CDK4/6 inhibitor resistance mechanisms and treatment strategies (Review)
- Authors:
- Jinyao Huang
- Liang Zheng
- Zicheng Sun
- Jie Li
-
Affiliations: Department of Breast and Thyroid Surgery, The First Affiliated Hospital, Sun Yat‑sen University, Guangzhou, Guangdong 510080, P.R. China, Department of Breast and Thyroid Surgery, Guangzhou Women and Children's Medical Center, Guangzhou, Guangdong 510623, P.R. China - Published online on: August 30, 2022 https://doi.org/10.3892/ijmm.2022.5184
- Article Number: 128
-
Copyright: © Huang et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
This article is mentioned in:
Abstract
 |
 |
 |
 |
 |
|
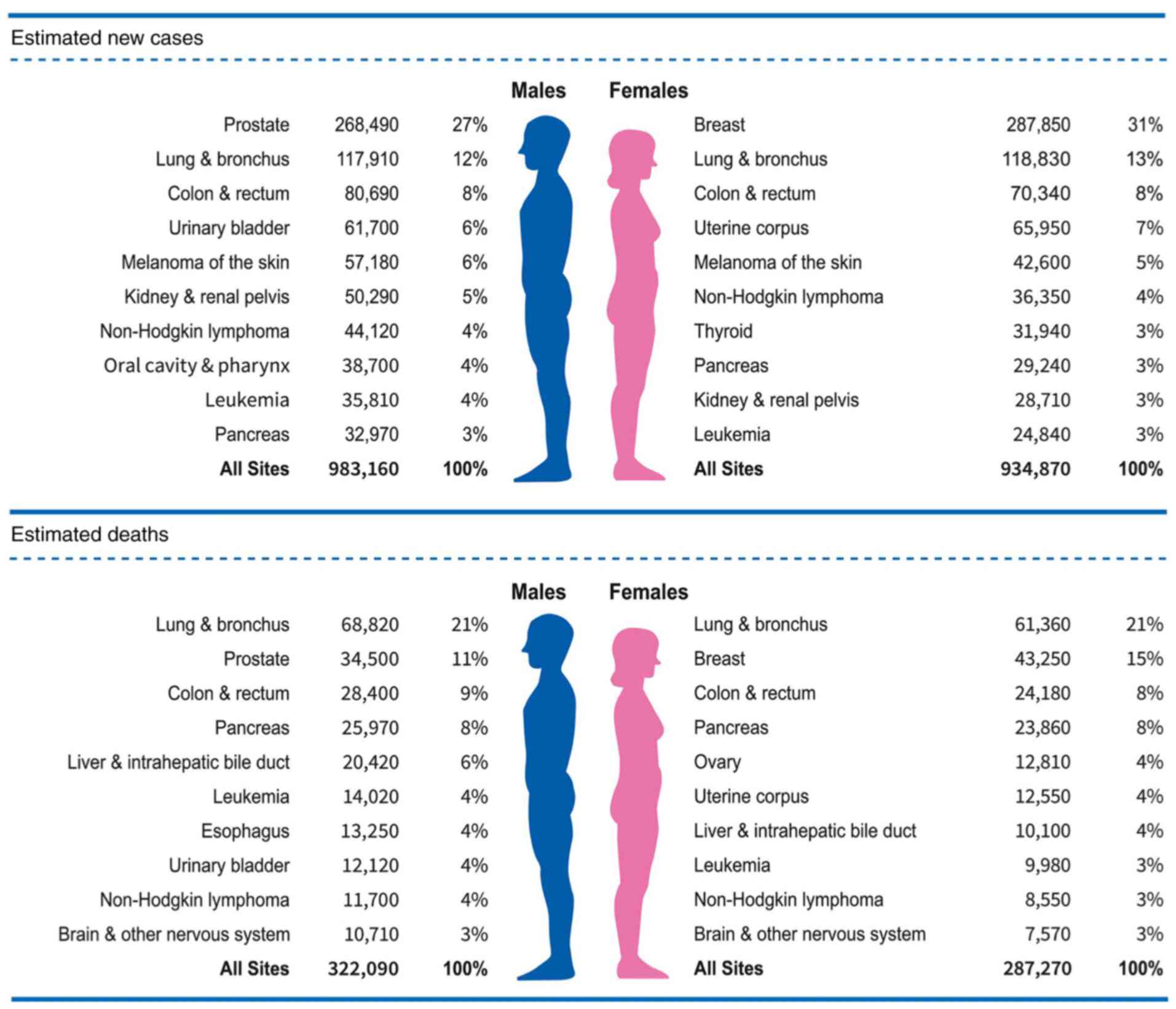
Siegel RL, Miller KD, Fuchs HE and Jemal A: Cancer statistics, 2022. CA Cancer J Clin. 72:7–33. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Dai X, Li T, Bai Z, Yang Y, Liu X, Zhan J and Shi B: Breast cancer intrinsic subtype classification, clinical use and future trends. Am J Cancer Res. 5:2929–2943. 2015.PubMed/NCBI | |
|
Cardoso F, Costa A, Senkus E, Aapro M, André F, Barrios CH, Bergh J, Bhattacharyya G, Biganzoli L, Cardoso MJ, et al: 3rd ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 3). Ann Oncol. 28:31112017. View Article : Google Scholar : PubMed/NCBI | |
|
Milani A, Geuna E, Mittica G and Valabrega G: Overcoming endocrine resistance in metastatic breast cancer: Current evidence and future directions. World J Clin Oncol. 5:990–1001. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Clarke R, Tyson JJ and Dixon JM: Endocrine resistance in breast cancer-an overview and update. Mol Cell Endocrinol. 418:220–234. 2015. View Article : Google Scholar : | |
|
Brufsky AM and Dickler MN: Estrogen receptor-positive breast cancer: Exploiting signaling pathways implicated in endocrine resistance. Oncologist. 23:528–539. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Zhu W and Xu B: Overcoming resistance to endocrine therapy in hormone receptor-positive human epidermal growth factor receptor 2-negative (HR+/HER2−) advanced breast cancer: A meta-analysis and systemic review of randomized clinical trials. Front Med. 15:208–220. 2021. View Article : Google Scholar | |
|
Nair BC and Vadlamudi RK: Regulation of hormonal therapy resistance by cell cycle machinery. Gene Ther Mol Biol. 12:3952008.PubMed/NCBI | |
|
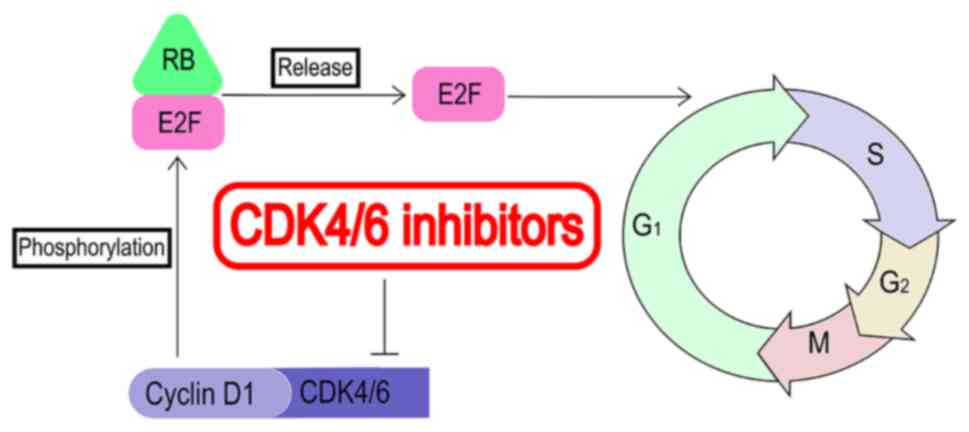
Spring LM, Wander SA, Andre F, Moy B, Turner NC and Bardia A: Cyclin-dependent kinase 4 and 6 inhibitors for hormone receptor-positive breast cancer: Past, present, and future. Lancet. 395:817–827. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Altucci L, Addeo R, Cicatiello L, Germano D, Pacilio C, Battista T, Cancemi M, Petrizzi VB, Bresciani F and Weisz A: Estrogen induces early and timed activation of cyclin-dependent kinases 4, 5, and 6 and increases cyclin messenger ribonucleic acid expression in rat uterus. Endocrinology. 138:978–984. 1997. View Article : Google Scholar : PubMed/NCBI | |
|
Geum D, Sun W, Paik SK, Lee CC and Kim K: Estrogen-induced cyclin D1 and D3 gene expressions during mouse uterine cell proliferation in vivo: Differential induction mechanism of cyclin D1 and D3. Mol Reprod Dev. 46:450–458. 1997. View Article : Google Scholar : PubMed/NCBI | |
|
Finn RS, Crown JP, Lang I, Boer K, Bondarenko IM, Kulyk SO, Ettl J, Patel R, Pinter T, Schmidt M, et al: The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): A randomised phase 2 study. Lancet Oncol. 16:25–35. 2015. View Article : Google Scholar | |
|
