Pancreatic stellate cells and the interleukin family: Linking fibrosis and immunity to pancreatic ductal adenocarcinoma (Review)
- Authors:
- Published online on: July 5, 2024 https://doi.org/10.3892/mmr.2024.13283
- Article Number: 159
-
Copyright: © Li et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Pancreatic ductal adenocarcinoma (PDAC), globally recognized as the ‘king of cancers’, is projected to become the second deadliest cancer in the United States by 2026, but its cause remains elusive (1,2). This lethal disease is typically diagnosed at an advanced metastatic stage owing to a lack of early clinical manifestations (3). The mortality rate of patients with PDAC closely mirrors the morbidity rate, with a brief disease course and a survival period typically <1 year (4). Early screening techniques such as endoscopic ultrasound, magnetic resonance/magnetic resonance pancreaticobiliary imaging and PDAC detection with artificial intelligence can accurately detect and classify pancreatic lesions using non-contrast CT (5,6). However, early diagnosis and screening for pancreatic cancer remain extremely limited in most countries (7).
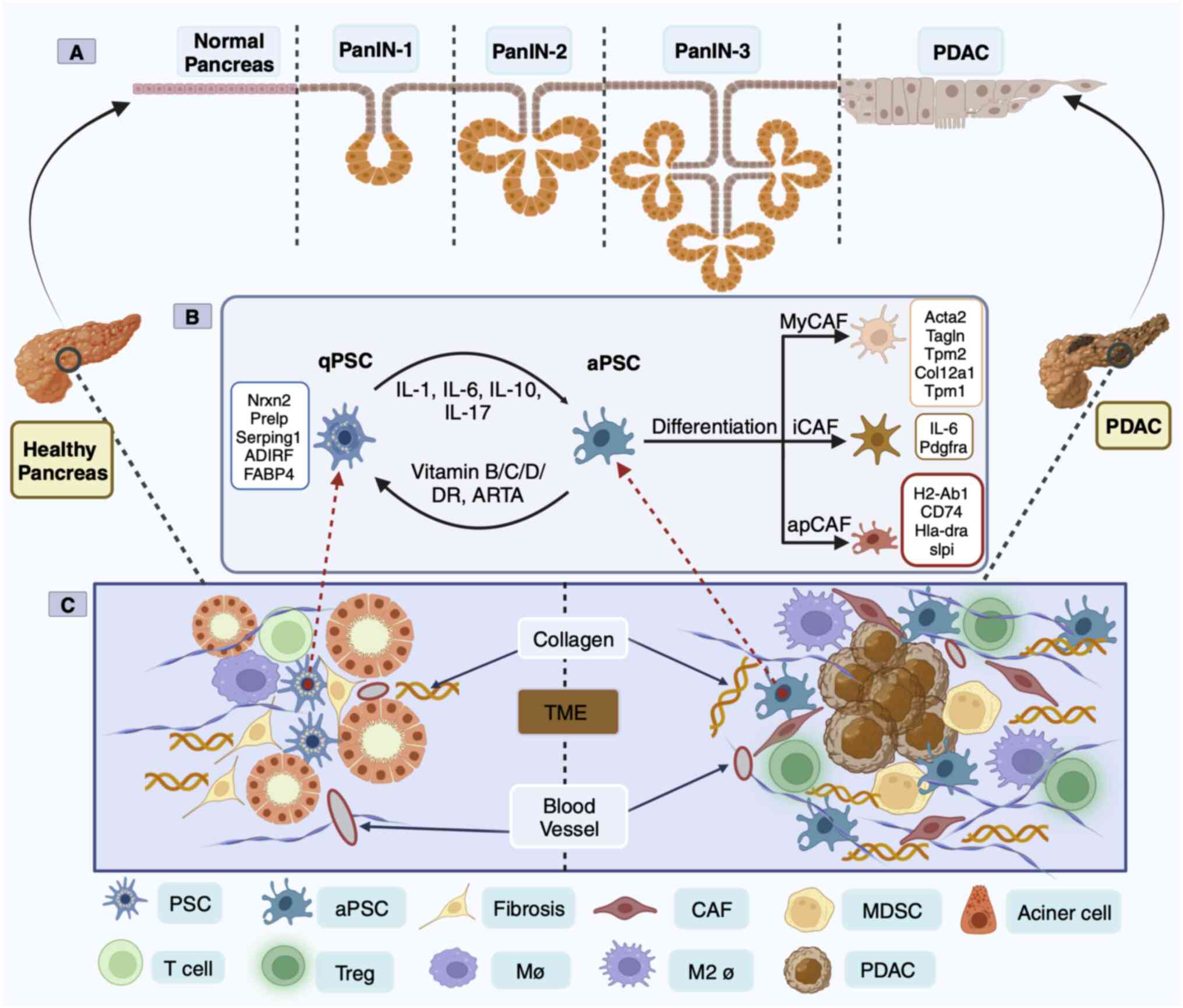
Yachida et al (8) proposed a model for the genetic evolution of PDAC, dividing its development into stages of precursor lesion pancreatic intraepithelial neoplasia (PanIN), invasive carcinoma to a metastatic mass and metastatic transmission to death. In the past few years, single-cell RNA sequencing (scRNA-seq) has revealed that the tumor microenvironment (TME) of PDAC is a complex ecosystem with intricate cellular composition and heterogeneity among populations (9). The TME is characterized by an abundance of extracellular matrix (ECM), including cell components such as fibroblasts and immune cells (10). Moreover, the progression from early to late PDAC is characterized by a decrease in the number of