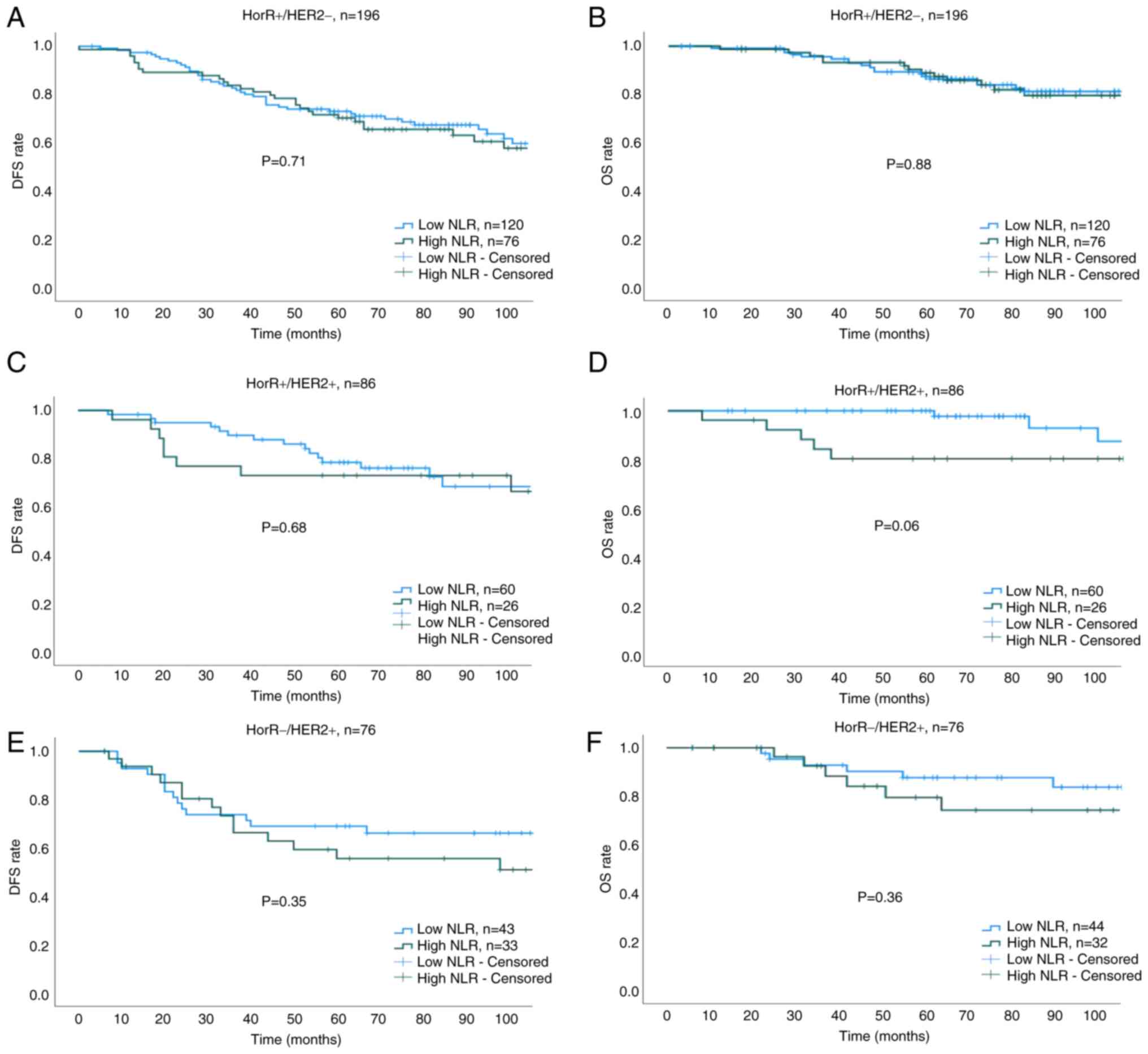
The median duration of follow-up was 77 months (interquartile range, 55–110 months). The 5-year DFS (75.8 vs. 64.9%; HR, 0.69; 95% CI, 0.50–0.94; P=0.02) and OS (90.3 vs. 81.9%; HR, 0.62; 95% CI, 0.39–0.98; P=0.04) rates were found to be higher in the low NLR group compared with those in the high NLR group (Fig. 3A and B). Cox regression analysis showed that a low NLR, T stage, N1 disease and pCR were associated with prolonged DFS and OS (Table III). Subgroup analysis of survival outcomes among molecular subtypes revealed statistical significance only in patients with TNBC (Fig. 4A and B). The 5-year DFS and OS rates for low vs. high NLR groups across all molecular subtypes were as follows: HorR+/HER2- (DFS: 73.4 vs. 70.6%; P=0.71; OS: 88.4 vs. 87.4%; P=0.88; Fig. 5A and B), HorR+/HER2+ (DFS: 78.5 vs. 73.1%; P=0.68; OS: 97.7 vs. 80.1%; P=0.06; Fig. 5C and D), HorR-/HER2+ (DFS: 69.3 vs. 55.7%; P=0.35; OS: 83.6 vs. 79.4%; P=0.36; Fig. 5E and F), and HorR-/HER2- (DFS: 81.6 vs. 51.1%; P<0.01; OS: 86.3 vs. 64.7%; P=0.04; Fig. 4A and B). Notably, among patients with TNBC with residual disease (no-pCR), those with a high pre-treatment NLR exhibited lower 5-year DFS (44.4 vs. 75.0%; P=0.02) and 5-year OS (55.9 vs. 84.5%, P=0.055) rates compared with those patients with a low pre-treatment NLR (Fig. 4C and D).
Table III.Cox regression analysis of variables associated with DFS and OS in patients with localized breast cancer who received neoadjuvant chemotherapy. |
Discussion
The present study aimed to evaluate the prognostic significance of the pre-treatment NLR for predicting a pCR following NAC in patients with LABC, in addition to its association with DFS and OS. The pre-treatment NLR was found to serve as a viable independent predictor of pCR after NAC in patients with LABC and as a prognostic factor for DFS and OS in patients with TNBC (including those with residual disease). This finding emphasizes the potential value of the NLR in BC prognostics, particularly in patients with TNBC, who frequently exhibit poorer clinical outcomes and have limited targeted treatment options.