Finn RS, Boer K, Bondarenko I, Patel R, Pinter T, Schmidt M, Shparyk YV, Thummala A, Voitko N, Bananis E, et al: Overall survival results from the randomized phase 2 study of palbociclib in combination with letrozole versus letrozole alone for first-line treatment of ER+/HER2- advanced breast cancer (PALOMA-1, TRIO-18). Breast Cancer Res Treat. 183:419–428. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Turner NC, Slamon DJ, Ro J, Bondarenko I, Im SA, Masuda N, Colleoni M, DeMichele A, Loi S, Verma S, et al: Overall survival with palbociclib and fulvestrant in advanced breast cancer. N Engl J Med. 379:1926–1936. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Hortobagyi GN, Stemmer SM, Burris HA, Yap YS, Sonke GS, Paluch-Shimon S, Campone M, Petrakova K, Blackwell KL, Winer EP, et al: Updated results from MONALEESA-2, a phase III trial of first-line ribociclib plus letrozole versus placebo plus letrozole in hormone receptor-positive, HER2-negative advanced breast cancer. Ann Oncol. 30:18422019. View Article : Google Scholar : PubMed/NCBI | |
|
Slamon DJ, Neven P, Chia S, Jerusalem G, De Laurentiis M, Im S, Petrakova K, Valeria Bianchi G, Martín M, Nusch A, et al: Ribociclib plus fulvestrant for postmenopausal women with hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer in the phase III randomized MONALEESA-3 trial: Updated overall survival. Ann Oncol. 32:1015–1024. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Sledge GW Jr, Toi M, Neven P, Sohn J, Inoue K, Pivot X, Burdaeva O, Okera M, Masuda N, Kaufman PA, et al: The effect of abemaciclib plus fulvestrant on overall survival in hormone receptor-positive, ERBB2-negative breast cancer that progressed on endocrine therapy-MONARCH 2: A randomized clinical trial. JAMA Oncol. 6:116–124. 2020. View Article : Google Scholar | |
|
Xu B, Zhang Q, Zhang P, Hu X, Li W, Tong Z, Sun T, Teng Y, Wu X, Ouyang Q, et al: Dalpiciclib versus placebo plus fulvestrant in HR+/HER2- advanced breast cancer that relapsed or progressed on previous endocrine therapy (DAWNA-1): A multi-center, randomized, phase 3 study. J Clin Oncol. 39(15 Suppl): S1002. 2021. View Article : Google Scholar | |
|
Xu B, Zhang Q, Zhang P, Hu X, Li W, Tong Z, Sun T, Teng Y, Wu X, Ouyang Q, et al: Dalpiciclib or placebo plus fulvestrant in hormone receptor-positive and HER2-negative advanced breast cancer: A randomized, phase 3 trial. Nat Med. 27:1904–1909. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang P, Xu B, Gui L, Wang W, Xiu M, Zhang X, Sun G, Zhu X and Zou J: A phase 1 study of dalpiciclib, a cyclin-dependent kinase 4/6 inhibitor in Chinese patients with advanced breast cancer. Biomark Res. 9:242021. View Article : Google Scholar : PubMed/NCBI | |
|
Malumbres M, Sotillo R, Santamaria D, Galán J, Cerezo A, Ortega S, Dubus P and Barbacid M: Mammalian cells cycle without the D-type cyclin-dependent kinases Cdk4 and Cdk6. Cell. 118:493–504. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Guarducci C, Bonechi M, Boccalini G, Benelli M, Risi E, Di Leo A, Malorni L and Migliaccio I: Mechanisms of resistance to CDK4/6 inhibitors in breast cancer and potential biomarkers of response. Breast Care (Basel). 12:304–308. 2017. View Article : Google Scholar | |
|
Siebert R, Willers CP and Opalka B: Role of the cyclin-dependent kinase 4 and 6 inhibitor gene family p15, p16, p18 and p19 in leukemia and lymphoma. Leuk Lymphoma. 23:505–520. 1996. View Article : Google Scholar : PubMed/NCBI | |
|
