Lymphangiogenesis: A new strategy for heart disease treatment (Review)
- Authors:
- Published online on: February 22, 2024 https://doi.org/10.3892/ijmm.2024.5359
- Article Number: 35
-
Copyright: © Bai et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
1. Introduction
A number of diseases such as atherosclerosis (AS), myocardial infarction (MI), heart failure (HF), hypertensive heart disease (HHD) and congenital heart disease (CHD), profoundly impact cardiac health (1). Despite the numerous treatment options available, heart disease remains the leading global cause of mortality, requiring innovative therapeutic approaches. The data show that there were 4.58 million estimated heart disease deaths in 2020; the age-standardized mortality rate (ASMR) was 245.39 per 100,000 in 2020 (2,3). The discovery of lymphatic marker genes, such as acid receptor 1 (LYVE-1) and vascular endothelial growth factor (VEGF) receptor (VEGFR)-3, alongside advancements in lymphatic function imaging and quantification techniques, has brought attention to the role of lymphatic vessels in the progression of heart disease (4). The lymphatic system is an open, low-pressure, one-way transmission network between the extracellular space and the veins that differs from closed, hypertensive circulatory vascular networks. This extensive lymphatic network plays a key role in maintaining tissue fluid homeostasis and monitoring cardiac immunity (5,6). Obstruction of cardiac lymph flow leads to edema and cardiac dysfunction (7), underscoring the clinical significance of elucidating the role of the lymphatic system in cardiovascular diseases. In the field of information science, scientometrics, a highly specialized branch of bibliometrics, offers a quantitative method leveraging mathematical and statistical approaches to visualize emerging research trends and hotspots in scientific literature (8). This method aids in identifying publications, researchers, journals and research institutions that significantly contribute to a field, and enhances our understanding of scientific citations (9). In the present review, scientometrics was used to analyze research hotspots of lymphatic vessels in heart disease and evaluate the impact of the relevant literature. Additionally, a detailed review of molecular mechanisms surrounding these hotspots is presented to advance the development of drugs targeting lymphatic vessels.
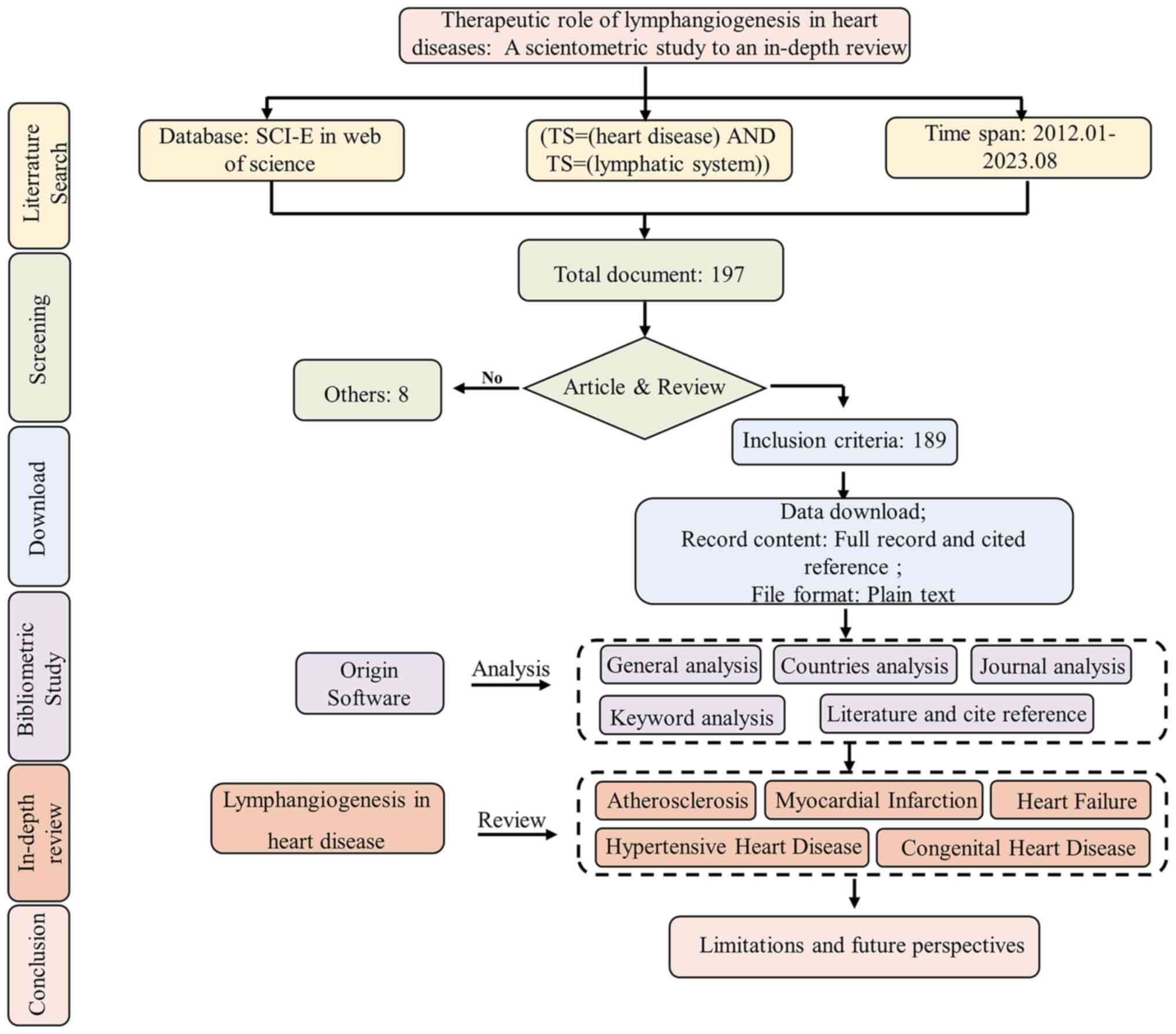
2. Scientometric study
Data and methods
A search in the Web of Science (https://webofscience.clarivate.cn/wos/woscc/basic-search) core collection database for documents published from Jan 1, 2012 to August 1, 2023, was conducted using the key words 'heart disease' and 'lymphatic system'. A total of 197 documents were included, articles and reviews were selected, and other types of documents such as proceeding paper, editorial material, book chapters and others were excluded, and finally a total of 189 documents were used. Subsequently, a scientometric analysis was conducted on both articles and reviews. Raw data, encompassing comprehensive records and cited references, were downloaded from the Web of Science. Using CiteSpace (version 6.1.R2) (10), primary information such as publication numbers per year, countries, journals, key words and literature titles were extracted from the raw data. The data were visualized using Microsoft® Excel® 2021MSO and Hiplot (https://hiplot.com.cn/home/index.html) (Fig. 1).
