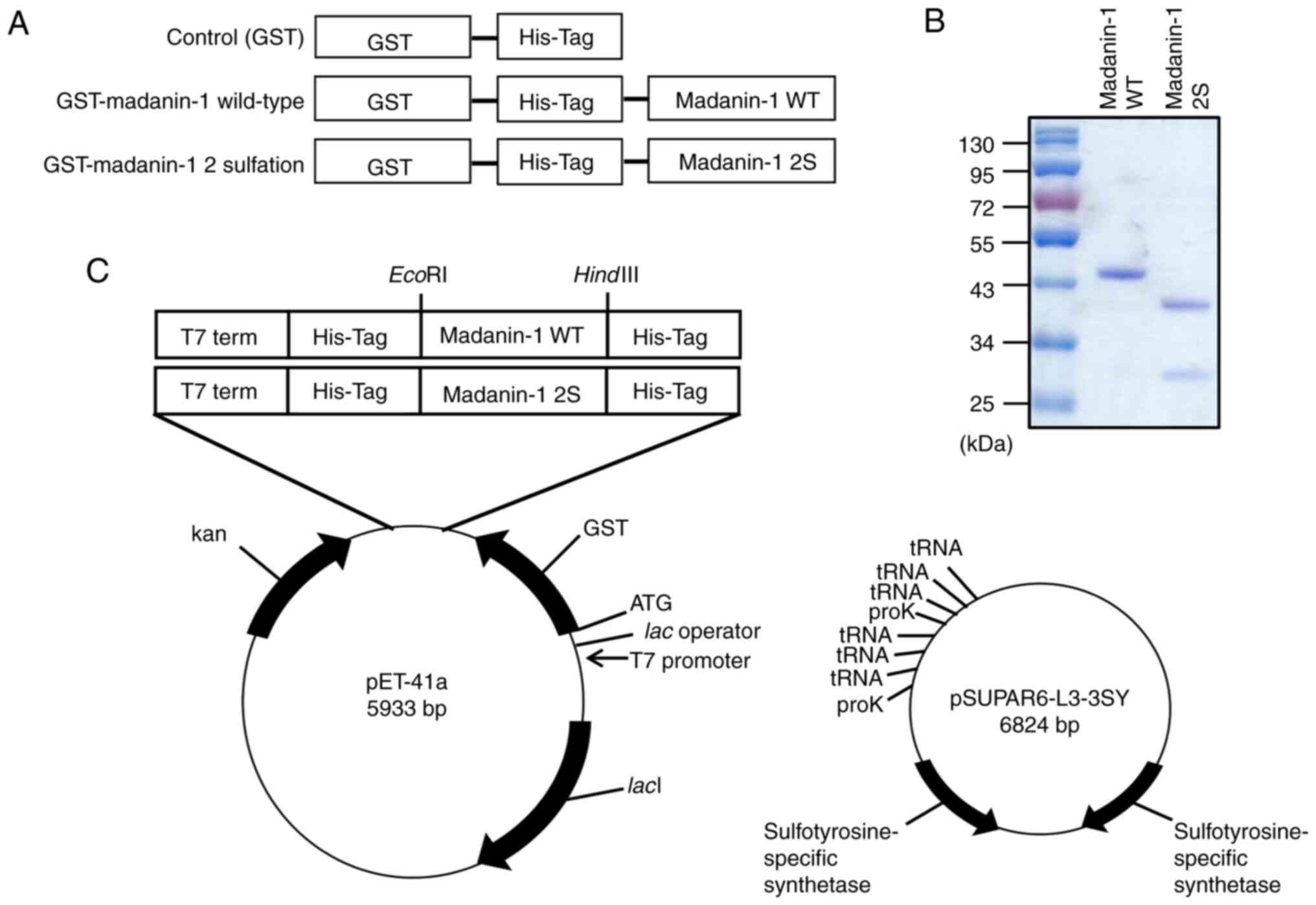
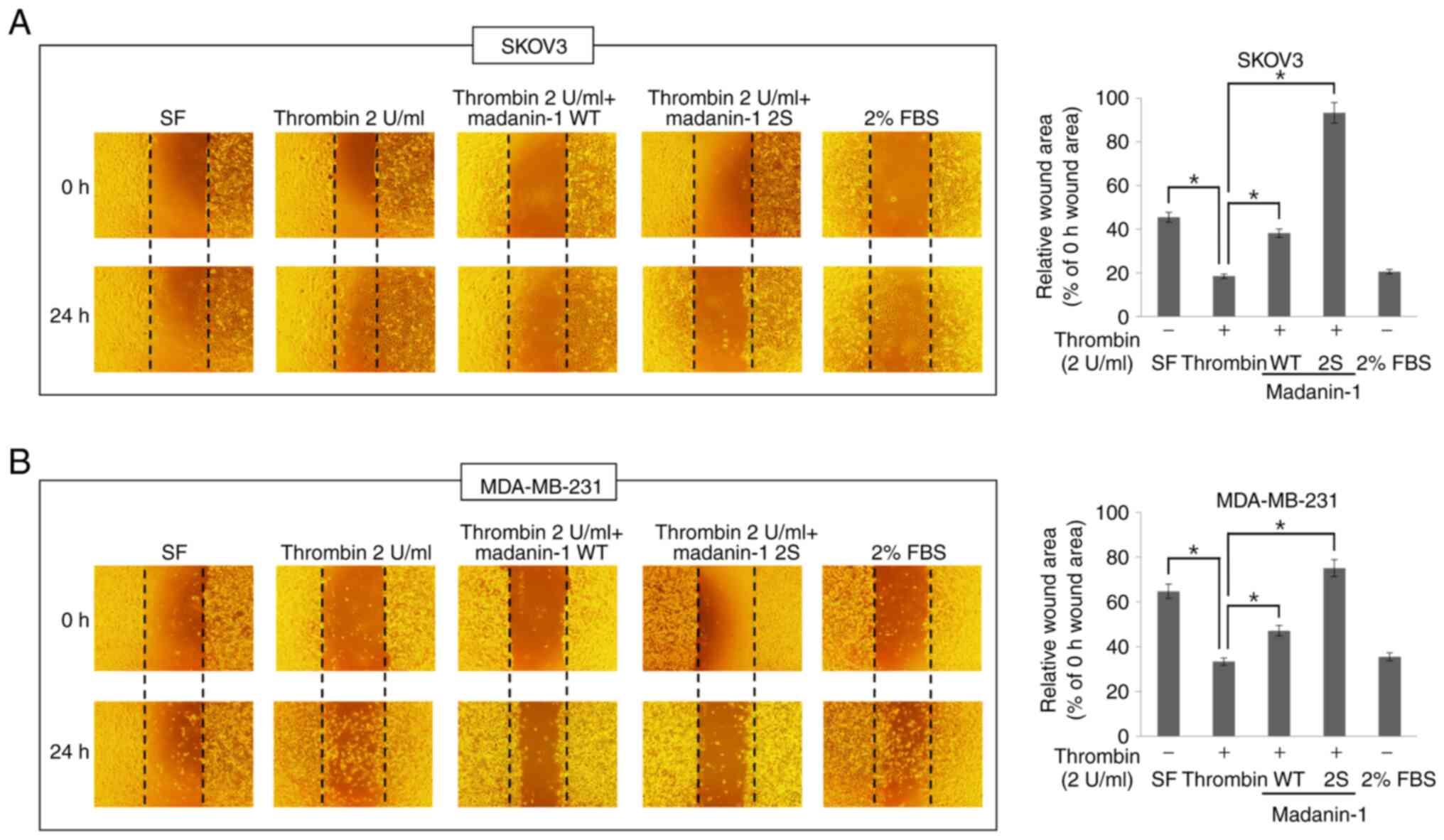
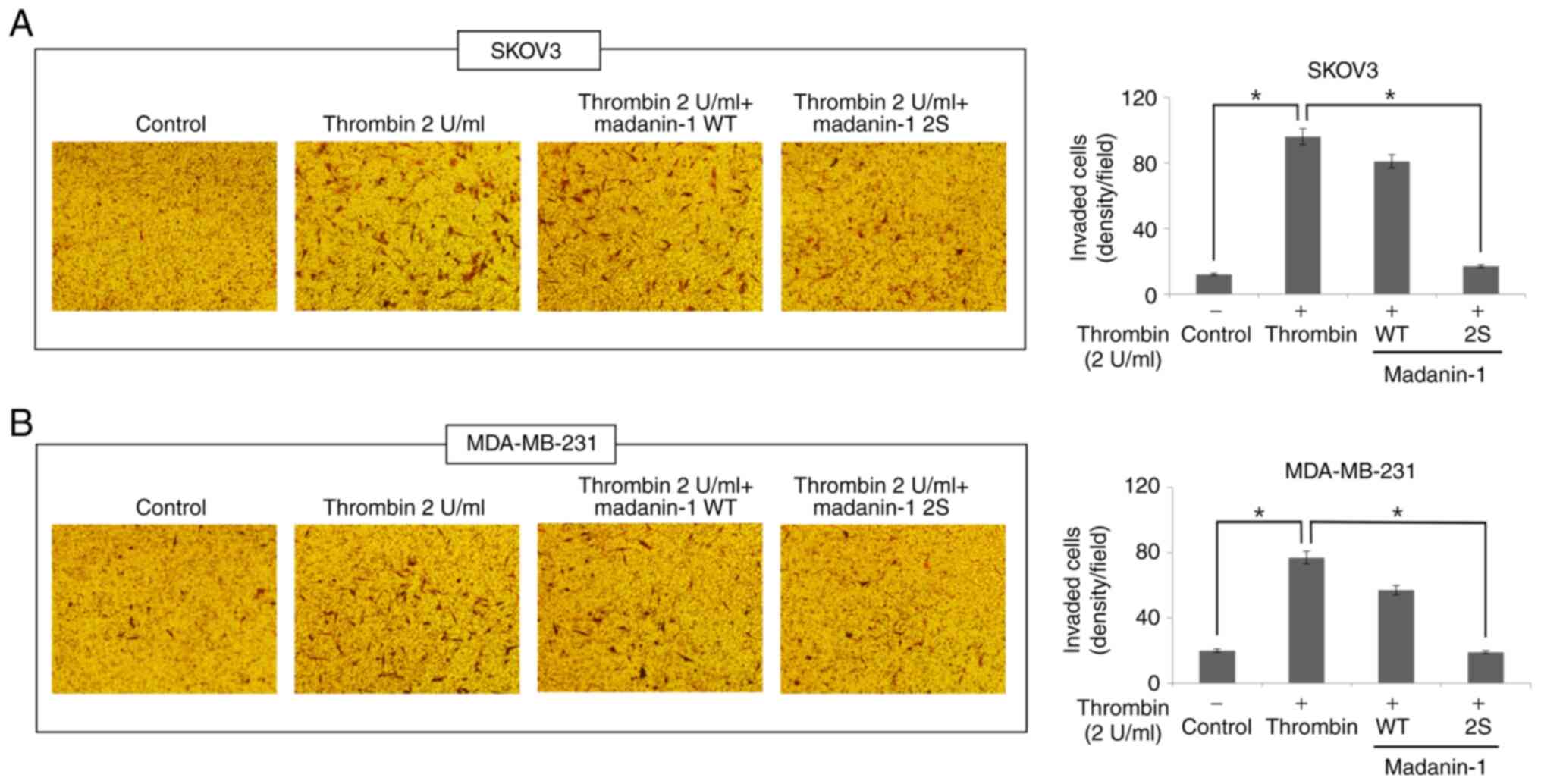
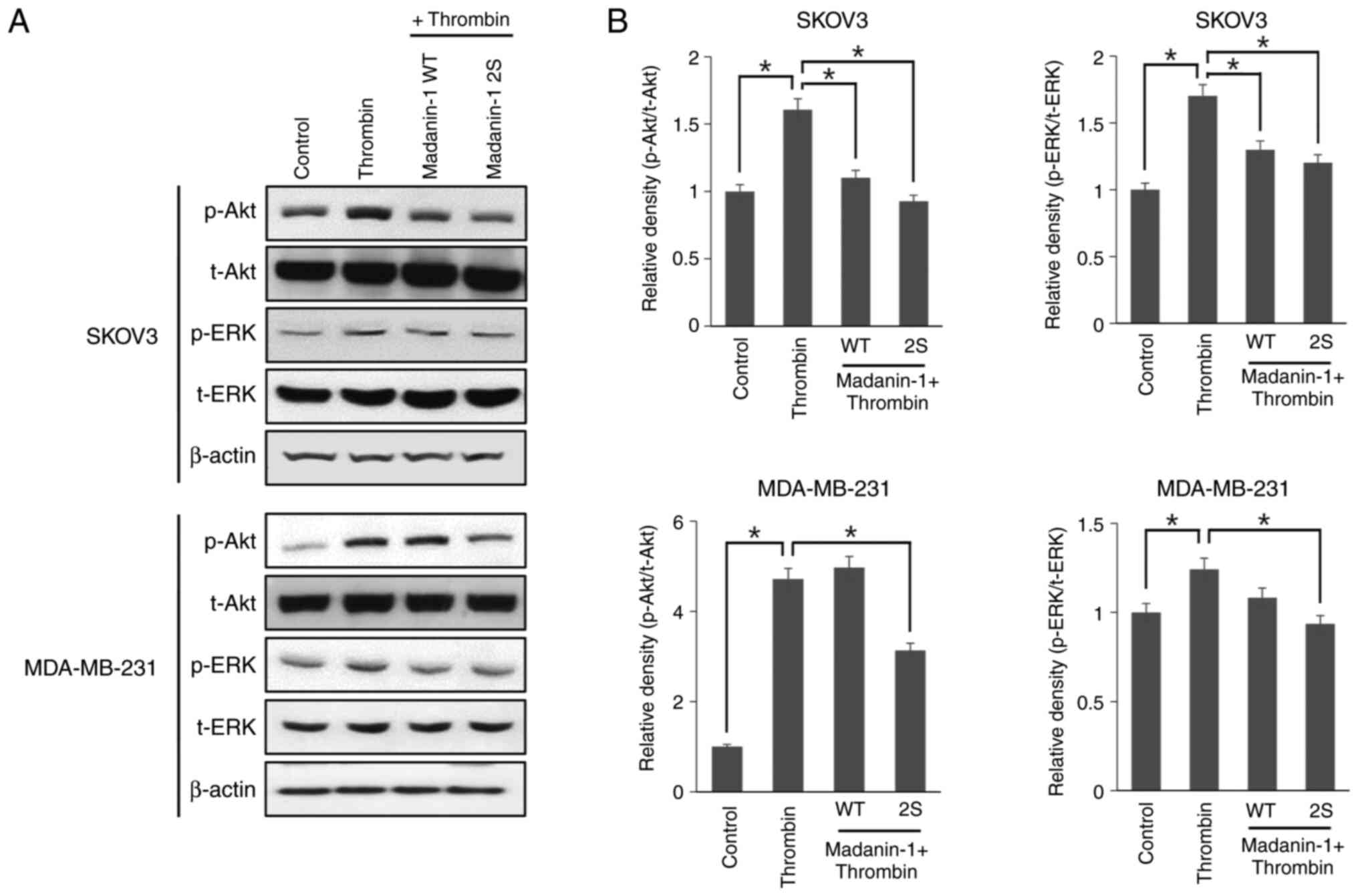
|
1
|
Larsen JB and Hvas AM: Thrombin: A pivotal
player in hemostasis and beyond. Semin Thromb Hemost. 47:759–774.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen LB and Buchanan JM: Mitogenic
activity of blood components. I. Thrombin and prothrombin. Proc
Natl Acad Sci USA. 72:131–135. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Maruyama I, Shigeta K, Miyahara H,
Nakajima T, Shin H, Ide S and Kitajima I: Thrombin activates
NF-kappa B through thrombin receptor and results in proliferation
of vascular smooth muscle cells: Role of thrombin in