Green JL, Okerberg ES, Sejd J, Palafox M, Monserrat L, Alemayehu S, Wu J, Sykes M, Aban A, Serra V and Nomanbhoy T: Direct CDKN2 modulation of CDK4 alters target engagement of CDK4 inhibitor drugs. Mol Cancer Ther. 18:771–779. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Finn RS, Crown JP, Ettl J, Schmidt M, Bondarenko IM, Lang I, Pinter T, Boer K, Patel R, Randolph S, et al: Efficacy and safety of palbociclib in combination with letrozole as first-line treatment of ER-positive, HER2-negative, advanced breast cancer: Expanded analyses of subgroups from the randomized pivotal trial PALOMA-1/TRIO-18. Breast Cancer Res. 18:672016. View Article : Google Scholar : PubMed/NCBI | |
|
Turner NC, Liu Y, Zhu Z, Loi S, Colleoni M, Loibl S, DeMichele A, Harbeck N, André F, Bayar MA, et al: Cyclin E1 expression and palbociclib efficacy in previously treated hormone receptor-positive metastatic breast cancer. J Clin Oncol. 37:1169–1178. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Finn RS, Liu Y, Zhu Z, Martin M, Rugo HS, Diéras V, Im SA, Gelmon KA, Harbeck N, Lu DR, et al: Biomarker analyses of response to cyclin-dependent kinase 4/6 inhibition and endocrine therapy in women with treatment-Naïve metastatic breast cancer. Clin Cancer Res. 26:110–121. 2020. View Article : Google Scholar | |
|
Wu A, Wu B, Guo J, Luo W, Wu D, Yang H, Zhen Y, Yu X, Wang H, Zhou Y, et al: Elevated expression of CDK4 in lung cancer. J Transl Med. 9:382011. View Article : Google Scholar : PubMed/NCBI | |
|
Olanich ME, Sun W, Hewitt SM, Abdullaev Z, Pack SD and Barr FG: CDK4 amplification reduces sensitivity to CDK4/6 inhibition in fusion-positive rhabdomyosarcoma. Clin Cancer Res. 21:4947–4959. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Yang C, Li Z, Bhatt T, Dickler M, Giri D, Scaltriti M, Baselga J, Rosen N and Chandarlapaty S: Acquired CDK6 amplification promotes breast cancer resistance to CDK4/6 inhibitors and loss of ER signaling and dependence. Oncogene. 36:2255–2264. 2017. View Article : Google Scholar : | |
|
Tigan AS, Bellutti F, Kollmann K, Tebb G and Sexl V: CDK6-a review of the past and a glimpse into the future: From cell-cycle control to transcriptional regulation. Oncogene. 35:3083–3091. 2016. View Article : Google Scholar | |
|
Kollmann K, Heller G, Schneckenleithner C, Warsch W, Scheicher R, Ott RG, Schäfer M, Fajmann S, Schlederer M, Schiefer AI, et al: A kinase-independent function of CDK6 links the cell cycle to tumor angiogenesis. Cancer Cell. 30:359–360. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Gacche RN and Assaraf YG: Redundant angiogenic signaling and tumor drug resistance. Drug Resist Updat. 36:47–76. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Ji W, Zhang W, Wang X, Shi Y, Yang F, Xie H, Zhou W, Wang S and Guan X: c-myc regulates the sensitivity of breast cancer cells to palbociclib via c-myc/miR-29b-3p/CDK6 axis. Cell Death Dis. 11:7602020. View Article : Google Scholar : PubMed/NCBI | |
|
Etemadmoghadam D, Au-Yeung G, Wall M, Mitchell C, Kansara M, Loehrer E, Batzios C, George J, Ftouni S, Weir BA, et al: Resistance to CDK2 inhibitors is associated with selection of polyploid cells in CCNE1-amplified ovarian cancer. Clin Cancer Res. 19:5960–5971. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Schachter MM, Merrick KA, Larochelle S, Hirschi A, Zhang C, Shokat KM, Rubin SM and Fisher RP: A Cdk7-Cdk4 T-loop phosphorylation cascade promotes G1 progression. Mol Cell. 50:250–260. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Martin LA, Pancholi S, Ribas R, Gao Q, Simigdala N, Nikitorowicz-Buniak J, Johnston SR and Dowsett M: Abstract P3-03-09: Resistance to palbociclib depends on multiple targetable mechanisms highlighting the potential of drug holidays and drug switching to improve therapeutic outcome. Cancer Res. 77(4 Suppl): P3-03-092017. View Article : Google Scholar | |