General analysis
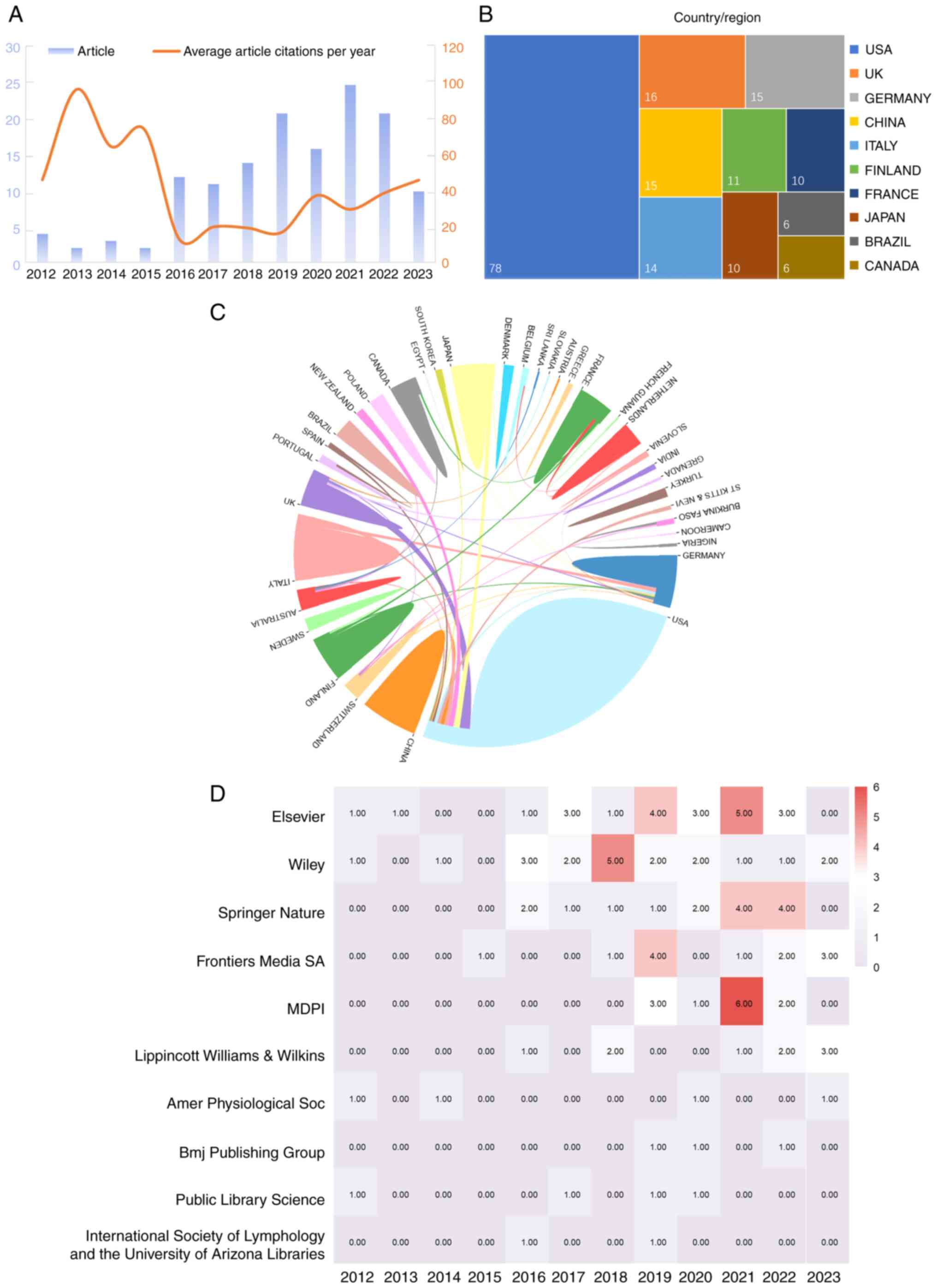
The search in the Web of Science core collection database revealed 189 publications related to the therapeutic effects of the lymphatic system on heart disease before August 1, 2023. On average, these documents were cited 28.06 times. In 2021, the highest number of articles was published, while the maximum number of citations was observed in 2013 (Fig. 2A). The observed trend indicates a growing body of research on the therapeutic effects of the lymphatic system on heart disease.