Although numerous studies have previously explored the association between the NLR and pathological response to NAC, results have been inconsistent (14–28). This is possibly due to the retrospective study designs, variations in sample sizes, factors influencing the NLR and heterogenous NLR cut-off values (15,30). In addition, the lack of a standardized NLR cut-off is a major limitation in its use as a prognostic factor; it is typically influenced by multiple factors, including ethnicity, age, comorbidities and medications, e.g., steroids, that affect neutrophil and lymphocyte counts. Therefore, in the present study, an NLR cut-off of 2.2 was derived from the study population using the ROC curve of the 465 patients. This was found to be in the range of 1.8–4 reported by previous studies (5,16,30,37–39).
Achieving a pCR in BC has been extensively studied. A pCR was found to be associated with an improved prognosis, which can guide adjuvant treatment planning and serve as a surrogate endpoint in clinical trials (5,8,39,40). Several prognostic factors associated with increased pCR in BC, including smaller tumor size, TNBC, HER2-positive disease, higher tumor grade, younger patient age, high tumor-infiltrating lymphocyte (TIL) count, and use of anthracycline and taxane-based regimens (5,8,41,42). In particular, a high number of TILs, among other immune cells, has emerged as an important indicator of chemotherapy response and survival in BC (42). The immunogenic nature of TNBC underscores its distinct characteristics, as evidenced by significantly increased pCR rates when pembrolizumab is added to a NAC protocol with four cycles of paclitaxel and carboplatin, and four cycles of doxorubicin and cyclophosphamide, compared with a placebo (64.8 vs. 51.2%), irrespective of programmed death-ligand 1 expression (43). These factors highlight the complex interplay among tumor biology, host immune response and treatment strategies in determining the likelihood of achieving a pCR in BC. Therefore, recognizing and comprehending these factors can facilitate the optimization of treatment strategies, potentially leading to improved patient outcomes. However, it is imperative to acknowledge that individual responses to treatment may vary. Although achieving a pCR is of importance, it is not the sole determinant of long-term survival.