|
Howell SJ, Krebs MG, Lord S, Kenny L, Bahl A, Clack G, Ainscow E, Arkenau HT, Mansi JL, Palmieri C, et al: 265P Study of samuraciclib (CT7001), a first-in-class, oral, selective inhibitor of CDK7, in combination with fulvestrant in patients with advanced hormone receptor positive HER2 negative breast cancer (HR+BC). Ann Oncol. 32(Suppl 5): S477–S478. 2021. View Article : Google Scholar | |
|
Orlando S, Gallastegui E, Besson A, Abril G, Aligué R, Pujol MJ and Bachs O: p27Kip1 and p21Cip1 collaborate in the regulation of transcription by recruiting cyclin-Cdk complexes on the promoters of target genes. Nucleic Acids Res. 43:6860–6873. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
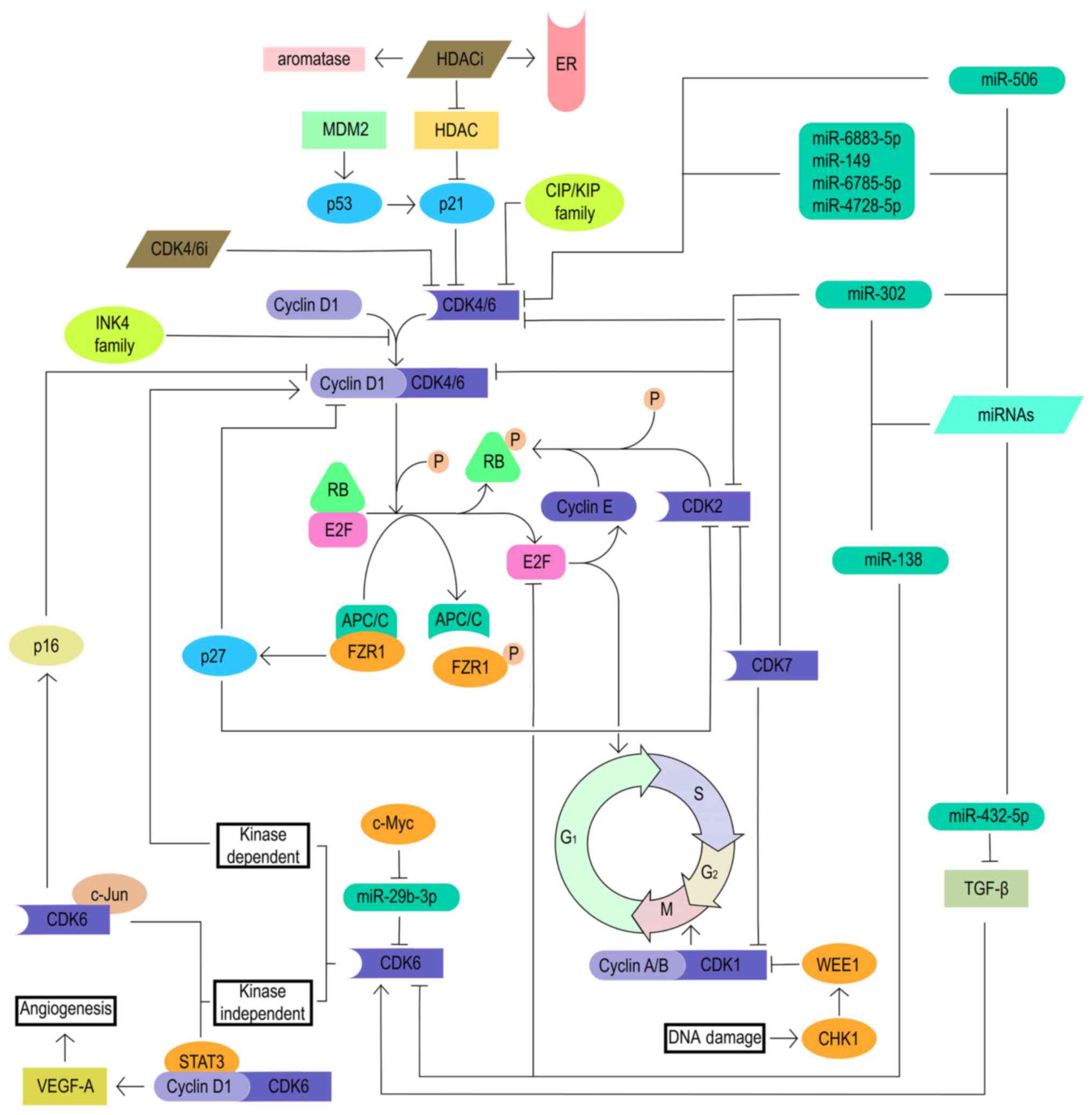
Álvarez-Fernández M and Malumbres M: Mechanisms of sensitivity and resistance to CDK4/6 inhibition. Cancer Cell. 37:514–529. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Sabnis GJ, Goloubeva O, Chumsri S, Nguyen N, Sukumar S and Brodie AM: Functional activation of the estrogen receptor-α and aromatase by the HDAC inhibitor entinostat sensitizes ER-negative tumors to letrozole. Cancer Res. 71:1893–1903. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Yardley DA, Ismail-Khan RR, Melichar B, Lichinitser M, Munster PN, Klein PM, Cruickshank S, Miller KD, Lee MJ and Trepel JB: Randomized phase II, double-blind, placebo-controlled study of exemestane with or without entinostat in postmenopausal women with locally recurrent or metastatic estrogen receptor-positive breast cancer progressing on treatment with a nonsteroidal aromatase inhibitor. J Clin Oncol. 31:2128–2135. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang Z, Li W, Hu X, Zhang Q, Sun T, Cui S, Wang S, Ouyang Q, Yin Y, Geng C, et al: Tucidinostat plus exemestane for postmenopausal patients with advanced, hormone receptor-positive breast cancer (ACE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 20:806–815. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Matheson CJ, Backos DS and Reigan P: Targeting WEE1 kinase in cancer. Trends Pharmacol Sci. 37:872–881. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Pandey K, An HJ, Kim SK, Lee SA, Kim S, Lim SM, Kim GM, Sohn J and Moon YW: Molecular mechanisms of resistance to CDK4/6 inhibitors in breast cancer: A review. Int J Cancer. 145:1179–1188. 2019. View Article : Google Scholar : | |
|
Ramanujan A and Tiwari S: APC/C and retinoblastoma interaction: Cross-talk of retinoblastoma protein with the ubiquitin proteasome pathway. Biosci Rep. 36:e003772016. View Article : Google Scholar : PubMed/NCBI | |
|
Fujita T, Liu W, Doihara H and Wan Y: Regulation of Skp2-p27 axis by the Cdh1/anaphase-promoting complex pathway in colorectal tumorigenesis. Am J Pathol. 173:217–228. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Laroche-Clary A, Chaire V, Algeo MP, Derieppe MA, Loarer FL and Italiano A: Combined targeting of MDM2 and CDK4 is synergistic in dedifferentiated liposarcomas. J Hematol Oncol. 10:1232017. View Article : Google Scholar : PubMed/NCBI | |
|
Cox LS: Multiple pathways control cell growth and transformation: Overlapping and independent activities of p53 and p21Cip1/WAF1/Sdi1. J Pathol. 183:134–140. 1997. View Article : Google Scholar : PubMed/NCBI | |
|