Country/region analysis
A total of 35 countries were represented in the publications included in the present review. The USA published the highest number of published articles, followed by United Kingdom, Germany, Italy, China, Japan, Finland, France, Brazil and Canada (Fig. 2B). Collaborative efforts were evident among several countries (Fig. 2C).
Publisher analysis
Publications originated from 53 different types of publishers, with the top 10 shown in Fig. 2D. Notably, Elsevier emerged as the most influential publisher, followed by Wiley, Springer Nature, Frontiers Media SA, MDPI, Lippincott Williams & Wilkins, American Physiological Society, BMJ Publishing Group, Public Library Science and International Society of Lymphology and the University of Arizona Libraries.
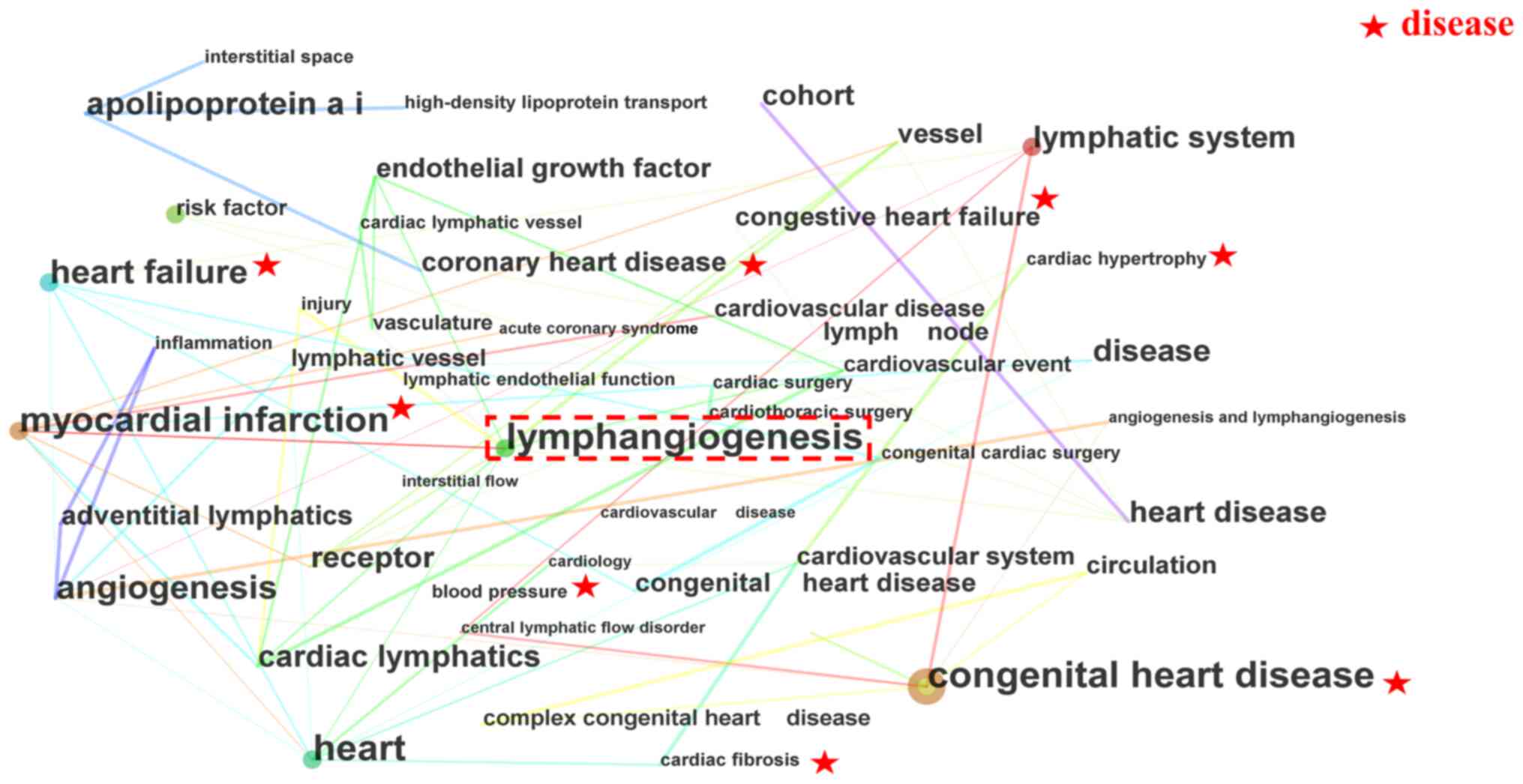
Key word analysis
Research key words reflect current trends, hotspots and the essence of a research document (11). The present review presents a co-occurrence network of key words (Fig. 3; Table I). In these clusters, 'lymphangiogenesis' emerged as a cross-key word, suggesting its potential impact on heart disease. The scientometric results further indicate that the lymphatic vessel is a research hotspot and potentially a promising treatment avenue for heart diseases, including MI, HF and CHD, with a focus on the cardiac lymphatic system.