The present study observed an overall pCR rate of 25.2%, with the highest rate observed in HER2-positive and TNBC subtypes, which is consistent with previous research (30,39,44). However, the pre-treatment NLR was independently associated with pCR across all molecular subtypes, highlighting its significance. Furthermore, the high pre-treatment NLR was associated with poor survival outcomes (DFS and OS), which aligns with the result of a previous meta-analysis that included 42 studies (45). Subgroup analysis revealed a statistically significant association between the NLR and survival outcomes only in patients with TNBC. The underlying mechanisms linking the NLR to pCR in BC subtypes, particularly TNBC, remain unclear. Hypotheses based on preclinical and clinical studies suggest that elevated neutrophil levels and reduced lymphocyte levels can contribute to tumor growth, invasion and metastasis (46,47). TNBC is characterized by an inflammatory microenvironment that can promote tumor progression through mechanisms such as angiogenesis, extracellular matrix remodeling, immune evasion and development of treatment-resistance (5,38–50). Additionally, TNBC frequently exhibits high levels of TILs, which is associated with increased pCR rates in response to neoadjuvant therapy (42). Furthermore, results from the present study align with those from the previous reports on the association between the pre-treatment NLR and survival outcomes in patients with residual TNBC (16,27). This finding may have implications for guiding further treatment decisions in the adjuvant setting of patients with residual disease TNBC.
While the present study primarily investigated the association between the pre-treatment NLR and pCR, the importance of distinguishing the unique implications of both preoperative and postoperative NLR values on patient prognosis cannot be overlooked. The postoperative NLR holds notable prognostic significance and merits attention in oncological investigations; it can reflect the systemic inflammatory response following tumor removal, providing insights into the host immune response, modulation of the tumor microenvironment and residual disease burden. Previous studies reported that elevated NLR levels, both immediately within 1 week postoperatively and later, 5 years after diagnosis, were associated with poorer survival outcomes in BC (51–53).
The major strengths of the present study include the utilization of a large sample size of patients with LABC who received NAC and data obtained from a prospective database, allowing for a comprehensive analysis of the NLR as a prognostic factor. Additionally, potential confounding factors that may impact the NLR were mitigated by following defined eligibility criteria and the NLR cut-off derived from the study population. However, limitations remain. The retrospective nature of the present study may introduce selection bias. In addition, the duration of follow-up may have been insufficient to fully capture long-term survival outcomes in patients with BC, particularly in molecular subtypes with a less aggressive disease course. The present study population was also drawn from a single institution, which may have limited the generalizability of the findings. Furthermore, the absence of tumor tissue data is a notable limitation, as it prevented the inclusion of TIL analysis, which could have provided additional insights into the prognostic landscape.
In conclusion, the present findings suggest that the pre-treatment NLR can serve as an independent prognostic factor for a pCR after NAC in patients with LABC and for survival outcomes, particularly in patients with TNBC (including those with residual disease). This highlights the potential of the NLR as a prognostic marker to identify patients who benefit most from NAC, particularly in TNBC, guiding treatment planning and patient counseling. However, further prospective multicenter studies are warranted to validate such findings and explore the underlying mechanisms linking the NLR and treatment response in different BC subtypes.
Acknowledgements
The early results of this study were presented as an E-Poster at the European Society for Medical Oncology (ESMO) 2020. The abstract was published in the Annals of Oncology as Supplement S344, available at http://www.annalsofoncology.org/article/S0923-7534(20)40069-9/fulltext.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be requested from the corresponding author.