Lin SL, Chang DC, Ying SY, Leu D and Wu DT: MicroRNA miR-302 inhibits the tumorigenecity of human pluripotent stem cells by coordinate suppression of the CDK2 and CDK4/6 cell cycle pathways. Cancer Res. 70:9473–9482. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Qiu S, Huang D, Yin D, Li F, Li X, Kung HF and Peng Y: Suppression of tumorigenicity by microRNA-138 through inhibition of EZH2-CDK4/6-pRb-E2F1 signal loop in glioblastoma multiforme. Biochim Biophys Acta. 1832:1697–1707. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Liu G, Sun Y, Ji P, Li X, Cogdell D, Yang D, Parker Kerrigan BC, Shmulevich I, Chen K, Sood AK, et al: MiR-506 suppresses proliferation and induces senescence by directly targeting the CDK4/6-FOXM1 axis in ovarian cancer. J Pathol. 233:308–318. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Lulla AR, Slifker MJ, Zhou Y, Lev A, Einarson MB, Dicker DT and El-Deiry WS: miR-6883 family miRNAs target CDK4/6 to induce G1 phase cell-cycle arrest in colon cancer cells. Cancer Res. 77:6902–6913. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
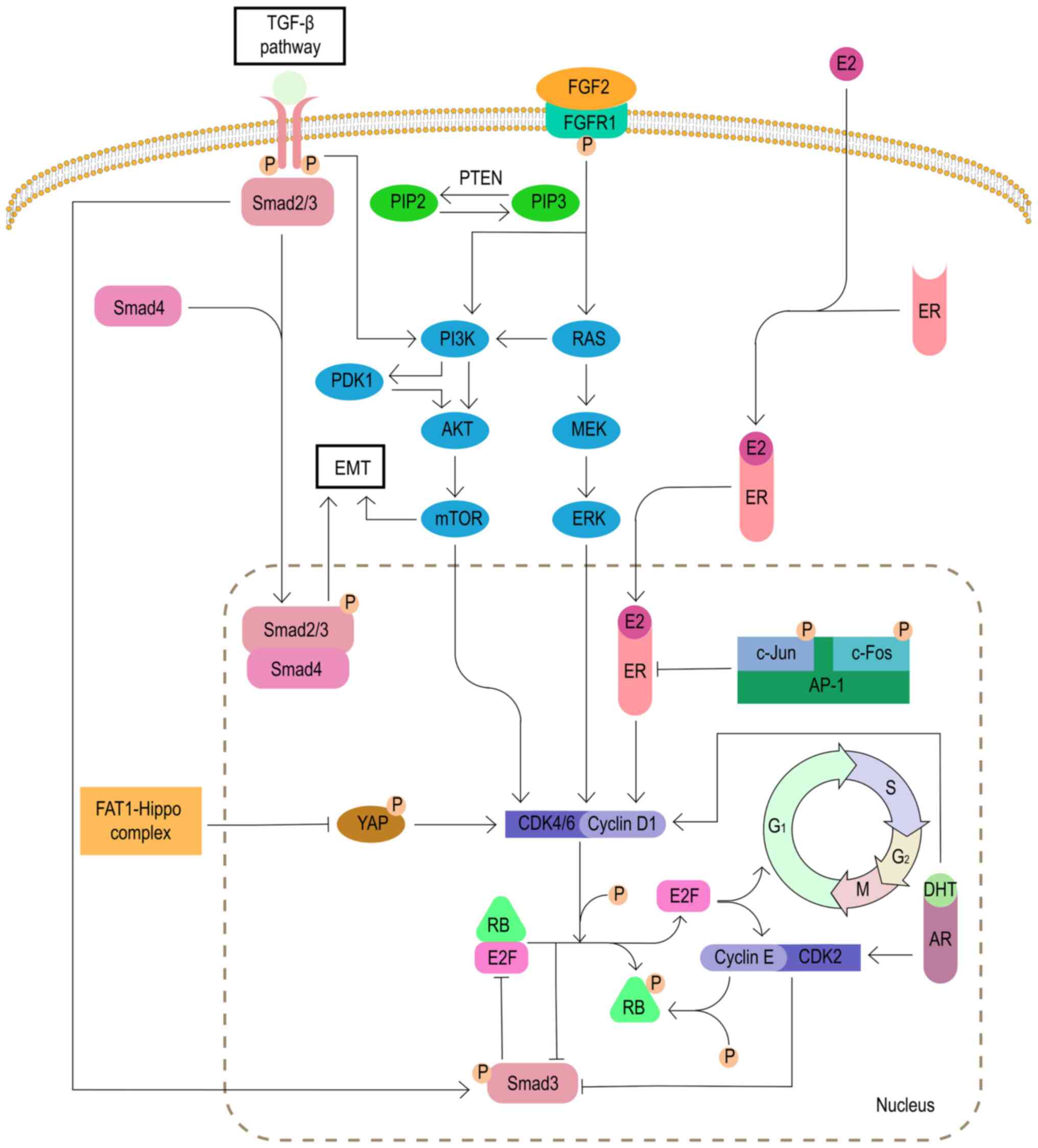
Cornell L, Wander SA, Visal T, Wagle N and Shapiro GI: MicroRNA-mediated suppression of the TGF-β pathway confers transmissible and reversible CDK4/6 inhibitor resistance. Cell Rep. 26:2667–2680.e7. 2019. View Article : Google Scholar | |
|
Turner N and Grose R: Fibroblast growth factor signalling: From development to cancer. Nat Rev Cancer. 10:116–129. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Turner N, Pearson A, Sharpe R, Lambros M, Geyer F, Lopez-Garcia MA, Natrajan R, Marchio C, Iorns E, Mackay A, et al: FGFR1 amplification drives endocrine therapy resistance and is a therapeutic target in breast cancer. Cancer Res. 70:2085–2094. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Formisano L, Lu Y, Servetto A, Hanker AB, Jansen VM, Bauer JA, Sudhan DR, Guerrero-Zotano AL, Croessmann S, Guo Y, et al: Aberrant FGFR signaling mediates resistance to CDK4/6 inhibitors in ER+ breast cancer. Nat Commun. 10:13732019. View Article : Google Scholar : PubMed/NCBI | |
|
Kilker RL and Planas-Silva MD: Cyclin D1 is necessary for tamoxifen-induced cell cycle progression in human breast cancer cells. Cancer Res. 66:11478–11484. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Costa C, Wang Y, Ly A, Hosono Y, Murchie E, Walmsley CS, Huynh T, Healy C, Peterson R, Yanase S, et al: PTEN loss mediates clinical cross-resistance to CDK4/6 and PI3Kα inhibitors in breast cancer. Cancer Discov. 10:72–85. 2020. View Article : Google Scholar | |
|
Bencivenga D, Caldarelli I, Stampone E, Mancini FP, Balestrieri ML, Della Ragione F and Borriello A: p27Kip1 and human cancers: A reappraisal of a still enigmatic protein. Cancer Lett. 403:354–365. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Finn RS, Aleshin A and Slamon DJ: Targeting the cyclin-dependent kinases (CDK) 4/6 in estrogen receptor-positive breast cancers. Breast Cancer Res. 18:172016. View Article : Google Scholar : PubMed/NCBI | |
|
Shaulian E and Karin M: AP-1 in cell proliferation and survival. Oncogene. 20:2390–2400. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Shen Q, Uray IP, Li Y, Zhang Y, Hill J, Xu XC, Young MR, Gunther EJ, Hilsenbeck SG, Colburn NH, et al: Targeting the activator protein 1 transcription factor for the prevention of estrogen receptor-negative mammary tumors. Cancer Prev Res (Phila). 1:45–55. 2008. View Article : Google Scholar | |
|
Tewari D, Nabavi SF, Nabavi SM, Sureda A, Farooqi AA, Atanasov AG, Vacca RA, Sethi G and Bishayee A: Targeting activator protein 1 signaling pathway by bioactive natural agents: Possible therapeutic strategy for cancer prevention and intervention. Pharmacol Res. 128:366–375. 2018. View Article : Google Scholar | |