Literature and cited reference analysis
Citations play a crucial role in acknowledging sources during the writing or editing of a manuscript, including both the cited literature and the specific content of the citation (12). In response to the analysis of the key words, current research interests and topic hotspots were explored through literature analysis and references. Table II presents the top 10 most-cited articles on the therapeutic effect of lymphangiogenesis on heart diseases. The most influential article highlights the critical role of the lymphatic vascular system in maintaining interstitial fluid homeostasis, immune cell transport, nutrient lipid uptake and transport, and reverse cholesterol transportation (13). The second most-cited article underscores that selective stimulation of cardiac lymphangiogenesis post-MI markedly improves myocardial edema and restores cardiac function (14). Additionally, Table III outlines the top 10 cited references on the therapeutic roles of lymphangiogenesis in heart diseases. These articles offer a comprehensive overview of the most influential studies in the field for further research into the therapeutic effect of lymphangiogenesis on heart diseases.
Table IITop 10 most-cited articles about the therapeutic effect of lymphangiogenesis on heart disease. |
Table IIITop 10 cited references about the therapeutic effect of lymphangiogenesis on heart disease. |
The clinical applications of lymphangiogenesis have been used to improve cardiac function (15). However, The clinical trial have yet to elucidate the specific signaling pathways regulating lymphangiogenesis for improved cardiac function. Therefore, animal experiments would be useful in establishing whether and how lymphangiogenesis improves cardiac function. The present review focused extensively on lymphangiogenesis in heart diseases. Furthermore, despite scientometric analysis indicating that lymphangiogenesis is a research hotspot and a promising treatment option for heart disease, a comprehensive and systematic exploration of relevant mechanisms is lacking. Therefore, in the current review, existing research at the mechanistic level on lymphangiogenesis and cardiovascular disease was examined.
3. Lymphangiogenesis and cardiovascular disease
Lymphangiogenesis and AS
AS is a chronic inflammatory disease characterized by the accumulation of cholesterol, lipoproteins and inflammatory cells in the arterial vessel wall (16). Lymphatics are present at atherosclerotic sites in the adventitia of the artery, adjacent to blood vessels and dilate within atherosclerotic plaques. With a deeper understanding of lymphatic vessel function, mounting evidence reveals the critical role of lymphangiogenesis in AS progression. Particularly, it is increasingly acknowledged that lymphatics are the main route for transporting high-density lipoprotein particles in cholesterol to the bloodstream (17). AS promotes lymphangiogenesis (18), which in turn, alleviates AS by facilitating reverse cholesterol transport and simultaneously draining local inflammation (19,20). The number and function of lymphatic vessels are limited. Consequently, promoting lymphangiogenesis holds great promise for the treatment of AS.
According to clinical and experimental studies, lymphangiogenesis in AS is regulated by multiple signaling pathways and molecules. Clinical evidence indicates that VEGF-C/D levels are notably and inversely associated with all-cause mortality among patients with suspected or known AS (21). Early treatment with VEGF-C inhibited plaque formation, promoting lymphatic molecular transport and triggering inflammatory cell migration (22). Besides VEGF-C/D, angiopoietin-2 (Angpt2) is associated with angiogenesis and inflammation. Clinical evidence suggests that the upregulation of Angpt2 may benefit patients with AS (23). This effect may be associated with the increase in VEGF-C/D secretion regulated by Angpt2, promoting lymphangiogenesis around atherosclerotic coronary arteries (23). R-spondin 2 (RSPO2), an important factor for regulating cell proliferation and differentiation, has previously shown potential in promoting lymphangiogenesis (24,25). A study indicated that the interaction between RSPO2 and G protein-coupled receptor 5 inhibits Wnt/β-catenin signaling (26), limiting lymphatic vessel formation, weakening lymphatic drainage of LDL cholesterol in the arterial wall and promoting AS development. A further study found that the phosphatidylinositol-3-hydroxykinase (PI3K)-protein kinase B (AKT) pathway is a downstream pathway of Wnt/β-catenin performs that promotes lymphangiogenesis (24). Additionally, the activation of the C-X-C chemokine receptor type 4 (CXCR4)/chemokine C-X-C motif ligand 12 (CXCL12) axis exacerbates inflammation by promoting leukocyte infiltration (27). A study suggested that the CXCL12/CXCR4 axis regulates cardiac adventitia lymphangiogenesis, reducing inflammation during AS by enhancing T-cell drainage (18). Details are provided in Table IV and Fig. 4.