cancer-associated fibroblasts (CAFs), which are composed of fibroblasts and stellate cells, and a progressive increase in the proportion of immune cells (11). Throughout these stages, a transition from a proinflammatory microenvironment to a highly fibrotic and immunosuppressive TME was observed (Fig. 1) (9,12). Therefore, fibrosis and immunity are indispensable factors in the progression of PanIN to PDAC. Activated pancreatic stellate cells (aPSCs), which constitute ~50% of the TME, dominate dense stroma composed of PDAC fibroblasts (13). These aPSCs are present at all stages of pancreatic cancer, including early PanIN lesions, where they accelerate the progression of high-grade PanIN lesions, thereby exhibiting tumor-promoting properties (14). Interleukins and related cytokines are a means of communication between innate and adaptive immune cells and non-immune cells and tissues (15). Members of the interleukin family have emerged as novel protein markers for pancreatic cancer risk in the China Kadoorie Biobank Chronic Disease Prospective Project study (16).
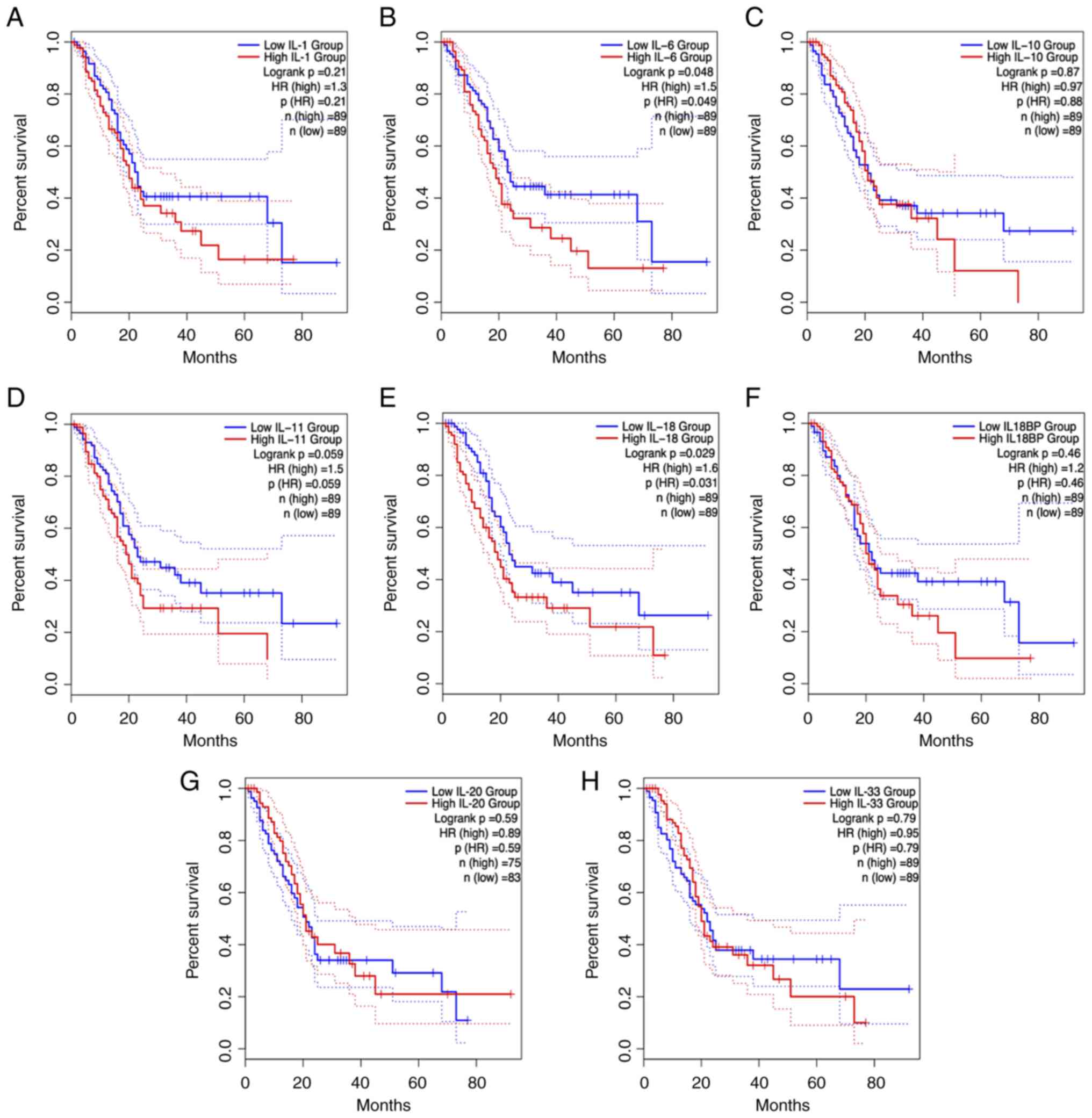
The present review focuses on the effects of aPSCs in the interleukin-1 (IL-1) family, interleukin-6 (IL-6) family, interleukin-10 (IL-10) family and interleukin-17A (IL-17A) cytokines on pancreatic cancer development (Fig. 2). In addition to the IL-1 family, the IL-6 family and IL-10 family of cytokines are involved in the survival of patients with PDAC (Fig. 3) highlighting their potential as critical targets for early cancer screening and diagnosis. These interleukin family cytokines are prospective candidates in the field of fibrosis as well as immune regulation and have considerable potential to improve the early developmental profile of PDAC (15–17). However, most other interleukin family members have only been evaluated in preclinical or clinical trials for immune regulatory signatures (Table I).