atherosclerosis and restenosis. Ann N Y Acad Sci. 811:429–436.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou S, Xiao W, Pan X, Zhu M, Yang Z,
Zhang F and Zheng C: Thrombin promotes proliferation of human lung
fibroblasts via protease activated receptor-1-dependent and
NF-κB-independent pathways. Cell Biol Int. 38:747–756. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Szaba FM and Smiley ST: Roles for thrombin
and fibrin(ogen) in cytokine/chemokine production and macrophage
adhesion in vivo. Blood. 99:1053–1059. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chiang HS, Yang RS and Huang TF: Thrombin
enhances the adhesion and migration of human colon adenocarcinoma
cells via increased beta 3-integrin expression on the tumour cell
surface and their inhibition by the snake venom peptide,
rhodostomin. Br J Cancer. 73:902–908. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nierodzik ML and Karpatkin S: Thrombin
induces tumor growth, metastasis, and angiogenesis: Evidence for a
thrombin-regulated dormant tumor phenotype. Cancer Cell.
10:355–362. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Franchini M and Mannucci PM: Thrombin and
cancer: From molecular basis to therapeutic implications. Semin
Thromb Hemost. 38:95–101. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cantrell R and Palumbo JS: The
thrombin-inflammation axis in cancer progression. Thromb Res. 191
(Suppl 1):S117–S122. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Covic L and Kuliopulos A:
Protease-activated receptor 1 as therapeutic target in breast,