Lymphangiogenesis and MI
The rupture of an unstable atherosclerotic plaque and subsequent coronary thrombosis often leads to MI (28). MI is often accompanied by myocardial edema, inflammation and fibrosis (29). Cardiac lymphatic flow begins from small endocardium lymphatic vessels, traverses through the myocardium and enters epicardial capillaries. These capillaries converge into large collecting lymphatic vessels, facilitating the transportation of excess water and immune cells, and inhibiting myocardial edema, inflammation and fibrosis (6). Lymphatic vessel formation is investigated as a potential target for MI in clinical practice (30). Lymphatic vessels play a crucial role in heart repair after MI, maintaining tissue fluid homeostasis and suppressing excessive immune responses (31). The VEGF family and their endothelial tyrosine kinase receptors induce lymphangiogenesis (32), but their efficiency is limited when injected intravenously or intracoronarily after MI due to short half-lives (33). Thus, researchers are exploring new pathways new pathways regulating lymphangiogenesis.
VEGF-C-VEGFR3, sphinogosine-1-phosphate (S1P)/S1P receptor 1 (S1PR1) is another noteworthy receptor-ligand complex. S1PR1 is a G-protein-coupled receptor, expressed in lymphatic endothelial cells, and modulates immune cell transportation (34). In injured hearts, S1PR1/S1P activates downstream signals, including extracellular regulated protein kinases/chemokine ligand 2 (ERK), promoting lymphangiogenesis in the injured heart, driving macrophage trafficking, immune modulation, and cardiac repair after MI (35,36). Estrogen receptor α (ERα)/estrogen ligand plays a protective role in MI, associated with regulating lymphangiogenesis (37). Overexpression of cardiomyocyte-specific ERα promotes lymphangiogenesis via inducing c-Jun N-terminal kinase phosphorylation, alleviating ventricular fibrosis (38). Spinster homolog 2 (SPNS2) and thymosin beta 4 (Tβ4) drive lymphangiogenesis by controlling lymphatic endothelial cell proliferation and differentiation. SPNS2, an S1P transporter, contributes to S1P release into extracellular space and the development of lymphatic vessels (39,40). Apelin/Apelin receptor signaling targets SPNS2, promoting lymphatic endothelial cell proliferation and maintaining lymphatic endothelial cell integrity, limiting persistent edema and inflammation after MI (41). Tβ4, which is a 43-amino-acid-sequestering peptide in the embryonic heart, is important for epicardial development and coronary artery formation (42). Tβ4 promotes the differentiation of epicardium-derived cells towards lymphatic endothelial cells, attenuating adverse myocardial remodeling, enhancing myocardial regeneration, and improving cardiac function post-MI (43). Connexin43 (Cx43) may control the function of nascent lymphatic vessels by determining endothelial cell connections. Cx43 modulates gap connection coupling between lymphatic endothelial cells and has been implicated in the pathological process of MI. A previous study showed that adrenomedullin targeted Cx43 to increase gap connections between human lymphatic endothelial cells, promoting the proliferation of the cardiac lymphatic vascular system and relieving MI-induced edema (44). Additionally, VEGF-C-VEGFR-3 indirectly promotes lymphangiogenesis, polarizing macrophage from M1 to M2 in the MI area via activation of downstream AKT/ERK1/2 and calcineurin/nuclear factor of activated T cells 1, forkhead box C2 signaling pathways, accelerating post-MI repair (45). Furthermore, when dead cells are engulfed by macrophages, more VEGF-C is released, further promoting myocardial lymphangiogenesis through its receptor (46). Details are provided in Table IV and Fig. 4.
Lymphangiogenesis and HF
At the end stage of heart disease, the clinical manifestation is HF, not a single pathological diagnosis but a clinical syndrome characterized by breathlessness, ankle swelling, fatigue, pulmonary crackles, peripheral edema and elevated jugular venous pressure (47,48). Common pathological manifestations of HF include myocarditis, myocardial edema, and myocardial fibrosis (49-51). Excessive inflammatory infiltration forms the biological basis for these manifestations (52), and edema results as a major consequence of the inflammatory response in the myocardium (53). Lymphatics are crucial to the progression of HF. Cardiac lymphatics play a crucial role in the progression of HF by adapting their number and function to the increased demand for inflammatory factors and fluid drainage, thus alleviating inflammation and edema (54,55).
Most studies suggest that VEGF-C-VEGFR3 signaling, as the key pathway of lymphangiogenesis, may be a therapeutic target for HF (45). Additionally, interleukin-1β (IL-1β), an inflammatory cytokine associated with MI, has been reported to exert a profound depressant effect on the contractility of lymphatic muscles (56). Prostaglandin E2 (PGE2), the main downstream product of cyclooxygenase-2 activation, induces relaxation by binding to PGE2 to prostaglandin E receptor 2 or 4, elevating intracellular cyclic adenosine monophosphate accumulation (57). A recent study showed that IL-1β reduced the contractility of cardiac lymphatic muscle cells through COX-2/PGE2 signal transduction, weakening the transport of local inflammation in the heart and promoting the progression of acute myocarditis to HF (58). Furthermore, angiotensin-2 (Ang II), is an important factor in inducing inflammation in the vascular system (59). Angiotensin, serotonin, and endothelin are also known to regulate lymphatic vessels (60). Ang II can activate AT1R to promote the expression and activity of proteasome catalytic subunits β2i and β5i, affecting MKP5 and VE-cadherin degradation and p38MAPK activation, resulting in lymphatic endothelial hyperpermeability (61). Moreover, cardiac fibroblasts, principal contributors to cardiac fibrosis leading to cardiac remodeling and HF, express vascular cell adhesion molecule-1 and promote lymphangiogenesis (62). This mobilizes lymphatic endothelial cells into the infarct zone, restoring ventricular wall mechanical properties. The mechanism may involve the formation of lymphatic vessels, inhibition of interstitial edema, and consequently, reduced inflammation and myocardial fibrosis (63). Details are provided in Table IV and Fig. 4.