|
McNamara KM, Yoda T, Takagi K, Miki Y, Suzuki T and Sasano H: Androgen receptor in triple negative breast cancer. J Steroid Biochem Mol Biol. 133:66–76. 2013. View Article : Google Scholar | |
|
Ji W, Shi Y, Wang X, He W, Tang L, Tian S, Jiang H, Shu Y and Guan X: Combined androgen receptor blockade overcomes the resistance of breast cancer cells to palbociclib. Int J Biol Sci. 15:522–532. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Li Z, Razavi P, Li Q, Toy W, Liu B, Ping C, Hsieh W, Sanchez-Vega F, Brown DN, Da Cruz Paula AF, et al: Loss of the FAT1 tumor suppressor promotes resistance to CDK4/6 inhibitors via the Hippo pathway. Cancer Cell. 34:893–905.e8. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Liu F and Korc M: Cdk4/6 inhibition induces epithelial-mesenchymal transition and enhances invasiveness in pancreatic cancer cells. Mol Cancer Ther. 11:2138–2148. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Moustakas A and Heldin CH: Non-Smad TGF-beta signals. J Cell Sci. 118:3573–3584. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Lamouille S, Xu J and Derynck R: Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Zelivianski S, Cooley A, Kall R and Jeruss JS: Cyclin-dependent kinase 4-mediated phosphorylation inhibits Smad3 activity in cyclin D-overexpressing breast cancer cells. Mol Cancer Res. 8:1375–1387. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Yang J, Song K, Krebs TL, Jackson MW and Danielpour D: Rb/E2F4 and Smad2/3 link survivin to TGF-beta-induced apoptosis and tumor progression. Oncogene. 27:5326–5338. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Decker JT, Wan L, Shea LD and Jeruss JS: Abstract P4-03-16: Cyclin E affects Smad3 pathway in trastuzumab resistant HER2+ breast cancer. Cancer Res. 78(4 Suppl): pp. P4-03-162018, View Article : Google Scholar | |
|
Jiang Z, Song E, Wang X, Wang H, Wang X, Wu J, Yin Y, Zhang Q, Chen J, Che W, et al: Guidelines of Chinese society of clinical oncology (CSCO) on diagnosis and treatment of breast cancer (2020 version). 2020. Transl Breast Cancer Res. 1:272020. View Article : Google Scholar | |
|
Johnston SRD, Harbeck N, Hegg R, Toi M, Martin M, Shao ZM, Zhang QY, Martinez Rodriguez JL, Campone M, Hamilton E, et al: Abemaciclib combined with endocrine therapy for the adjuvant treatment of HR+, HER2-, node-positive, high-risk, early breast cancer (monarchE). J Clin Oncol. 38:3987–3998. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Hamilton E, Cortes J, Ozyilkan O, Chen SC, Petrakova K, Manikhas A, Jerusalem G, Hegg R, Huober J, Chapman SC, et al: nextMONARCH: Abemaciclib monotherapy or combined with tamoxifen for metastatic breast cancer. Clin Breast Cancer. 21:181–190.e2. 2021. View Article : Google Scholar | |
|
Wander SA, Zangardi M, Niemierko A, Kambadakone A, Kim LSL, Xi J, Pandey AK, Spring L, Stein C, Juric D, et al: A multicenter analysis of abemaciclib after progression on palbociclib in patients (pts) with hormone receptor-positive (HR+)/HER2-metastatic breast cancer (MBC). J Clin Oncol. 37(15 Suppl): S10572019. View Article : Google Scholar | |
|
Kalinsky K, Accordino MK, Chiuzan C, Mundi PS, Trivedi MS, Novik Y, Tiersten A, Raptis G, Baer LN, Oh SY, et al: A randomized, phase II trial of fulvestrant or exemestane with or without ribociclib after progression on anti-estrogen therapy plus cyclin-dependent kinase 4/6 inhibition (CDK 4/6i) in patients (pts) with unresectable or hormone receptor-positive (HR+), HER2-negative metastatic breast cancer (MBC): MAINTAIN trial. J Clin Oncol. 40(17 Suppl): LBA1004. 2022. View Article : Google Scholar | |