Table I.Preclinical or clinical trials of IL family members on PanIN/PDAC in the previous five years. |
Previous and current understanding of pancreatic stellate cells (PSCs) in PDAC
In 1998, PSCs from healthy human and rat pancreases were successfully isolated and cultured for the first time (18). Most of the initial understanding of PSCs was based on their similarity with hepatic stellate cells, but in-depth exploration has led to a more comprehensive understanding of the morphology and location of PSCs, as well as their unique characteristics of being able to store lipid droplets and vitamin A (19). Transcriptome analysis of human and mouse PSCs revealed two distinct clusters of PSCs including ‘activate’ [enrichment of ECM genes, collagen type 1 α1 (COL1A1) and fibronectin 1] and ‘quiescent’ (enrichment of adipose genes, adipogenesis regulatory factor and fatty acid binding protein 4) (20). Under physiological conditions, PSCs are in a quiescent state and are the only pancreatic cells that store vitamin A (21). PSC activation is characterized by a spindle-like shape in vitro and the disappearance of vitamin A lipid droplets, although the mechanism of the disappearance or absence of vitamin A in PDAC progression has yet to be elucidated (22). In the aPSC state, there are α-smooth mucle actin (SMA) and collagen fibers, ECM deposition, and the release of epithelial-mesenchymal transition (EMT)-associated soluble inflammatory factors (23). Simultaneously, aPSCs begin to proliferate and manifest the proinflammatory phenotype, releasing chemokines, cytokines and growth factors that recruit other inflammatory factors into the pancreas, perpetuating the inflammatory response (24). aPSCs can also promote an immunosuppressive microenvironment in mouse models of pancreatic cancer (25). Sustained aPSCs can create a TME conducive to cancer cell growth (Fig. 1C).
