MicroRNA‑mediated regulation of muscular atrophy: Exploring molecular pathways and therapeutics (Review)
- Authors:
- Woohyeong Jung
- Uijin Juang
- Suhwan Gwon
- Hounggiang Nguyen
- Qingzhi Huang
- Soohyeon Lee
- Beomwoo Lee
- So-Hee Kwon
- Seon-Hwan Kim
- Jongsun Park
-
Affiliations: Department of Pharmacology, College of Medicine, Chungnam National University, Daejeon 35015, Republic of Korea, College of Pharmacy, Yonsei Institute of Pharmaceutical Sciences, Yonsei University, Incheon 21983, Republic of Korea, Department of Neurosurgery, Institute for Cancer Research, College of Medicine, Chungnam National University, Daejeon 35015, Republic of Korea - Published online on: April 9, 2024 https://doi.org/10.3892/mmr.2024.13222
- Article Number: 98
-
Copyright: © Jung et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
This article is mentioned in:
Abstract
 |
 |
 |
 |
 |
 |
|
Duan K, Gao X and Zhu D: The clinical relevance and mechanism of skeletal muscle wasting. Clin Nutr. 40:27–37. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Larsson L, Degens H, Li M, Salviati L, Lee YI, Thompson W, Kirkland JL and Sandri M: Sarcopenia: Aging-Related loss of muscle mass and function. Physiol Rev. 99:427–511. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Damluji AA, Alfaraidhy M, AlHajri N, Rohant NN, Kumar M, Al Malouf C, Bahrainy S, Ji Kwak M, Batchelor WB, Forman DE, et al: Sarcopenia and cardiovascular diseases. Circulation. 147:1534–1553. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Nishio H, Niba ETE, Saito T, Okamoto K, Takeshima Y and Awano H: Spinal muscular atrophy: The past, present, and future of diagnosis and treatment. Int J Mol Sci. 24:119392023. View Article : Google Scholar : PubMed/NCBI | |
|
O'Brien J, Hayder H, Zayed Y and Peng C: Overview of MicroRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol (Lausanne). 9:4022018. View Article : Google Scholar : PubMed/NCBI | |
|
Gebert LFR and MacRae IJ: Regulation of microRNA function in animals. Nat Rev Mol Cell Biol. 20:21–37. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Brzeszczynska J, Brzeszczynski F, Hamilton DF, McGregor R and Simpson AHRW: Role of microRNA in muscle regeneration and diseases related to muscle dysfunction in atrophy, cachexia, osteoporosis, and osteoarthritis. Bone Joint Res. 9:798–807. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
De Paepe B: Progressive skeletal muscle atrophy in muscular dystrophies: A role for toll-like receptor-signaling in disease pathogenesis. Int J Mol Sci. 21:44402020. View Article : Google Scholar : PubMed/NCBI | |
|
Vo TT, Kong G, Kim C, Juang U, Gwon S, Jung W, Nguyen H, Kim SH and Park J: Exploring scavenger receptor class F member 2 and the importance of scavenger receptor family in prediagnostic diseases. Toxicol Res. 39:341–353. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Jun L, Robinson M, Geetha T, Broderick TL and Babu JR: Prevalence and mechanisms of skeletal muscle atrophy in metabolic conditions. Int J Mol Sci. 24:29732023. View Article : Google Scholar : PubMed/NCBI | |