Lymphangiogenesis and HHD
HHD is an organic heart disease caused by long-term poorly controlled hypertension, characterized by cardiac hypertrophy due to progressively increasing ventricular load (64). Studies indicated that high sodium intake contributes to hypertension development (65). The large accumulation of sodium leads to electrolyte disorders and promotes edema formation (66). Lymphatics is a key route in draining interstitial fluid, and in HHD, lymphangiogenesis is an effective strategy for reducing cardiac interstitial edema. As a cytokine regulating lymphangiogenesis, VEGF-C relieves edema caused by electrolyte imbalances (67). VEGF-C treatment notably improves cardiac function, preserving cardiac function, reducing cardiac hypertrophy and fibrosis, accelerating edema clearance by inducing skin lymphangiogenesis and enhancing endothelial nitric oxide (NO) synthase expression in blood vessels to lower blood pressure (68).
Lymphangiogenesis plays an important role not only in maintaining interstitial fluid balance but also in facilitating immune cell transport in HHD. The activation of the VEGF-C-VEGFR3 pathway is activated in mononuclear phagocytes, triggered by sodium salt accumulation, and stimulates skin lymphangiogenesis (69). Systemic overexpression of VEGF-C enhances lymphangiogenesis within the myocardial interstitium, attenuating local infiltration of macrophages in the heart (70). This dual mechanism of eliminating excess interstitial fluid and transporting immune cells contributes to the reduction of cardiac decompensation-induced myocardial edema and inflammation. In HHD, tonicity enhancer binding protein (TonEBP) and silencing regulatory protein 3 (SIRT3) play key roles in the upstream regulation of VEGFC-VEGFR3 signaling. TonEBP, a key regulator of cellular responses to hypertonic stress, binds to the promoter region of VEGF-C, enhancing its secretion and promoting lymphangiogenesis (71). This, in turn, inhibits left ventricular remodeling induced by high salt intake (72). SIRT3 is a major mitochondrial deacetylase closely associated with angiogenesis. A recent study indicated that SIRT3 activation upregulates the VEGF-C-VEGFR3 signaling axis protein expression, thereby promoting migration and proliferation of lymphatic endothelial cells (LECs), and lymphangiogenesis in AngⅡ-induced hypertensive heart models (73,74). Details are provided in Table IV and Fig. 4.
Lymphangiogenesis in CHD
CHD, a common congenital cardiovascular anomaly (75), significantly affects the lymphatic system due to associated hemodynamic changes (76). The development of lymphatic structures relies on mechanical forces generated by fluid accumulation in the interstitial space. However, chronic elevation of lymph flow can lead to lymphatic system dysfunction under prolonged mechanical stimuli (77). In models of CHD with increased pulmonary blood flow, mechanical stimulation and shear stress upregulate HIF-1 α expression and induce structural disorders in LECs (78). NO plays a pivotal role in lymphangiogenesis and lymphatic function maintenance, with a notable reduction in NO bioavailability observed in congenital heart defect models (79). PPAR-γ, a ligand-activated transcription factor, increases NO bioavailable in LECs (80). Kruppel Factor 2 (KLF2) induces LEC proliferation and lymphatic sprouting under laminar flow conditions (81). The KLF2-mediated PPAR-γ signal, however, inhibits NO production, aggravating lymphatic vessel dysfunction (82). Understanding the role of lymphatic vessels in hemorheology provides innovative treatment strategies for congenital heart defects and other lymphatic disorders. Details are provided in Table IV and Fig. 4.
4. Traditional Chinese Medicine regulating lymphangiogenesis
The rapid advancements in modernization research within Traditional Chinese Medicine (TCM) have garnered international recognition for its efficacy in disease treatment. TCM is characterized by personalized treatment, fewer side effects and a focus on treating both symptoms and root causes and promoting self-healing ability of the body. A study reported the explicitly regulatory effects of TCM on lymphatic vessels, particularly in promoting lymphangiogenesis (83).
Due to its core role in tissue inflammation (20), lymphangiogenesis holds the potential to augment the excretion of extracellular fluid, thereby reducing edema during both acute and chronic inflammatory attacks (84). Han et al (85) and Chen et al (86) identified that Du-huo-ji-sheng-tang and Fang-Ji-Huang-Qi-Tang exhibit the capability to enhance lymphangiogenesis and promote lymphatic drainage function. This enhancement proves pivotal in alleviating local edema and tissue damage caused by inflammation. Additionally, a study demonstrated the efficacy of ginsenoside Rg1, a tetracyclic triterpenoid compound extracted from ginseng, in promoting the recovery of lymphatic drainage function, leading to a reduction in joint swelling (87).