|
Mayer EL, Wander SA, Regan MM, DeMichele A, Forero-Torres A, Rimawi MF, Ma CX, Cristofanilli M, Anders CK, Bartlett CH, et al: Palbociclib after CDK and endocrine therapy (PACE): A randomized phase II study of fulvestrant, palbociclib, and avelumab for endocrine pre-treated ER+/HER2-metastatic breast cancer. J Clin Oncol. 36(15 Suppl): TPS11042018. View Article : Google Scholar | |
|
Bardia A, Aftimos P, Bihani T, Anderson-Villaluz AT, Jung J, Conlan MG and Kaklamani VG: EMERALD: Phase III trial of elacestrant (RAD1901) vs endocrine therapy for previously treated ER+ advanced breast cancer. Future Oncol. 15:3209–3218. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Bidard FC, Kaklamani VG, Neven P, Streich G, Montero AJ, Forget F, Mouret-Reynier MA, Sohn JH, Taylor D, Harnden KK, et al: Elacestrant (oral selective estrogen receptor degrader) versus standard endocrine therapy for estrogen receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: Results from the randomized phase III EMERALD trial. J Clin Oncol. JCO22003382022.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI | |
|
Mosele F, Stefanovska B, Lusque A, Tran Dien A, Garberis I, Droin N, Le Tourneau C, Sablin MP, Lacroix L, Enrico D, et al: Outcome and molecular landscape of patients with PIK3CA-mutated metastatic breast cancer. Ann Oncol. 31:377–386. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
André F, Ciruelos EM, Juric D, Loibl S, Campone M, Mayer IA, Rubovszky G, Yamashita T, Kaufman B, Lu YS, et al: Alpelisib plus fulvestrant for PIK3CA-mutated, hormone receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: Final overall survival results from SOLAR-1. Ann Oncol. 32:208–217. 2021. View Article : Google Scholar | |
|
Rugo HS, Lerebours F, Ciruelos E, Drullinsky P, Borrego MR, Neven P, Park YH, Prat A, Bachelot T, Juric D, et al: Alpelisib (ALP) + fulvestrant (FUL) in patients (pts) with PIK3CA-mutated (mut) hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) advanced breast cancer (ABC) previously treated with cyclin-dependent kinase 4/6 inhibitor (CDKi) + aromatase inhibitor (AI): BYLieve study results. J Clin Oncol. 38(15 Suppl): S10062020. View Article : Google Scholar | |
|
Bartsch R: ASCO 2020: Highlights in breast cancer. Memo. 14:58–61. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
André F, Ciruelos E, Rubovszky G, Campone M, Loibl S, Rugo HS, Iwata H, Conte P, Mayer IA, Kaufman B, et al: Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N Engl J Med. 380:1929–1940. 2019. View Article : Google Scholar | |
|
Suppan C: Post San Antonio update-my top three abstracts! Memo-Mag Eur Med Oncol. 14:244–246. 2021. | |
|
Piccart M, Hortobagyi GN, Campone M, Pritchard KI, Lebrun F, Ito Y, Noguchi S, Perez A, Rugo HS, Deleu I, et al: Everolimus plus exemestane for hormone-receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: Overall survival results from BOLERO-2†. Ann Oncol. 25:2357–2362. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Bardia A, Hurvitz SA, DeMichele A, Clark AS, Zelnak AB, Yardley DA, Karuturi MS, Sanft TB, Blau S, Hart LL, et al: Triplet therapy (continuous ribociclib, everolimus, exemestane) in HR+/HER2-advanced breast cancer postprogression on a CDK4/6 inhibitor (TRINITI-1): Efficacy, safety, and biomarker results. J Clin Oncol. 37(15 Suppl): S10162019. View Article : Google Scholar | |
|