|
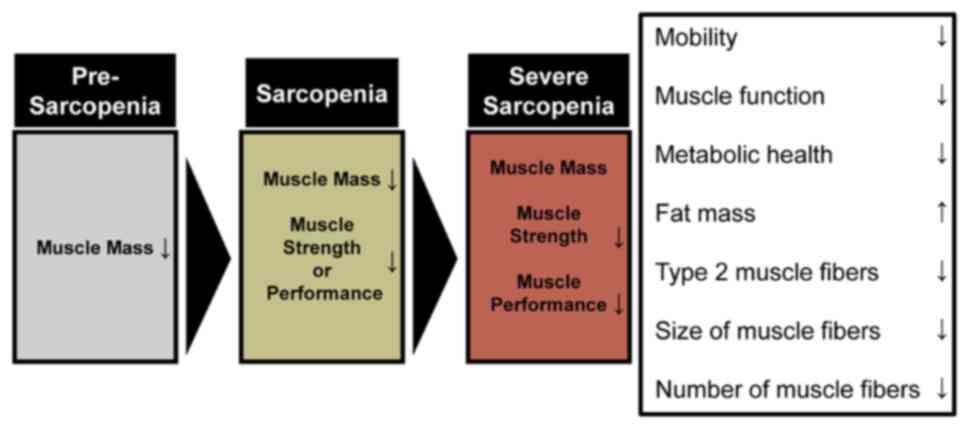
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, et al: Sarcopenia: European consensus on definition and diagnosis: Report of the European working group on sarcopenia in older people. Age Ageing. 39:412–423. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Cho MR, Lee S and Song SK: A review of sarcopenia pathophysiology, diagnosis, treatment and future direction. J Korean Med Sci. 37:e1462022. View Article : Google Scholar : PubMed/NCBI | |
|
Jang JY, Kim D and Kim ND: Pathogenesis, intervention, and current status of drug development for sarcopenia: A review. Biomedicines. 11:16352023. View Article : Google Scholar : PubMed/NCBI | |
|
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyere O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, et al: Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing. 48:16–31. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Guttikonda D and Smith AL: Sarcopenia assessment techniques. Clin Liver Dis (Hoboken). 18:189–192. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Koo BK: Assessment of muscle quantity, quality and function. J Obes Metab Syndr. 31:9–16. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Cheng KY, Chow SK, Hung VW, Wong CH, Wong RM, Tsang CS, Kwok T and Cheung WH: Diagnosis of sarcopenia by evaluating skeletal muscle mass by adjusted bioimpedance analysis validated with dual-energy X-ray absorptiometry. J Cachexia Sarcopenia Muscle. 12:2163–2173. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Faron A, Sprinkart AM, Kuetting DLR, Feisst A, Isaak A, Endler C, Chang J, Nowak S, Block W, Thomas D, et al: Body composition analysis using CT and MRI: intra-individual intermodal comparison of muscle mass and myosteatosis. Sci Rep. 10:117652020. View Article : Google Scholar : PubMed/NCBI | |
|
Dufour AB, Hannan MT, Murabito JM, Kiel DP and McLean RR: Sarcopenia definitions considering body size and fat mass are associated with mobility limitations: The Framingham Study. J Gerontol A Biol Sci Med Sci. 68:168–174. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Singh H, Kim D, Kim E, Bemben MG, Anderson M, Seo DI and Bemben DA: Jump test performance and sarcopenia status in men and women, 55 to 75 years of age. J Geriatr Phys Ther. 37:76–82. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Lee J, Hong YP, Shin HJ and Lee W: Associations of sarcopenia and sarcopenic obesity with metabolic syndrome considering both muscle mass and muscle strength. J Prev Med Public Health. 49:35–44. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Hunter GR, McCarthy JP and Bamman MM: Effects of resistance training on older adults. Sports Med. 34:329–348. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Hepple RT: Sarcopenia-a critical perspective. Sci Aging Knowledge Environ. 2003:pe312003. View Article : Google Scholar : PubMed/NCBI | |