PSC-derived CAFs in human and mouse PDAC were analyzed by scRNA-seq technology and classified into myofibroblast CAFs (myCAFs), inflammatory CAFs (iCAFs) and antigen-presenting CAFs (apCAFs) based on positional and functional characteristics (26,27). These three heterogeneous CAF subsets function in mutual conversion (26,27). However, apCAFs are rarely found in patients with PDAC (9). MyCAFs dominate connective tissue proliferation and form a physical protective barrier outside the cells of pancreatic cancers, protecting them from drug intervention and immune recognition (28). ECM depletion has been proposed to remove the fibrous barrier, but it has not been successful in the treatment of pancreatic cancer (29). The depletion of SMA+ myCAFs led to a reduction in the tumor stroma in mice (30). This accelerates PanIN and PDAC formation and development and reduces survival (30). The second CAF subgroup includes iCAFs. In a broad sense, myCAFs appear to be involved in EMT and ECM remodeling, whereas iCAFs appear to be associated with inflammation and ECM deposition (31). Mouse pancreatic tumor scRNA-seq revealed that the IL-1/JAK/STAT3 and TGFβ/Smad3 signaling are key pathways that regulate iCAF and MyCAF heterogeneity and function (32). Furthermore, myCAFs and iCAFs are considered to play opposing roles in cancer (33). The third subpopulation of CAFs is apCAFs, which express a wide range of fibroblast markers including COL1A1, COL1A2, decorin and podoplanin as well as major histocompatibility complex class II (MHC II)-related genes that are limited to antigen-presenting cells (APC) of the immune system and present model antigens to CD4+ T cells in an antigen-dependent manner (26). The source and nature of the antigen presented by apCAFs are not known, which is an important mystery in the study of the interaction between CAFs and tumor-infiltrating T cells.
PSC-derived CAFs are classified as cancer-promoting CAFs (pCAFs) or cancer-restraining CAFs (rCAFs) based on their role in fighting cancer (26,34). Fibroblasts that maintain homeostasis and innately suppress tumorigenesis are collectively referred to as rCAFs (35). However, the nature and characteristic markers of rCAFs are unknown. Meflin is highly expressed in quiescent PSCs, oncogenic meflin-tagged rCAFs appear around cells in PanIN, meflin expression decreases during PDAC progression, and α-SMA expression increases, leading to phenomena such as pCAFs (34). pCAFs secrete matrix components such as collagen, fibronectin and proteoglycans that are involved in EMT, invasion, metastasis and tumor angiogenesis and have been extensively studied (35). Currently, using pharmacological methods to synthesize the unnatural retinoid Am80, clinical trials on the conversion of pCAFs to rCAFs for the treatment of pancreatic cancer have been initiated (clinical trial NCT05064618). Recently, several vitamin analogs that downregulate α-SMA, reduce the movement and activation of PSCs, restore PSCs to quiescence, and lead to increased apoptosis in neighboring cancer cells in combination with chemotherapy drugs have entered clinical trials in patients with PDAC (https://clinicaltrials.gov/; Table II). However, PSC-derived CAF subpopulations are diverse (26,27,34). Thus, interfering with PSC subpopulations is one of the more challenging therapeutic strategies for pancreatic cancer.