Several studies highlight the efficacy of Chinese herbal extracts and monomers in alleviating lymphedema by promoting lymphangiogenesis. Ishii et al (88) reported that piper retrofractum extract and piperine, isolated from piper retrofractum, notably enhanced the proliferation, migration and tube formation in human dermal lymphatic microvascular endothelial cells, thereby promoting lymphangiogenesis through the AKT and ERK pathways and effectively alleviating lymphedema. T total saponin of Panax notoginseng (PNS) can markedly promote lymphangiogenesis, and its mechanism may be related to the activation of the ERK1/2, PI3K and P38-MAPK signaling pathways, which stimulate the release of VEGF-C. PNS is a promising therapeutic option for managing secondary lymphedema or other lymphatic system disorder (89).
Notably, there is a limited number of TCM studies addressing the regulation of lymphangiogenesis in the treatment of cardiovascular diseases (90). Studies have indicated that salvianolic acid B (SAB), a water-soluble phenolic compound isolated from Danshen, exhibits therapeutic effects on dilated cardiomyopathy, which is associated with lymphangiogenesis deficiency (91). Mechanistically, SAB promotes the proliferation and migration of LECs by upregulating the TNF-α/NF-κB/VEGF-C signaling pathway, thus regulating the formation of lymphatic vessels and inhibiting the development of the pathological process of dilated heart disease (92).
The lymphatic vessel emerges as a key conduit for local inflammation, playing an important role in various diseases, including osteoarthritis, lymphedema and cardiovascular disease (31,93). Notably, a study also highlighted the rich distribution of lymphatic vessels in the cornea (94). Furthermore, the discovery of meningeal lymphatic vessels challenges the prevailing notion of the absence of lymphatic systems in the brain. This discovery opens up new avenues for efficiently eliminating metabolic products produced by brain parenchyma (95), offering potential treatment ideas for Alzheimer's disease, insomnia and meningitis (96-98).
A study indicated a growing interest in the regulation of lymphatic vessels by TCM (99). Although current research primarily focuses on arthritis and lymphedema, there are numerous promising signs that TCM can improve microcirculation in the eyes and brain, while also regulating local oxidative stress and immune responses (100,101). These findings prompt us to consider the potential role of TCM in regulating lymphatic vessels, providing a promising perspective for further exploration.
5. Lymphatic dysfunction as the biological basis of phlegm/phlegm dampness in TCM
According to TCM theory, all normal bodily fluids, including inter-tissue fluids and fluids within internal organs fluids such as gastric and intestinal fluids, are categorized as body fluids (102). The generation, distribution and excretion of body fluids are intricately balanced in a healthy body. An imbalance between body fluid generation and excretion can lead to insufficient body fluid and symptoms such as thirst and dry skin (103). TCM syndromes, including phlegm dampness or phlegm retention, stem from abnormal fluid distribution and slow flow within the body (104).
Understanding the biological basis of TCM syndrome has long presented a scientific challenge. Some authors propose that the biological basis of phlegm retention or phlegm dampness lies in the accumulation of tissue fluid (105), hyperlipidemia (106), aggregation of adhesion factors (107) and localized immune inflammation (108). Modern medicine recognizes lymphatic functions that involve the clearance of low-density lipoprotein in macrophages, transportation of immune cells and drainage of tissue fluid (109). Phlegm and dampness in TCM can also be understood as local inflammation, lipid accumulation and fluid retention resulting from the obstruction of lymphatic reflux (110). Through extensive study of phlegm and dampness, it is hypothesized that obstruction of lymphatic reflux might offer a more appropriate explanation.
6. Conclusion and future perspectives
Scientometrics is a branch of information science that uses quantitative analysis and statistics to study scholarly literature, including publications, citations and author collaboration. Researchers can use it to identify research trends in a specific area, assess the impact of related research, understand research progress and identify partners. In the present review, scientometric analysis was applied to identify new research hotspots in cardiovascular-related diseases, specifically analyzing the therapeutic role of lymphangiogenesis. The lymphatic system, as another important circulatory pathway alongside blood circulation, facilitates the circulation of body fluids together with the blood circulatory system. It achieves this by transporting lymphatic fluid containing water and macromolecules, contributing to overall body metabolism, while maintaining the stability of internal lymphangiogenesis (111). Currently, it is well established that the VEGF family is one of the potent lymphangiogenic factors (112). Additional factors, including IL-6 (113), platelet-derived growth factor (114), neuropilin-2 (115) and calcium-binding EGF domain-containing protein 1 (116) have also been identified for their role in stimulating lymphangiogenesis. Various indirect factors, such as activated macrophages secreting lymphatic vessel-promoting factors, also influence lymphangiogenesis (117). Considerable attention is currently directed towards the VEGF-C/D-VEGFR3 pathway as a potential therapeutic target. In relevant experiments, recombinant VEGF-C-C156S has demonstrated the ability to promote lymphangiogenesis and improve prognosis after MI. Furthermore, certain proteins, such as Polydom (118) and Notch (119) play an important role in lymphangiogenesis. Therefore, gaining a better understanding of the cellular and molecular mechanisms underlying specific lymphatic vessel diseases, coupled with the development of new drugs, holds the potential to limit the development and progression of heart diseases.