|
Hunter GR, Singh H, Carter SJ, Bryan DR and Fisher G: Sarcopenia and its implications for metabolic health. J Obes. 2019:80317052019. View Article : Google Scholar : PubMed/NCBI | |
|
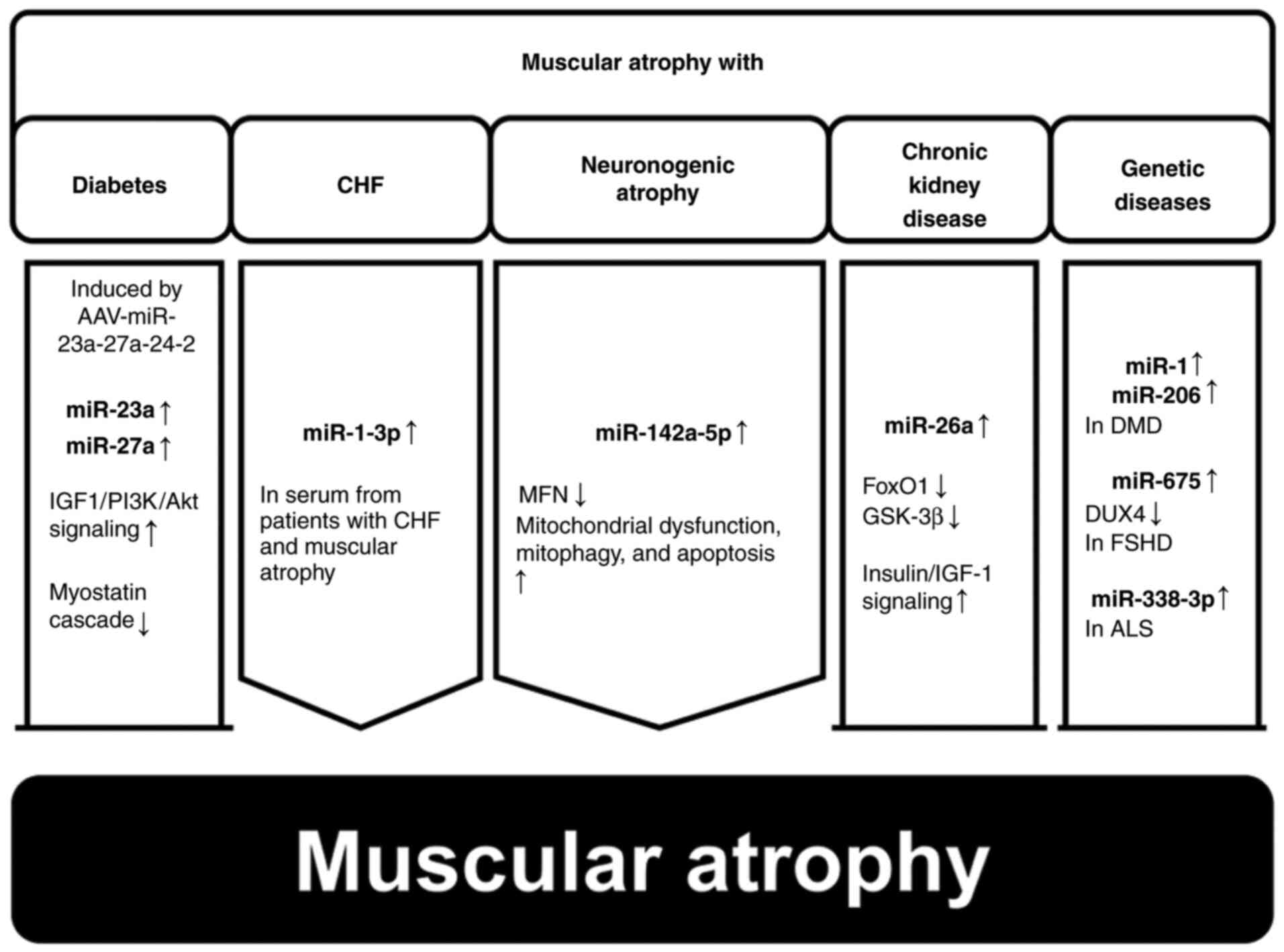
Zhang A, Li M, Wang B, Klein JD, Price SR and Wang XH: miRNA-23a/27a attenuates muscle atrophy and renal fibrosis through muscle-kidney crosstalk. J Cachexia Sarcopenia Muscle. 9:755–770. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Xu R, Cui S, Chen L, Chen XC, Ma LL, Yang HN and Wen FM: Circulating miRNA-1-3p as biomarker of accelerated sarcopenia in patients diagnosed with chronic heart failure. Rev Invest Clin. 74:276–268. 2022.PubMed/NCBI | |
|
Yang X, Xue P, Chen H, Yuan M, Kang Y, Duscher D, Machens HG and Chen Z: Denervation drives skeletal muscle atrophy and induces mitochondrial dysfunction, mitophagy and apoptosis via miR-142a-5p/MFN1 axis. Theranostics. 10:1415–1432. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Wang B, Zhang A, Wang H, Klein JD, Tan L, Wang ZM, Du J, Naqvi N, Liu BC and Wang XH: miR-26a limits muscle wasting and cardiac fibrosis through exosome-mediated microRNA transfer in chronic kidney disease. Theranostics. 9:1864–1877. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Oikawa S, Yuan S, Kato Y and Akimoto T: Skeletal muscle-enriched miRNAs are highly unstable in vivo and may be regulated in a Dicer-independent manner. FEBS J. 290:5692–5703. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Meng Q, Zhang J, Zhong J, Zeng D and Lan D: Novel miRNA biomarkers for patients with duchenne muscular dystrophy. Front Neurol. 13:9217852022. View Article : Google Scholar : PubMed/NCBI | |
|
Saad NY, Al-Kharsan M, Garwick-Coppens SE, Chermahini GA, Harper MA, Palo A, Boudreau RL and Harper SQ: Human miRNA miR-675 inhibits DUX4 expression and may be exploited as a potential treatment for Facioscapulohumeral muscular dystrophy. Nat Commun. 12:71282021. View Article : Google Scholar : PubMed/NCBI | |
|
De Felice B, Annunziata A, Fiorentino G, Borra M, Biffali E, Coppola C, Cotrufo R, Brettschneider J, Giordana ML, Dalmay T, et al: miR-338-3p is over-expressed in blood, CFS, serum and spinal cord from sporadic amyotrophic lateral sclerosis patients. Neurogenetics. 15:243–253. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Dumont NA, Wang YX and Rudnicki MA: Intrinsic and extrinsic mechanisms regulating satellite cell function. Development. 142:1572–1581. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Feige P, Brun CE, Ritso M and Rudnicki MA: Orienting muscle stem cells for regeneration in homeostasis, aging, and disease. Cell Stem Cell. 23:653–664. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Yafe A, Shklover J, Weisman-Shomer P, Bengal E and Fry M: Differential binding of quadruplex structures of muscle-specific genes regulatory sequences by MyoD, MRF4 and myogenin. Nucleic Acids Res. 36:3916–3925. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Gunther S, Kim J, Kostin S, Lepper C, Fan CM and Braun T: Myf5-positive satellite cells contribute to Pax7-dependent long-term maintenance of adult muscle stem cells. Cell Stem Cell. 13:590–601. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
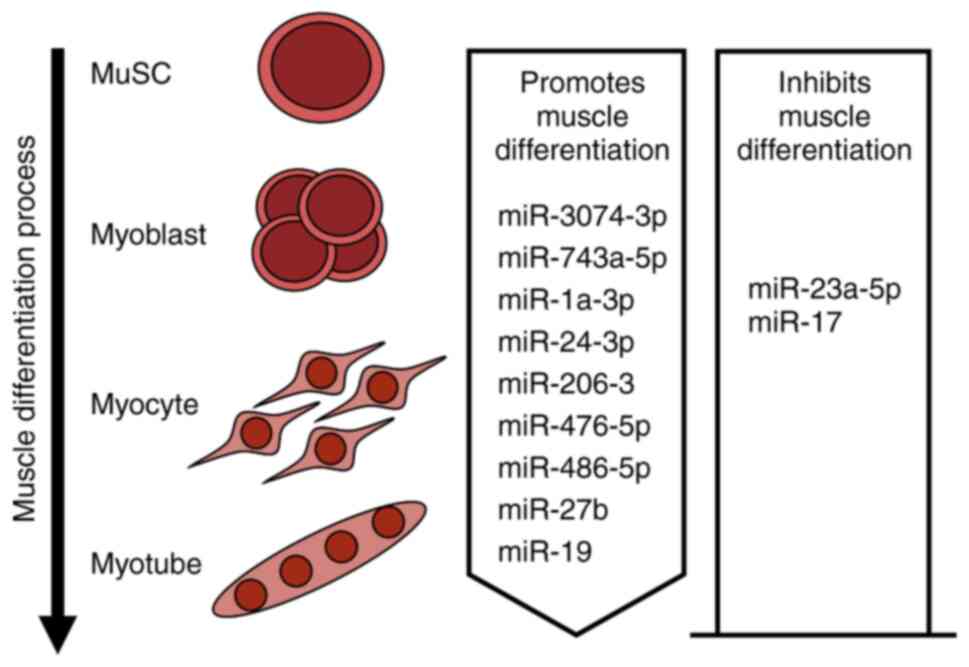
Chen JF, Mandel EM, Thomson JM, Wu Q, Callis TE, Hammond SM, Conlon FL and Wang DZ: The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat Genet. 38:228–233. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Lee SW, Yang J, Kim SY, Jeong HK, Lee J, Kim WJ, Lee EJ and Kim HS: MicroRNA-26a induced by hypoxia targets HDAC6 in myogenic differentiation of embryonic stem cells. Nucleic Acids Res. 43:2057–2073. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Wu R, Li H, Zhai L, Zou X, Meng J, Zhong R, Li C, Wang H, Zhang Y and Zhu D: MicroRNA-431 accelerates muscle regeneration and ameliorates muscular dystrophy by targeting Pax7 in mice. Nat Commun. 6:77132015. View Article : Google Scholar : PubMed/NCBI | |
|
Ma G, Wang Y, Li Y, Cui L, Zhao Y, Zhao B and Li K: MiR-206, a key modulator of skeletal muscle development and disease. Int J Biol Sci. 11:345–352. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Dey P, Soyer MA and Dey BK: MicroRNA-24-3p promotes skeletal muscle differentiation and regeneration by regulating HMGA1. Cell Mol Life Sci. 79:1702022. View Article : Google Scholar : PubMed/NCBI | |
|
Lee B, Shin YJ, Lee SM, Son YH, Yang YR and Lee KP: miR-3074-3p promotes myoblast differentiation by targeting Cav1. BMB Rep. 53:278–283. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang Y, Yao Y, Wang Z, Lu D, Zhang Y, Adetula AA, Liu S, Zhu M, Yang Y, Fan X, et al: MiR-743a-5p regulates differentiation of myoblast by targeting Mob1b in skeletal muscle development and regeneration. Genes Dis. 9:1038–1048. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Holstein I, Singh AK, Pohl F, Misiak D, Braun J, Leitner L, Huttelmaier S and Posern G: Post-transcriptional regulation of MRTF-A by miRNAs during myogenic differentiation of myoblasts. Nucleic Acids Res. 48:8927–8942. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhao X, Gu H, Wang L, Zhang P, Du J, Shen L, Jiang D, Wang J, Li X, Zhang S, et al: MicroRNA-23a-5p mediates the proliferation and differentiation of C2C12 myoblasts. Mol Med Rep. 22:3705–3714. 2020.PubMed/NCBI | |
|