Bardia A, Hurvitz SA, DeMichele A, Clark AS, Zelnak A, Yardley DA, Karuturi M, Sanft T, Blau S, Hart L, et al: Phase I/II trial of exemestane, ribociclib, and everolimus in women with HR+/HER2− advanced breast cancer after progression on CDK4/6 inhibitors (TRINITI-1). Clin Cancer Res. 27:4177–4185. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Wander SA, Juric D, Supko JG, Micalizzi DS, Spring L, Vidula N, Beeler M, Habin KR, Viscosi E, Fitzgerald DM, et al: Phase Ib trial to evaluate safety and anti-tumor activity of the AKT inhibitor, ipatasertib, in combination with endocrine therapy and a CDK4/6 inhibitor for patients with hormone receptor positive (HR+)/HER2 negative metastatic breast cancer (MBC) (TAKTIC). J Clin Oncol. 38(15 Suppl): S10662020. View Article : Google Scholar | |
|
Martin LA and Dowsett M: BCL-2: A new therapeutic target in estrogen receptor-positive breast cancer? Cancer Cell. 24:7–9. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Lok SW, Whittle JR, Vaillant F, The CE, Lo LL, Policheni AN, Bergin ART, Desai J, Ftouni S, Gandolfo LC, et al: A phase Ib dose-escalation and expansion study of the BCL2 inhibitor venetoclax combined with tamoxifen in ER and BCL2-positive metastatic breast cancer. Cancer Discov. 9:354–369. 2019. View Article : Google Scholar | |
|
A phase II study comparing the efficacy of venetoclax + fulvestrant vs fulvestrant in women with estrogen receptor-positive, Her2-negative locally advanced or metastatic breast cancer who experienced disease recurrence or progression during or after CDK4/6 inhibitor therapy (Veronica). | |
|
Lindeman GJ, Bowen R, Jerzak KJ, Song X, Decker T, Boyle FM, McCune SL, Armstrong A, Shannon CM, Bertelli G, et al: Results from VERONICA: A randomized, phase II study of second-/third-line venetoclax (VEN) + fulvestrant (F) versus F alone in estrogen receptor (ER)-positive, HER2-negative, locally advanced, or metastatic breast cancer (LA/MBC). J Clin Oncol. 39(15 Suppl): pp. S10042021, View Article : Google Scholar | |
|
Goel S, DeCristo MJ, Watt AC, BrinJones H, Sceneay J, Li BB, Khan N, Ubellacker JM, Xie S, Metzger-Filho O, et al: CDK4/6 inhibition triggers anti-tumour immunity. Nature. 548:471–475. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
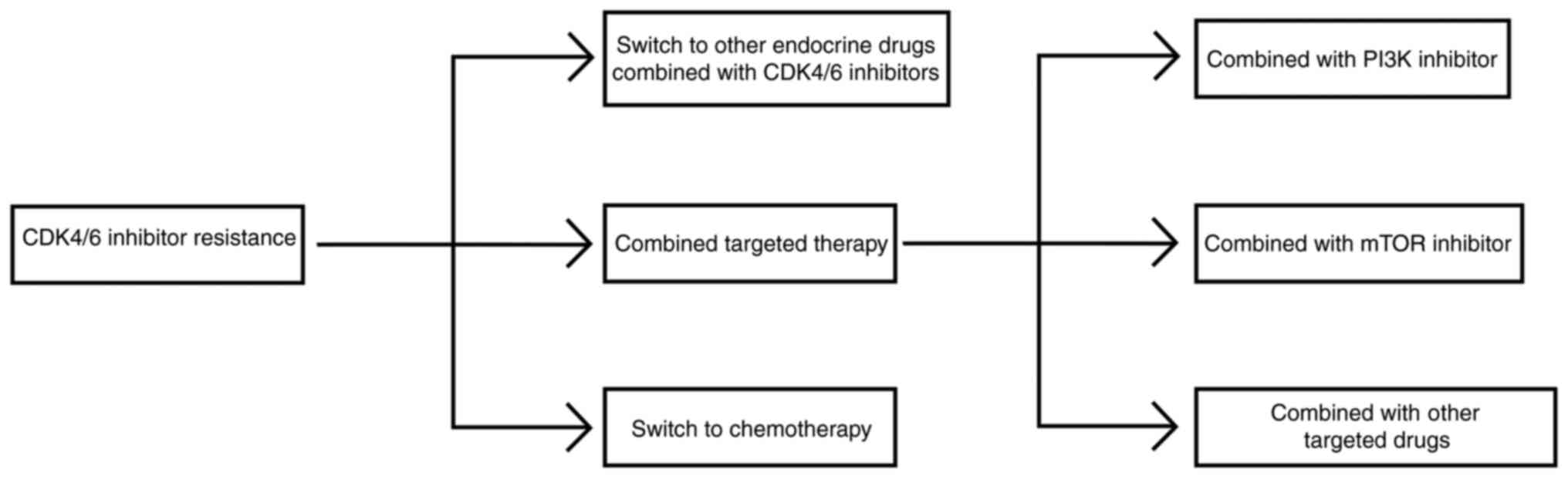
Ogata R, Kishino E, Saitoh W, Koike Y and Kurebayashi J: Resistance to cyclin-dependent kinase (CDK) 4/6 inhibitors confers cross-resistance to other CDK inhibitors but not to chemotherapeutic agents in breast cancer cells. Breast Cancer. 28:206–215. 2021. View Article : Google Scholar | |
|
Rugo HS, Cristofanilli M, Loibl S, Harbeck N, DeMichele A, Iwata H, Park YH, Brufsky A, Theall KP, Huang X, et al: Prognostic factors for overall survival in patients with hormone receptor-positive advanced breast cancer: Analyses from PALOMA-3. Oncologist. 26:e1339–e1346. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Princic N, Aizer A, Tang DH, Smith DM, Johnson W and Bardia A: Predictors of systemic therapy sequences following a CDK 4/6 inhibitor-based regimen in post-menopausal women with hormone receptor positive, HEGFR-2 negative metastatic breast cancer. Curr Med Res Opin. 35:73–80. 2019. View Article : Google Scholar | |
|
Kolyadina IV, Bolotina L, Zhukova L, Vladimirova LU, Sultanbaev A, Karabina E, Ganshina I, Ovchinnikova E, Kolyadina IV, Antonova G, et al: The effectiveness and safety of eribulin therapy in HR-positive HER2-negative metastatic breast cancer post-CDK4/6 inhibitor therapy in Russian clinical practice. J Clin Oncol. 39(15 Suppl): e130352021. View Article : Google Scholar |