Authors' contributions
BA conceptualized the present study. MAE, MA, NF, AB, AA, KS and BA performed data collection. AA, KS and TAT were involved in designing and managing the patient and BC database. BA and TAT confirm the authenticity of all of the raw data. BA and TAT performed a literature review and wrote the first draft of the manuscript. TE and BA performed the analysis and prepared tables and figures. All authors edited and approved the final version of the manuscript.
Ethics approval and consent to participate
The study was approved by the RAC of King Faisal Specialist Hospital and Research Centre (Riyadh, Saudi Arabia; approval no. 2051-029). The RAC committee at King Faisal Specialist Hospital and Research Centre waived the requirement for informed consent. All methods were performed in accordance with relevant guidelines and regulations.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Glossary
Abbreviations
Abbreviations:
|
HER2 |
human epidermal growth factor receptor 2 |
|
HR |
hazard ratio |
|
HorR |
hormone receptor |
|
LABC |
locally advanced breast cancer |
|
NAC |
neoadjuvant chemotherapy |
|
NLR |
neutrophil-to-lymphocyte ratio |
|
OS |
overall survival |
|
pCR |
pathological complete response |
|
RAC |
Research Advisory Council |
|
ROC |
receiver operating characteristic |
|
TIL |
tumor-infiltrating lymphocyte |
|
TNBC |
triple-negative breast cancer |
References
|
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 74:229–263. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Kaufmann M, Hortobagyi GN, Goldhirsch A, Scholl S, Makris A, Valagussa P, Blohmer JU, Eiermann W, Jackesz R, Jonat W, et al: Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: An update. J Clin Oncol. 24:1940–1949. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Rastogi P, Anderson SJ, Bear HD, Geyer CE, Kahlenberg MS, Robidoux A, Margolese RG, Hoehn JL, Vogel VG, Dakhil SR, et al: Preoperative chemotherapy: Updates of national surgical adjuvant breast and bowel project protocols B-18 and B-27. J Clin Oncol. 26:778–785. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Early Breast Cancer Trialists' Collaborative Group (EBCTCG), . Long-term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: Meta-analysis of individual patient data from ten randomised trials. Lancet Oncol. 19:27–39. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, et al: Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 30:1796–1804. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Fayanju OM, Ren Y, Thomas SM, Greenup RA, Plichta JK, Rosenberger LH, Tamirisa N, Force J, Boughey JC, Hyslop T and Hwang ES: The clinical significance of breast-only and node-only pathologic complete response (PCR) after neoadjuvant chemotherapy (NACT): A review of 20,000 breast cancer patients in the national cancer data base (NCDB). Ann Surg. 268:591–601. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Viale G: Characterization and clinical impact of residual disease after neoadjuvant chemotherapy. Breast. 22 (Suppl 2):S88–S91. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, Bonnefoi H, Cameron D, Gianni L, Valagussa P, et al: Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet. 384:164–172. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Masuda N, Lee SJ, Ohtani S, Im YH, Lee ES, Yokota I, Kuroi K, Im SA, Park BW, Kim SB, et al: Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N Engl J Med. 376:2147–2159. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Von Minckwitz G, Huang CS, Mano MS, Loibl S, Mamounas EP, Untch M, Wolmark N, Rastogi P, Schneeweiss A, Redondo A, et al: Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med. 380:617–628. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Templeton AJ, McNamara MG, Šeruga B, Vera-Badillo FE, Aneja P, Ocaña A, Leibowitz-Amit R, Sonpavde G, Knox JJ, Tran B, et al: Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: A systematic review and meta-analysis. J Natl Cancer Inst. 106:dju1242014. View Article : Google Scholar : PubMed/NCBI | |