Crist CG, Montarras D, Pallafacchina G, Rocancourt D, Cumano A, Conway SJ and Buckingham M: Muscle stem cell behavior is modified by microRNA-27 regulation of Pax3 expression. Proc Natl Acad Sci USA. 106:13383–13387. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Kong D, He M, Yang L, Zhou R, Yan YQ, Liang Y and Teng CB: MiR-17 and miR-19 cooperatively promote skeletal muscle cell differentiation. Cell Mol Life Sci. 76:5041–5054. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Attaix D, Combaret L, Bechet D and Taillandier D: Role of the ubiquitin-proteasome pathway in muscle atrophy in cachexia. Curr Opin Support Palliat Care. 2:262–266. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Hartmann-Petersen R and Gordon C: Proteins interacting with the 26S proteasome. Cell Mol Life Sci. 61:1589–1595. 2004.PubMed/NCBI | |
|
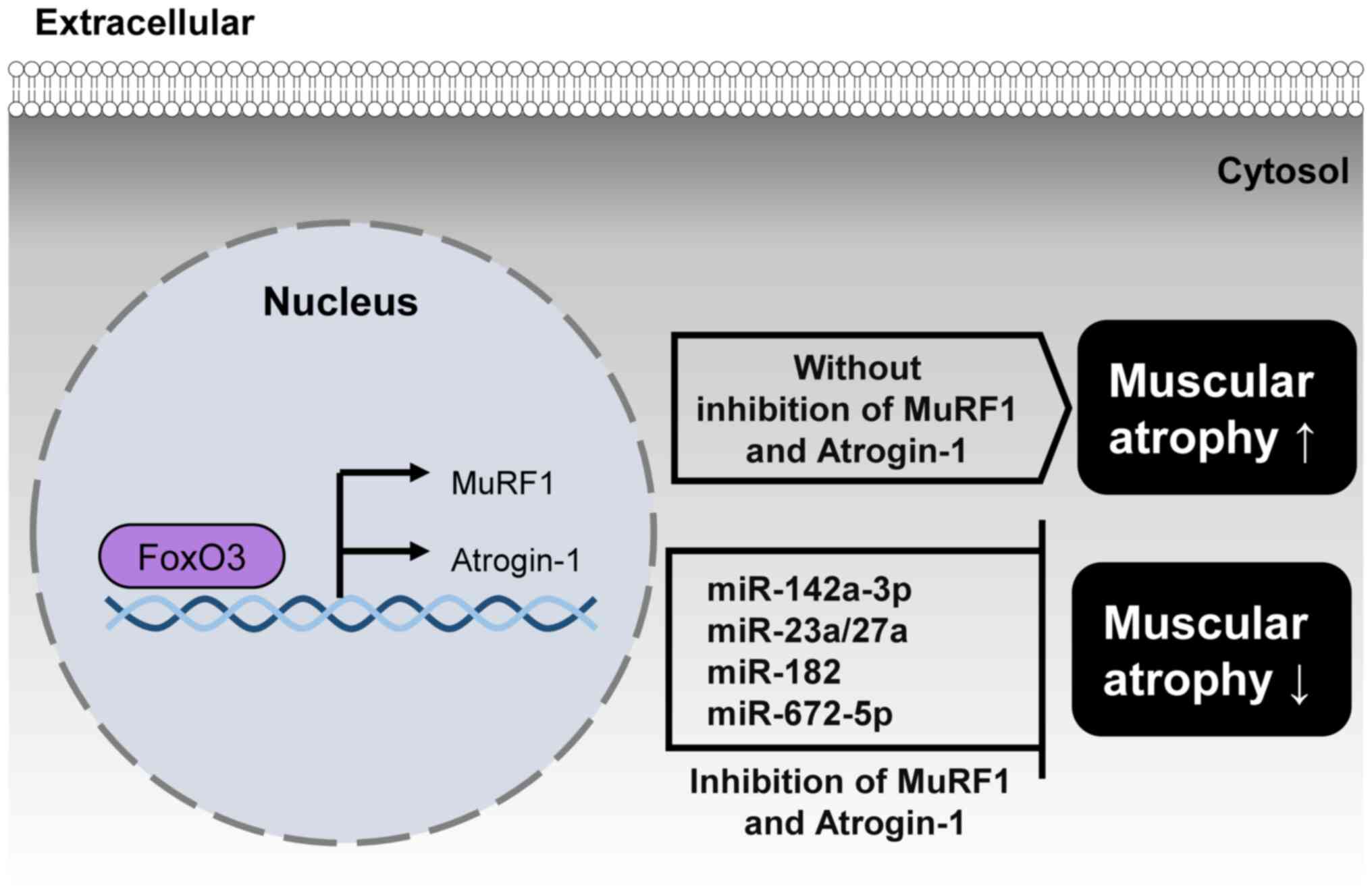
Bodine SC, Latres E, Baumhueter S, Lai VK, Nunez L, Clarke BA, Poueymirou WT, Panaro FJ, Na E, Dharmarajan K, et al: Identification of ubiquitin ligases required for skeletal muscle atrophy. Science. 294:1704–1708. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Eddins MJ, Marblestone JG, Suresh Kumar KG, Leach CA, Sterner DE, Mattern MR and Nicholson B: Targeting the ubiquitin E3 ligase MuRF1 to inhibit muscle atrophy. Cell Biochem Biophys. 60:113–118. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Clavel S, Coldefy AS, Kurkdjian E, Salles J, Margaritis I and Derijard B: Atrophy-related ubiquitin ligases, atrogin-1 and MuRF1 are up-regulated in aged rat Tibialis Anterior muscle. Mech Ageing Dev. 127:794–801. 2006. View Article : Google Scholar : PubMed/NCBI | |
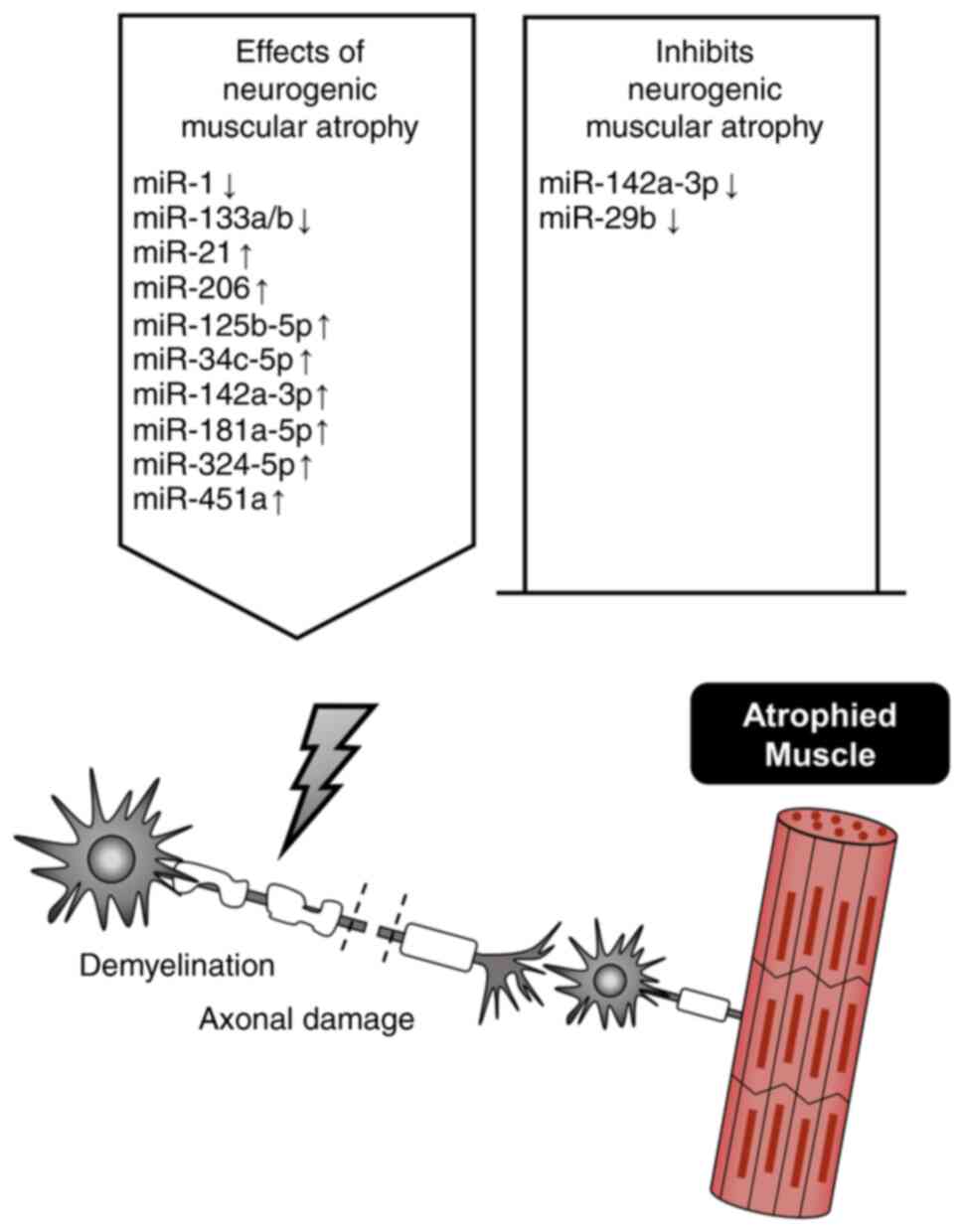
|
Gu X, Wang S, Li D, Jin B, Qi Z, Deng J, Huang C and Yin X: MicroRNA-142a-3p regulates neurogenic skeletal muscle atrophy by targeting Mef2a. Mol Ther Nucleic Acids. 33:191–204. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Xhuti D, Nilsson MI, Manta K, Tarnopolsky MA and Nederveen JP: Circulating exosome-like vesicle and skeletal muscle microRNAs are altered with age and resistance training. J Physiol. 601:5051–5073. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Ahmad N, Kushwaha P, Karvande A, Tripathi AK, Kothari P, Adhikary S, Khedgikar V, Mishra VK and Trivedi R: MicroRNA-672-5p identified during weaning reverses osteopenia and sarcopenia in ovariectomized mice. Mol Ther Nucleic Acids. 14:536–549. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Webster JM, Kempen LJAP, Hardy RS and Langen RCJ: Inflammation and skeletal muscle wasting during cachexia. Front Physiol. 11:5976752020. View Article : Google Scholar : PubMed/NCBI | |
|
Emery PW, Edwards RH, Rennie MJ, Souhami RL and Halliday D: Protein synthesis in muscle measured in vivo in cachectic patients with cancer. Br Med J (Clin Res Ed). 289:584–586. 1984. View Article : Google Scholar : PubMed/NCBI | |
|
Warnold I, Lundholm K and Schersten T: Energy balance and body composition in cancer patients. Cancer Res. 38:1801–1807. 1978.PubMed/NCBI | |
|