The therapeutic strategy aimed at specifically targeting lymphangiogenesis in treating heart disease has been thoroughly validated in animal models. This includes significant improvements in lipid transport, cardiac function, and immune response observed after heart transplantation through the administration of VEGF-C (120-122). However, clinical studies on the treatment of heart disease with lymphatic neovascularization, particularly involving VEGF, remain limited. An injection of VEGF-2 into the endocardium has shown notable improvement in angina pectoris symptoms and was proven to be relatively safe. Conducting a larger phase III trial is deemed necessary for further validation (123). AdVEGF-DΔNΔC gene therapy is safe, well tolerated, and notably improves myocardial perfusion reserve, enhancing the quality of life for patients with MI during year 1 of follow-up (124). Moreover, researchers are actively exploring changes in drug dosage forms to target lesion sites and enhance drug efficacy. This includes innovative approaches such as using nano and hyaluronic acid hydrogel-wrapped VEGF to achieve site-specific delivery. This method of drug delivery not only avoids the short half-life associated with intravenous VEGF injection, but also alleviates the disadvantages of easy degradation in vivo, making it suitable for clinical lymphangiogenesis treatment (125,126). In conclusion, the exploration of lymphatic neovascularization factors and related pathways, along with the development of corresponding therapeutic drugs and dosage forms, represents a promising therapeutic strategy for heart disease treatment.
TCM, as a natural medicine, holds a marked advantage in regulating lymphangiogenesis. Current research indicates that TCM exerts remarkable anti-inflammatory effects by regulating lymphatic neovascularization (99). A major effect of TCM lies in its ability to regulate lymphatic vessel function, including lipid transport, tissue fluid homeostasis and immunity (127). The structure of lymphatic vessels plays a crucial role in their function. The mature lymphatic network comprises initial lymphatics, precollectors and collecting ducts (128). Due to structural differences, these vessels serve distinct functions. Blind-ended initial lymphatics take up lymphatic fluid containing fluid, macromolecules and immune cells (129). Precollectors and collecting ducts possess valvular structures that effectively prevent lymph fluid reflux (130). At present, TCM research on lymphatic vessels mainly focuses on their quantity and function, with limited attention to their structures and mechanisms (99). Consequently, future research should focus on further exploring the structure of lymphatic vessels to enhance our understanding of the way TCM operates. Although lymphangiogenesis is gradually recognized as a novel target for intervention in cardiovascular diseases, relatively few studies have been reported on the TCM for the treatment of cardiovascular diseases focusing on lymphangiogenesis (90) By contrast, TCM has been reported to treat arthritis and lymphedema by promoting lymphangiogenesis (131,132). Since lymphangiogenesis helps to impede the pathological processes of cardiovascular diseases, including fibrosis, edema and plaque formation by reversing cholesterol transport, draining local inflammation and eliminating excessive interstitial fluid, the role of TCM in the regulation of lymphangiogenesis will be a promising research direction. While the majority of drugs are absorbed into the bloodstream after further breakdown in the intestines, lymphatic vessels are widely distributed throughout the intestinal tract, ultimately directing lymph into the veins. It would be of interest to investigate the presence of small molecules of TCM in lymphatic fluid, enabling direct administration of drugs through lymphatic vessels. This approach could notably reduce the first-pass effect of drugs, thereby enhancing their efficacy.
Availability of data and materials
Not applicable.
Authors' contributions
BL carried out investigation and visualization, and wrote the original draft of the manuscript. WY conceptualized the study, acquired funding, developed methodology and wrote the original draft of the manuscript. DS carried out investigation and data curation. SY carried out investigation and used software. CL acquired funding, reviewed the manuscript and completed language editing. LL developed methodology, supervised the project, and reviewed and edited the manuscript. LY conceptualized the study, supervised the project, and reviewed and edited the manuscript. All authors have read and approved the final version of the manuscript. Data authentication not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Abbreviations:
|
AS |
atherosclerosis |
|
Angpt2 |
angiopoietin-2 |
|
AKT |
protein kinase B |
|
CXCL12 |
chemokine C-X-C motif ligand 12 |
|
CXCR4 |
C-X-C chemokine receptor type 4 |
|
CHD |
congenital heart disease |
|
Cx43 |
connexin43 |
|
ERK |
extracellular regulated protein kinase |
|
ERα |
estrogen receptor α |
|
HHD |
hypertensive heart disease |
|
HF |
heart failure |
|
IL-1β |
interleukin-1β |
|
KLF2 |
Kruppel factor 2 |
|
LEC |
lymphatic endothelial cell |
|
MI |
myocardial infarction |
|
NO |
nitric oxide |
|
PGE2 |
prostaglandin E2 |
|
PI3K |
phosphatidylinositol-3-hydroxykinase |
|
RSPO2 |
R-spondin 2 |
|
S1P |
sphinogosine-1-phosphate |
|
SPNS2 |
spinster homolog 2 |
|
S1PR1 |
sphinogosine-1-phosphate receptor 1 |
|
SIRT3 |
silencing regulatory protein 3 |
|
Tβ4 |
thymosin β 4 |
|
TCM |
Traditional Chinese medicine |
|
TonEBP |
tonicity enhancer binding protein |
|
VEGF-C/D |
vascular endothelial growth factor-C/D |
Acknowledgments
The authors would like to thank Shanghai Tengyun Biotechnology Co., Ltd. for providing technical assistance and valuable tools for data analysis and visualization using the Hiplot Pro platform (https://hiplot.com.cn/).
Funding
The present study was supported by the National Natural Science Foundation of China (grant nos. 82204867 and 82174205) and Tianjin Municipal Health Commission (grant no. 2023145).