|
Paramanathan A, Saxena A and Morris DL: A systematic review and meta-analysis on the impact of pre-operative neutrophil lymphocyte ratio on long term outcomes after curative intent resection of solid tumours. Surg Oncol. 23:31–39. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Faria SS, Fernandes PC Jr, Silva MJ, Lima VC, Fontes W, Freitas-Junior R, Eterovic AK and Forget P: The neutrophil-to-lymphocyte ratio: A narrative review. Ecancermedicalscience. 10:7022016.PubMed/NCBI | |
|
Qian Y, Tao J, Li X, Chen H, Lu Q, Yang J, Pan H, Wang C, Zhou W and Liu X: Peripheral inflammation/immune indicators of chemosensitivity and prognosis in breast cancer patients treated with neoadjuvant chemotherapy. Onco Targets Ther. 11:1423–1432. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, Chen K, Xiao X, Nie Y, Qu S, Gong C, Su F and Song E: Pretreatment neutrophil-to-lymphocyte ratio is correlated with response to neoadjuvant chemotherapy as an independent prognostic indicator in breast cancer patients: A retrospective study. BMC Cancer. 16:3202016. View Article : Google Scholar : PubMed/NCBI | |
|
Bae SJ, Cha YJ, Yoon C, Kim D, Lee J, Park S, Cha C, Kim JY, Ahn SG, Park HS, et al: Prognostic value of neutrophil-to-lymphocyte ratio in human epidermal growth factor receptor 2-negative breast cancer patients who received neoadjuvant chemotherapy. Sci Rep. 10:130782020. View Article : Google Scholar : PubMed/NCBI | |
|
Asano Y, Kashiwagi S, Onoda N, Noda S, Kawajiri H, Takashima T, Ohsawa M, Kitagawa S and Hirakawa K: Predictive value of neutrophil/lymphocyte ratio for efficacy of preoperative chemotherapy in triple-negative breast cancer. Ann Surg Oncol. 23:1104–1110. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Chae S, Kang KM, Kim HJ, Kang E, Park SY, Kim JH, Kim SH, Kim SW and Kim EK: Neutrophil-lymphocyte ratio predicts response to chemotherapy in triple-negative breast cancer. Curr Oncol. 25:e113–e119. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Rao JS, Hanumappa HK, Joseph EP, Chowdappa RG and Ramesh R: Elevated neutrophil-lymphocyte ratio in luminal-type locally advanced breast cancer to circumvent neo-adjuvant chemotherapy. Indian J Surg Oncol. 10:454–459. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Rivas M, Acevedo F, Dominguez F, Galindo H, Camus M, Oddo D, Villarroel A, Razmilic D, Peña J, Munoz Medel M, et al: The neutrophil to lymphocyte ratio predicts the response to neoadjuvant chemotherapy in luminal B breast cancer. Asian Pac J Cancer Prev. 20:2209–2212. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Coleman N, Rom FM, Brennan A, Bennett K, Jayaram AK and Kennedy MJ: Does baseline absolute neutrophil-to-lymphocyte ratio (Nlr) correlate with pathological complete response (Pcr) in neoadjuvant breast cancer (Bc)? Ann Oncol. 25 (Suppl 4):IV1132014. View Article : Google Scholar | |
|
Eryilmaz MK, Mutlu H, Salim DK, Musri FY, Tural D and Coskun HS: The neutrophil to lymphocyte ratio has a high negative predictive value for pathologic complete response in locally advanced breast cancer patients receiving neoadjuvant chemotherapy. Asian Pac J Cancer Prev. 15:7737–7740. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Koh YW, Lee HJ, Ahn JH, Lee JW and Gong G: Prognostic significance of the ratio of absolute neutrophil to lymphocyte counts for breast cancer patients with ER/PR-positivity and HER2-negativity in neoadjuvant setting. Tumour Biol. 35:9823–9830. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Suppan C, Bjelic-Radisic V, La Garde M, Groselj-Strele A, Eberhard K, Samonigg H, Loibner H, Dandachi N and Balic M: Neutrophil/lymphocyte ratio has no predictive or prognostic value in breast cancer patients undergoing preoperative systemic therapy. BMC Cancer. 15:10272015. View Article : Google Scholar : PubMed/NCBI | |