Chang VT, Xia Q and Kasimis B: The functional assessment of anorexia/cachexia therapy (FAACT) Appetite Scale in veteran cancer patients. J Support Oncol. 3:377–382. 2005.PubMed/NCBI | |
|
Martin L, Birdsell L, Macdonald N, Reiman T, Clandinin MT, McCargar LJ, Murphy R, Ghosh S, Sawyer MB and Baracos VE: Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J Clin Oncol. 31:1539–1547. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Yang W, Huang J, Wu H, Wang Y, Du Z, Ling Y, Wang W, Wu Q and Gao W: Molecular mechanisms of cancer cachexia-induced muscle atrophy (Review). Mol Med Rep. 22:4967–4980. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Bilodeau PA, Coyne ES and Wing SS: The ubiquitin proteasome system in atrophying skeletal muscle: Roles and regulation. Am J Physiol Cell Physiol. 311:C392–C403. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
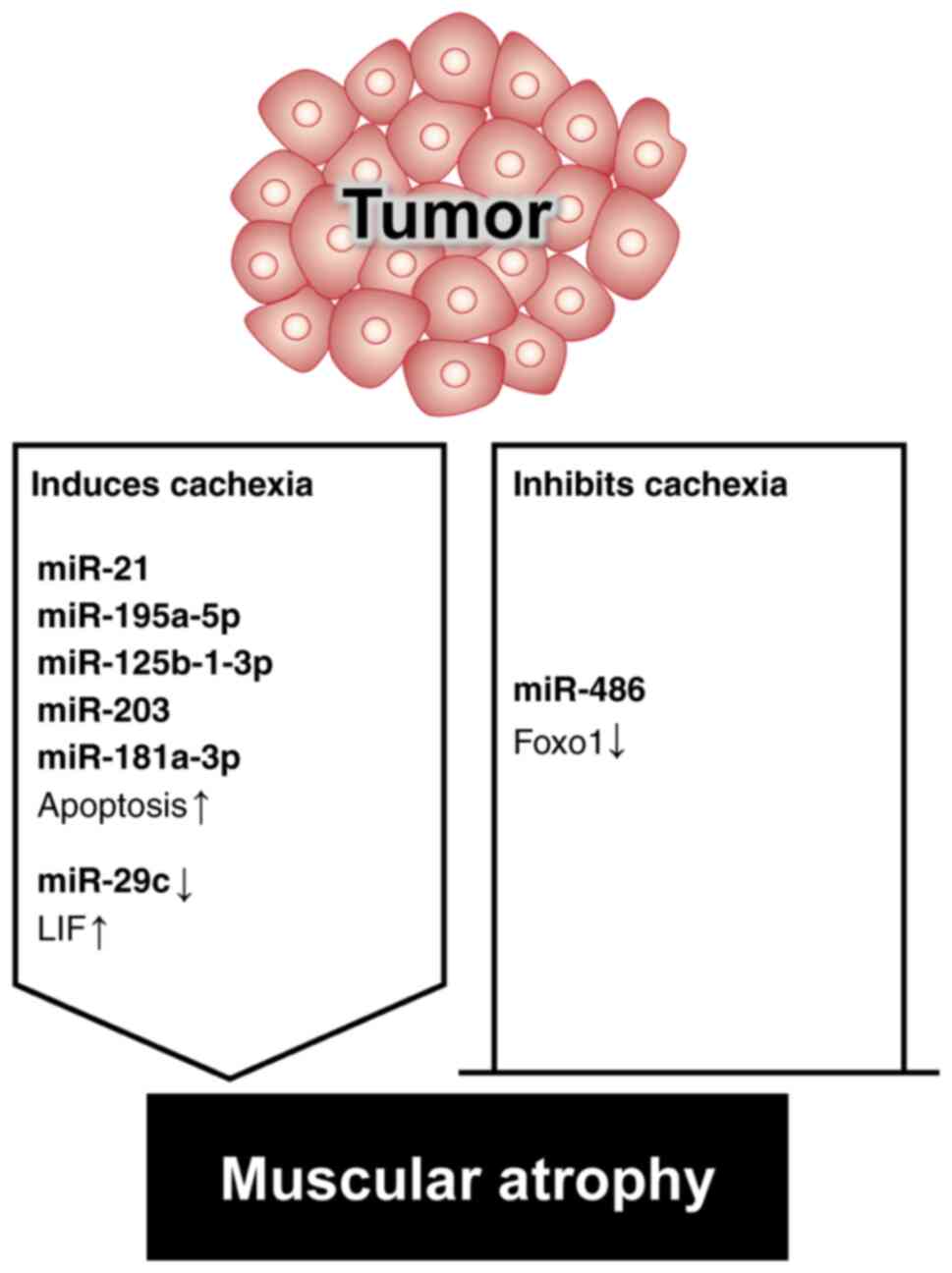
Reed SA, Sandesara PB, Senf SM and Judge AR: Inhibition of FoxO transcriptional activity prevents muscle fiber atrophy during cachexia and induces hypertrophy. FASEB J. 26:987–1000. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Xu J, Li R, Workeneh B, Dong Y, Wang X and Hu Z: Transcription factor FoxO1, the dominant mediator of muscle wasting in chronic kidney disease, is inhibited by microRNA-486. Kidney Int. 82:401–411. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
He WA, Calore F, Londhe P, Canella A, Guttridge DC and Croce CM: Microvesicles containing miRNAs promote muscle cell death in cancer cachexia via TLR7. Proc Natl Acad Sci USA. 111:4525–4529. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Xie K, Xiong H, Xiao W, Xiong Z, Hu W, Ye J, Xu N, Shi J, Yuan C, Chen Z, et al: Downregulation of miR-29c promotes muscle wasting by modulating the activity of leukemia inhibitory factor in lung cancer cachexia. Cancer Cell Int. 21:6272021. View Article : Google Scholar : PubMed/NCBI | |
|
Miao C, Zhang W, Feng F, Gu X, Shen Q, Lu S, Fan M, Li Y, Guo X, Ma Y, et al: Cancer-derived exosome miRNAs induce skeletal muscle wasting by Bcl-2-mediated apoptosis in colon cancer cachexia. Mol Ther Nucleic Acids. 24:923–938. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Okugawa Y, Toiyama Y, Hur K, Yamamoto A, Yin C, Ide S, Kitajima T, Fujikawa H, Yasuda H, Koike Y, et al: Circulating miR-203 derived from metastatic tissues promotes myopenia in colorectal cancer patients. J Cachexia Sarcopenia Muscle. 10:536–548. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Qiu L, Chen W, Wu C, Yuan Y and Li Y: Exosomes of oral squamous cell carcinoma cells containing miR-181a-3p induce muscle cell atrophy and apoptosis by transmissible endoplasmic reticulum stress signaling. Biochem Biophys Res Commun. 533:831–837. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Su SF, Chang YW, Andreu-Vieyra C, Fang JY, Yang Y, Han B, Lee AS and Liang G: miR-30d, miR-181a and miR-199a-5p cooperatively suppress the endoplasmic reticulum chaperone and signaling regulator GRP78 in cancer. Oncogene. 32:4694–4701. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Liu J, Huang Y, Cai F, Dang Y, Liu C and Wang J: MicroRNA-181a regulates endoplasmic reticulum stress in offspring of mice following prenatal microcystin-LR exposure. Chemosphere. 240:1249052020. View Article : Google Scholar : PubMed/NCBI | |
|
Wei Y, Tao X, Xu H, Chen Y, Zhu L, Tang G, Li M, Jiang A, Shuai S, Ma J, et al: Role of miR-181a-5p and endoplasmic reticulum stress in the regulation of myogenic differentiation. Gene. 592:60–70. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang M, Zhang Q, Hu Y, Xu L, Jiang Y, Zhang C, Ding L, Jiang R, Sun J, Sun H and Yan G: miR-181a increases FoxO1 acetylation and promotes granulosa cell apoptosis via SIRT1 downregulation. Cell Death Dis. 8:e30882017. View Article : Google Scholar : PubMed/NCBI | |
|
Cisterna BA, Vargas AA, Puebla C, Fernandez P, Escamilla R, Lagos CF, Matus MF, Vilos C, Cea LA, Barnafi E, et al: Active acetylcholine receptors prevent the atrophy of skeletal muscles and favor reinnervation. Nat Commun. 11:10732020. View Article : Google Scholar : PubMed/NCBI | |
|
Burke RE: Sir Charles Sherrington's the integrative action of the nervous system: A centenary appreciation. Brain. 130:887–894. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Dulhunty AF: Excitation-contraction coupling from the 1950s into the new millennium. Clin Exp Pharmacol Physiol. 33:763–772. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Canfora I, Tarantino N and Pierno S: Metabolic pathways and ion channels involved in skeletal muscle atrophy: A starting point for potential therapeutic strategies. Cells. 11:25662022. View Article : Google Scholar : PubMed/NCBI | |
|
Bruusgaard JC and Gundersen K: In vivo time-lapse microscopy reveals no loss of murine myonuclei during weeks of muscle atrophy. J Clin Invest. 118:1450–1457. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
De Gasperi R, Hamidi S, Harlow LM, Ksiezak-Reding H, Bauman WA and Cardozo CP: Denervation-related alterations and biological activity of miRNAs contained in exosomes released by skeletal muscle fibers. Sci Rep. 7:128882017. View Article : Google Scholar : PubMed/NCBI | |
|
Magnusson C, Svensson A, Christerson U and Tagerud S: Denervation-induced alterations in gene expression in mouse skeletal muscle. Eur J Neurosci. 21:577–580. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Ehmsen JT and Hoke A: Cellular and molecular features of neurogenic skeletal muscle atrophy. Exp Neurol. 331:1133792020. View Article : Google Scholar : PubMed/NCBI | |
|
Daeschler SC, Feinberg K, Harhaus L, Kneser U, Gordon T and Borschel GH: Advancing nerve regeneration: Translational perspectives of tacrolimus (FK506). Int J Mol Sci. 24:127712023. View Article : Google Scholar : PubMed/NCBI | |