lung, and ovarian cancer: Pepducin approach. Int J Mol Sci.
19:22372018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu X, Yu J, Song S, Yue X and Li Q:
Protease-activated receptor-1 (PAR-1): A promising molecular target
for cancer. Oncotarget. 8:107334–107345. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jacobs JW, Cupp EW, Sardana M and Friedman
PA: Isolation and characterization of a coagulation factor Xa
inhibitor from black fly salivary glands. Thromb Haemost.
64:235–238. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Waxman L, Smith DE, Arcuri KE and Vlasuk
GP: Tick anticoagulant peptide (TAP) is a novel inhibitor of blood
coagulation factor Xa. Science. 248:593–596. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Salzet M: Leech thrombin inhibitors. Curr
Pharm Des. 8:493–503. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Figueiredo AC, de Sanctis D and Pereira
PJ: The tick-derived anticoagulant madanin is processed by thrombin
and factor Xa. PLoS One. 8:e718662013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Iwanaga S, Okada M, Isawa H, Morita A,
Yuda M and Chinzei Y: Identification and characterization of novel
salivary thrombin inhibitors from the ixodidae tick,
Haemaphysalis longicornis. Eur J Biochem. 270:1926–1934.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Thompson RE, Liu X, Ripoll-Rozada J,
Alonso-Garcia N, Parker BL, Pereira PJB and Payne RJ: Tyrosine
sulfation modulates activity of tick-derived thrombin inhibitors.