|
Graziano V, Grassadonia A, Iezzi L, Vici P, Pizzuti L, Barba M, Quinzii A, Camplese A, Di Marino P, Peri M, et al: Combination of peripheral neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio is predictive of pathological complete response after neoadjuvant chemotherapy in breast cancer patients. Breast. 44:33–38. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Marín Hernández C, Piñero Madrona A, Gil Vázquez PJ, Galindo Fernández PJ, Ruiz Merino G, Alonso Romero JL and Parrilla Paricio P: Usefulness of lymphocyte-to-monocyte, neutrophil-to-monocyte and neutrophil-to-lymphocyte ratios as prognostic markers in breast cancer patients treated with neoadjuvant chemotherapy. Clin Transl Oncol. 20:476–483. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Muñoz-Montaño W, Cabrera-Galeana P, Alvarado-Miranda A, Villarreal-Garza C, Mohar A, Olvera A, Bargallo-Rocha E, Lara-Medina F and Arrieta O: Prognostic value of the pretreatment neutrophil-to-lymphocyte ratio in different phenotypes of locally advanced breast cancer during neoadjuvant systemic treatment. Clin Breast Cancer. 20:307–316.e1. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Peng Y, Chen R, Qu F, Ye Y, Fu Y, Tang Z, Wang Y, Zong B, Yu H, Luo F and Liu S: Low pretreatment lymphocyte/monocyte ratio is associated with the better efficacy of neoadjuvant chemotherapy in breast cancer patients. Cancer Biol Ther. 21:189–196. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Bulut G and Ozdemir ZN: Significance of neutrophil-lymphocyte ratio and Trombocyte-lymphocyte ratio in predicting complete pathological response in patients with local advanced breast cancer. Eurasian J Med Invest. 61:78–83. 2022. | |
|
Cullinane C, Creavin B, O'Leary DP, O'Sullivan MJ, Kelly L, Redmond HP and Corrigan MA: Can the neutrophil to lymphocyte ratio predict complete pathologic response to neoadjuvant breast cancer treatment? A systematic review and meta-analysis. Clin Breast Cancer. 20:e675–e681. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M and Clark GM; Statistics Subcommittee of NCI-EORTC Working Group on Cancer Diagnostics, : REporting recommendations for tumor MARKer prognostic studies (REMARK). Breast Cancer Res Treat. 100:229–235. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Sauerbrei W, Taube SE, McShane LM, Cavenagh MM and Altman DG: Reporting recommendations for tumor marker prognostic studies (REMARK): An abridged explanation and elaboration. J Natl Cancer Inst. 110:803–811. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Hammond ME, Hayes DF, Wolff AC, Mangu PB and Temin S: American society of clinical oncology/college of american pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Oncol Pract. 6:195–197. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, Dowsett M, Fitzgibbons PL, Hanna WM, Langer A, et al: American society of clinical oncology/college of American pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 25:118–145. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Wolff AC, Hammond ME, Hicks DG, Dowsett M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M, Fitzgibbons P, et al: Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American society of clinical oncology/college of American pathologists clinical practice guideline update. J Clin Oncol. 31:3997–4013. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Amin MB, Edge SB, Greene FL, Byrd DR, Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR, Sullivan DC, et al: AJCC cancer staging manual. 8th edition. Springer; 2017 | |
|
Liu X, Qu JK, Zhang J, Yan Y, Zhao XX, Wang JZ, Qu HY, Liu L, Wang JS and Duan XY: Prognostic role of pretreatment neutrophil to lymphocyte ratio in breast cancer patients: A meta-analysis. Medicine (Baltimore). 96:e81012017. View Article : Google Scholar : PubMed/NCBI | |