|
Zheng H, Liu X, Katsurada K and Patel KP: Renal denervation improves sodium excretion in rats with chronic heart failure: Effects on expression of renal ENaC and AQP2. Am J Physiol Heart Circ Physiol. 317:H958–H968. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Tokinoya K, Shirai T, Ota Y, Takemasa T and Takekoshi K: Denervation-induced muscle atrophy suppression in renalase-deficient mice via increased protein synthesis. Physiol Rep. 8:e144752020. View Article : Google Scholar : PubMed/NCBI | |
|
Sandri M: Protein breakdown in muscle wasting: Role of autophagy-lysosome and ubiquitin-proteasome. Int J Biochem Cell Biol. 45:2121–2129. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Bongers KS, Fox DK, Ebert SM, Kunkel SD, Dyle MC, Bullard SA, Dierdorff JM and Adams CM: Skeletal muscle denervation causes skeletal muscle atrophy through a pathway that involves both Gadd45a and HDAC4. Am J Physiol Endocrinol Metab. 305:E907–E915. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Weng J, Zhang P, Yin X and Jiang B: The whole transcriptome involved in denervated muscle atrophy following peripheral nerve injury. Front Mol Neurosci. 11:692018. View Article : Google Scholar : PubMed/NCBI | |
|
Nimmo R, Ciau-Uitz A, Ruiz-Herguido C, Soneji S, Bigas A, Patient R and Enver T: MiR-142-3p controls the specification of definitive hemangioblasts during ontogeny. Dev Cell. 26:237–249. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Huang QK, Qiao HY, Fu MH, Li G, Li WB, Chen Z, Wei J and Liang BS: MiR-206 Attenuates Denervation-Induced Skeletal Muscle Atrophy in Rats Through Regulation of Satellite Cell Differentiation via TGF-beta1, Smad3, and HDAC4 Signaling. Med Sci Monit. 22:1161–1170. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Li J, Wang L, Hua X, Tang H, Chen R, Yang T, Das S and Xiao J: CRISPR/Cas9-Mediated miR-29b editing as a treatment of different types of muscle atrophy in mice. Mol Ther. 28:1359–1372. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Srivastava S, Rathor R, Singh SN and Suryakumar G: Emerging role of MyomiRs as biomarkers and therapeutic targets in skeletal muscle diseases. Am J Physiol Cell Physiol. 321:C859–C875. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Gu XY, Jin B, Qi ZD and Yin XF: MicroRNA is a potential target for therapies to improve the physiological function of skeletal muscle after trauma. Neural Regen Res. 17:1617–1622. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Abiusi E, Infante P, Cagnoli C, Lospinoso Severini L, Pane M, Coratti G, Pera MC, D'Amico A, Diano F, Novelli A, et al: SMA-miRs (miR-181a-5p, −324-5p, and −451a) are overexpressed in spinal muscular atrophy skeletal muscle and serum samples. Elife. 10:e680542021. View Article : Google Scholar : PubMed/NCBI |