Nat Chem. 9:909–917. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Khorana AA, Mackman N, Falanga A, Pabinger
I, Noble S, Ageno W, Moik F and Lee AYY: Cancer-associated venous
thromboembolism. Nat Rev Dis Primers. 8:112022. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Asanuma K, Wakabayashi H, Okamoto T,
Asanuma Y, Akita N, Yoshikawa T, Hayashi T, Matsumine A, Uchida A
and Sudo A: The thrombin inhibitor, argatroban, inhibits breast
cancer metastasis to bone. Breast Cancer. 20:241–246. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Alexander ET, Minton AR, Peters MC, van
Ryn J and Gilmour SK: Thrombin inhibition and cisplatin block tumor
progression in ovarian cancer by alleviating the immunosuppressive
microenvironment. Oncotarget. 7:85291–85305. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chanakira A, Westmark PR, Ong IM and
Sheehan JP: Tissue factor-factor VIIa complex triggers protease
activated receptor 2-dependent growth factor release and migration
in ovarian cancer. Gynecol Oncol. 145:167–175. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chaffer CL and Weinberg RA: A perspective
on cancer cell metastasis. Science. 331:1559–1564. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Radjabi AR, Sawada K, Jagadeeswaran S,
Eichbichler A, Kenny HA, Montag A, Bruno K and Lengyel E: Thrombin
induces tumor invasion through the induction and association of
matrix metalloproteinase-9 and beta1-integrin on the cell surface.
J Biol Chem. 283:2822–2834. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim J, Lee JW, Kim SI, Choi YJ, Lee WK,
Jeong MJ, Cha SH, Lee HJ, Chun W and Kim SS: Thrombin-induced
migration and matrix metalloproteinase-9 expression are regulated
by MAPK and PI3K pathways in C6 glioma cells. Korean J Physiol
Pharmacol. 15:211–216. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhong YC, Zhang T, Di W and Li WP:
Thrombin promotes epithelial ovarian cancer cell invasion by
inducing epithelial-mesenchymal transition. J Gynecol Oncol.
24:265–272. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Justus CR, Leffler N, Ruiz-Echevarria M
and Yang LV: In vitro cell migration and invasion assays. J Vis
Exp. 510462014.PubMed/NCBI
|
|
27
|
Liu CC, Cellitti SE, Geierstanger BH and
Schultz PG: Efficient expression of tyrosine-sulfated proteins in
E. coli using an expanded genetic code. Nat Protoc.
4:1784–1789. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kirwan CC, Descamps T and Castle J:
Circulating tumour cells and hypercoagulability: A lethal
relationship in metastatic breast cancer. Clin Transl Oncol.
22:870–877. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Reddel CJ, Tan CW and Chen VM: Thrombin
generation and cancer: Contributors and consequences. Cancers
(Basel). 11:1002019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shaker H, Bundred NJ, Landberg G,
Pritchard SA, Albadry H, Nicholson SL, Harries LJ, Heah JYE, Castle
J and Kirwan CC: Breast cancer stromal clotting activation (tissue
factor and thrombin): A pre-invasive phenomena that is prognostic
in invasion. Cancer Med. 9:1768–1778. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ossovskaya VS and Bunnett NW:
Protease-activated receptors: Contribution to physiology and
disease. Physiol Rev. 84:579–621. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wojtukiewicz MZ, Hempel D, Sierko E,
Tucker SC and Honn KV: Protease-activated receptors (PARs)-biology
and role in cancer invasion and metastasis. Cancer Metastasis Rev.
34:775–796. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Alexander ET and Gilmour SK:
Immunomodulatory role of thrombin in cancer progression. Mol
Carcinog. 61:527–536. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ohshiro K, Bui-Nguyen TM, Divijendra Natha
RS, Schwartz AM, Levine P and Kumar R: Thrombin stimulation of
inflammatory breast cancer cells leads to aggressiveness via the
EGFR-PAR1-Pak1 pathway. Int J Biol Markers. 27:e305–e313. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhao B, Wu M, Hu Z, Wang T, Yu J, Ma Y,
Wang Q, Zhang Y, Chen D, Li T, et al: A novel oncotherapy strategy:
Direct thrombin inhibitors suppress progression, dissemination and
spontaneous metastasis in non-small cell lung cancer. Br J
Pharmacol. 179:5056–5073. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang YS, Wang CC, Chen BH, Hou YH, Hung KS
and Mao YC: Tyrosine sulfation as a protein post-translational
modification. Molecules. 20:2138–2164. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Corral-Rodriguez MA, Macedo-Ribeiro S,
Pereira PJ and Fuentes-Prior P: Leech-derived thrombin inhibitors:
From structures to mechanisms to clinical applications. J Med Chem.