Interleukin family synergizes the role of the PSC in fibrosis and immune regulation in PanIN and PDAC
Spatial transcriptomics and scRNA-seq have revealed distinct transcriptomic features of PanIN fibroblasts and macrophages in healthy adult organ donors and patients with PDAC (36). Furthermore, macrophages are observed in close proximity to PSCs, and aPSCs drive anti-inflammatory M2 type macrophages to produce an immunosuppressive environment for pancreatic cancer, which may explain the aberrant interactions between PSCs and immune cells (37,38). scRNA-seq analysis conducted on PDAC samples before and after chemotherapy revealed that chemotherapy might enhance resistance to immunotherapy (9). However, T cells and macrophages are the primary populations shaping the immune landscape in the TME (39). Baron et al (20) conducted scRNA-seq on four cases of PDAC and found that PSC, in addition to expressing genes associated with the ECM, were also closely associated with high interleukin expression. Interleukins are cytokines with immunomodulatory functions that serve as a means of communication between cells and tissues (20). Interleukins cultivate an environment conducive to cancer growth and are critical for tumor immunotherapy and targeting (15).
IL-1 family
The IL-1 family was originally used to generalize the products of macrophage-induced inflammation. A total of seven agonist cytokines (IL-1α, IL-1β, IL-18, IL-33, IL-36α, IL-36β and IL-36γ), three receptor antagonists (IL-1Ra, IL-36Ra and IL-38) and one anti-inflammatory cytokine (IL-37) currently belong to the IL-1 family (40,41). With the notable exception of IL-1Ra, each family member is produced as a precursor (40). Most of these precursors, except for IL-1α and IL-33, which are biologically inactive until they are cleaved and mature, do not activate their receptors (40). The antagonistic function of the IL-1 family and its own proinflammatory effects confer a wide range of biological activities, including proinflammatory and anti-inflammatory activities (42). The receptor structure contains one or three extracellular immunoglobulin structural domains and a transmembrane structural domain (42). Apart from type 2 IL-1 receptor (IL-1R2), other receptors share a conserved intracellular Toll/IL-1 receptor signaling domain, which is the structural basis for the IL-1 response to immunology (43). Therefore, the regulation of these signals is critical for the regulation of the immune system. In summary, the IL-1 family is a powerful toolbox.
IL-1
IL-1α and IL-1β were the first cytokines in the IL-1 family to be discovered (44). The expression of iCAF marker genes [IL-1α, IL-6, leukemia inhibitory factor, CXC motif chemokine ligand 1 (CXCL1) and granulocyte colony-stimulating factor 3] were significantly upregulated after IL-α and IL-1β induction in PSCs, whereas the expression of myCAF marker genes (actin α2, smooth muscle and connective tissue growth factor) were reduced (32). In particular, the mature form of IL-α enhances crosstalk between CAFs and PDAC cells, resulting in a chronic inflammatory tumor environment, characterized by the expression of the inflammatory factors IL-6 and CXCL8, but no downstream mechanism has been reported (32,45). Schwann cells enhance the proliferation and migration of PDAC cells by promoting the switch of CAFs to malignant subtypes of iCAFs via IL-1α (46). These preclinical studies have encouraged early phase I clinical trials. The maximum established tolerated dose of XB2001 (an IL-1α antagonist), in combination with irinotecan liposome injection (trade name ‘ONIVYDE’) plus 5-FU/LV (+ folinic acid), is being implemented in a PDAC clinical trial (clinical trial NCT04825288). IL-1β promotes immunosuppression by regulating PSC activation and the secretory phenotype in the PanIN microenvironment (47). Inflammatory loops are induced between tumor cells and IL-1β-expressing macrophages, a subset of macrophages elicited by synergy between prostaglandin E2 (PGE2) and tumor necrosis factor (TNF) (48). The PGE2-IL-1β axis may enable preventive or therapeutic strategies to reprogram immune dynamics in pancreatic cancer (48). However, IL-1β is associated with poor outcomes after neoadjuvant therapy of PDAC (49). IL-1 is essential for the formation of both iCAFs and myCAFs but it does not serve as a successful target for specific inhibition of mesothelial cell transition to apCAFs (49). Simultaneously, IL-1 inhibits the expression of MHC II and APC-related genes in CAFs, while IL-1R2 receptors, which are highly expressed by regulatory T cells (Tregs) in the TME, decoy IL-1 to block IL-1 CAF signaling and enhance the ability to deliver APCs, thereby increasing the number of Tregs infiltrating tumors (50). IL-1Ra binds to and blocks proinflammatory IL-1α and IL-1β (42). The IL-1Ra protein signature significantly distinguishes benign pancreatic tissue from PDAC (51). Given the wealth of preclinical studies of IL-1 signaling in iCAFs, anakinra, a human recombinant IL-1Ra, is currently being clinically evaluated in combination with standard chemotherapy (clinical trial NCT02550327).