|
Ethier JL, Desautels D, Templeton A, Shah PS and Amir E: Prognostic role of neutrophil-to-lymphocyte ratio in breast cancer: A systematic review and meta-analysis. Breast Cancer Res. 19:22017. View Article : Google Scholar : PubMed/NCBI | |
|
Spring LM, Fell G, Arfe A, Sharma C, Greenup R, Reynolds KL, Smith BL, Alexander B, Moy B, Isakoff SJ, et al: Pathologic complete response after neoadjuvant chemotherapy and impact on breast cancer recurrence and survival: A comprehensive meta-analysis. Clin Cancer Res. 26:2838–2848. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou Q, Dong J, Sun Q, Lu N, Pan Y and Han X: Role of neutrophil-to-lymphocyte ratio as a prognostic biomarker in patients with breast cancer receiving neoadjuvant chemotherapy: A meta-analysis. BMJ Open. 11:e0479572021. View Article : Google Scholar : PubMed/NCBI | |
|
Mieog JSD, Van Der Hage JA and Van De Velde CJH: Preoperative chemotherapy for women with operable breast cancer. Cochrane Database Syst Rev. 2007:CD0050022007.PubMed/NCBI | |
|
Denkert C, von Minckwitz G, Darb-Esfahani S, Lederer B, Heppner BI, Weber KE, Budczies J, Huober J, Klauschen F, Furlanetto J, et al: Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 19:40–50. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Schmid P, Cortes J, Dent R, Pusztai L, McArthur H, Kümmel S, Bergh J, Denkert C, Park YH, Hui R, et al: Event-free survival with pembrolizumab in early triple-negative breast cancer. N Engl J Med. 386:556–567. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
von Au A, Shencoru S, Uhlmann L, Mayer L, Michel L, Wallwiener M, Hennigs A, Deutsch T, Riedel F, Heil J, et al: Predictive value of neutrophil-to-lymphocyte-ratio in neoadjuvant-treated patients with breast cancer. Arch Gynecol Obstet. 307:1105–1113. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Savioli F, Morrow ES, Dolan RD, Romics L, Lannigan A, Edwards J and McMillan DC: Prognostic role of preoperative circulating systemic inflammatory response markers in primary breast cancer: Meta-analysis. Br J Surg. 109:1206–1215. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Hanahan D and Weinberg RA: Hallmarks of cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Mantovani A, Romero P, Palucka AK and Marincola FM: Tumour immunity: Effector response to tumour and role of the microenvironment. Lancet. 371:771–783. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Quail DF and Joyce JA: Microenvironmental regulation of tumor progression and metastasis. Nat Med. 19:1423–1437. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
DeNardo DG, Brennan DJ, Rexhepaj E, Ruffell B, Shiao SL, Madden SF, Gallagher WM, Wadhwani N, Keil SD, Junaid SA, et al: Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov. 1:54–67. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Larionova I, Cherdyntseva N, Liu T, Patysheva M, Rakina M and Kzhyshkowska J: Interaction of tumor-associated macrophages and cancer chemotherapy. Oncoimmunology. 8:15960042019. View Article : Google Scholar : PubMed/NCBI | |
|
Ren Z, Yang J, Liang J, Xu Y, Lu G, Han Y, Zhu J, Tan H, Xu T and Ren M: Monitoring of postoperative neutrophil-to-lymphocyte ratio, D-dimer, and CA153 in: Diagnostic value for recurrent and metastatic breast cancer. Front Surg. 9:9274912023. View Article : Google Scholar : PubMed/NCBI | |
|
Lee SK, Choi MY, Bae SY, Lee JH, Lee HC, Kil WH, Lee JE, Kim SW and Nam SJ: Immediate postoperative inflammation is an important prognostic factor in breast cancer. Oncology. 88:337–344. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Moon G, Noh H, Cho IJ, Lee JI and Han A: Prediction of late recurrence in patients with breast cancer: Elevated neutrophil to lymphocyte ratio (NLR) at 5 years after diagnosis and late recurrence. Breast Cancer. 27:54–61. 2020. View Article : Google Scholar : PubMed/NCBI |