53:3847–3861. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ouyang YB and Moore KL: Molecular cloning
and expression of human and mouse tyrosylprotein sulfotransferase-2
and a tyrosylprotein sulfotransferase homologue in Caenorhabditis
elegans. J Biol Chem. 273:24770–24774. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Dongre A and Weinberg RA: New insights
into the mechanisms of epithelial-mesenchymal transition and
implications for cancer. Nat Rev Mol Cell Biol. 20:69–84. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huber MA, Kraut N and Beug H: Molecular
requirements for epithelial-mesenchymal transition during tumor
progression. Curr Opin Cell Biol. 17:548–558. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhu X, Wang X, Gong Y and Deng J:
E-cadherin on epithelial-mesenchymal transition in thyroid cancer.
Cancer Cell Int. 21:6952021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Mrozik KM, Blaschuk OW, Cheong CM,
Zannettino ACW and Vandyke K: N-cadherin in cancer metastasis, its
emerging role in haematological malignancies and potential as a
therapeutic target in cancer. BMC Cancer. 18:9392018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Song Y, Ye M, Zhou J, Wang ZW and Zhu X:
Restoring E-cadherin expression by natural compounds for anticancer
therapies in genital and urinary cancers. Mol Ther Oncolytics.
14:130–138. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Song Y, Ye M, Zhou J, Wang Z and Zhu X:
Targeting E-cadherin expression with small molecules for digestive
cancer treatment. Am J Transl Res. 11:3932–3944. 2019.PubMed/NCBI
|
|
45
|
Otsuki T, Fujimoto D, Hirono Y, Goi T and
Yamaguchi A: Thrombin conducts epithelial-mesenchymal transition
via protease-activated receptor-1 in human gastric cancer. Int J
Oncol. 45:2287–2294. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cao Z, Liao Q, Su M, Huang K, Jin J and
Cao D: AKT and ERK dual inhibitors: The way forward? Cancer Lett.
459:30–40. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Asati V, Mahapatra DK and Bharti SK:
PI3K/Akt/mTOR and Ras/Raf/MEK/ERK signaling pathways inhibitors as
anticancer agents: Structural and pharmacological perspectives. Eur
J Med Chem. 109:314–341. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Karimi Roshan M, Soltani A, Soleimani A,
Rezaie Kahkhaie K, Afshari AR and Soukhtanloo M: Role of AKT and
mTOR signaling pathways in the induction of epithelial-mesenchymal
transition (EMT) process. Biochimie. 165:229–234. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Li Q, Li Z, Luo T and Shi H: Targeting the
PI3K/AKT/mTOR and RAF/MEK/ERK pathways for cancer therapy. Mol
Biomed. 3:472022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Narayan P, Prowell TM, Gao JJ, Fernandes
LL, Li E, Jiang X, Qiu J, Fan J, Song P, Yu J, et al: FDA approval
summary: Alpelisib plus fulvestrant for patients with HR-positive,
HER2-negative, PIK3CA-mutated, advanced or metastatic breast
cancer. Clin Cancer Res. 27:1842–1849. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yuan Y, Jiang YC, Sun CK and Chen QM: Role
of the tumor microenvironment in tumor progression and the clinical
applications (review). Oncol Rep. 35:2499–2515. 2016. View Article : Google Scholar : PubMed/NCBI
|