Interleukin-18 (IL-18)
IL-18 is mainly secreted by macrophages, dendritic cells and epithelial cells, and can stimulate a variety of cell types and has numerous biological functions (52). IL-18 exists in an inactive form within cells (53). Structurally, IL-18-like IL-1β is an important effector molecule downstream of the NLRP3 and NLRP1 inflammasomes (53). IL-18 mediates the MyD88-NFκΒ signaling pathway in combination with its heterodimeric receptor (IL-18Rα/βR), is activated by the rapid release of caspase-1 excised precursor peptides from the inflammasome during inflammation and is considered a proinflammatory cytokine (53,54). Preliminary studies have revealed that IL-18 plays an instrumental role in the development of the pancreas from the acute to the chronic disease stage, while activating the PSC to promote fibrosis in the pancreas (55,56). Concurrently, elevated levels of IL-1β and IL-18 proteins in chronic pancreatitis (CP) have been attributed to the direct involvement of the NLRP3 inflammasome in PSC activation both in vivo and in vitro (57,58). The subsequent promotion of pancreatic fibrosis is mediated by pathogen-associated molecular patterns (57,58).
In the search for specific cytokines in the TME, the application of scRNA-seq technology to tumor-infiltrating lymphocytes revealed specifically high expression of IL-18 and its receptor (59). High expression of IL-18 in the stroma is associated with poor prognosis in patients (60). One function of IL-18 is to stimulate the production of IFNγ by natural killer (NK) cells and Th1 cells and synergize with IL-12 to enhance cytotoxicity against tumor cells (53,54). Therefore, IL-18 has been used in tumor immunotherapy, however, this approach has failed in phase II clinical trials (61). This raises the question of why IL-18 is ineffective against solid tumors. First, the Cancer Genome Atlas (TCGA) database and tissue microarrays revealed that IL-18 binding protein (IL-18BP) is more widely distributed than IL-18R in various solid tumor tissues and sera. Second, IL-18BP binds IL-18 with high affinity (1.1 pM), prevents it from binding to the receptor and reduces the IFNγ-secreting activity of IL-18 (62,63). Thus, IL-18BP is a major barrier to IL-18 immunotherapy (62). The next question was whether bypassing IL-18BP would exert an antitumor effect. A mutant decoy-resistant IL-18 (DR-18) variant, which combines with IL-18Rα but not with IL-18BP, has been screened by directed evolutionary means for its full mobilization of a variety of immune cells, including CD8+ T cells, NK cells and intratumor cell-like T cells (60). DR-18 exhibits good antitumor activity, and its efficacy alone is superior to that of anti-PD-1 monotherapy (60). In 2022, Simcha Therapeutics (60) commenced a phase I clinical trial on the safety and bioactivity of ST-067 (DR-18) in multiple solid tumor types based on this basic study. In a second study, IL-37 was shown to have high homology with IL-18 (61). IL-37 binds to IL-18Rα after maturation via caspase-1 cleavage but binds less efficiently than does IL-18 (62). However, IL-37 can act as a binder for IL-18BP and antagonize the binding of IL-18/IL-18Rα to IL-18Rβ, whereas the low-affinity dimer IL-18/IL-18Rα needs to bind to IL-18Rβ for cell signaling, thus inhibiting IL-18 innate immunity and reducing IFNγ expression (61,64). IL-18 is a promising alternative therapeutic target for pancreatitis, PanIN and pancreatic cancer.
Interleukin-33 (IL-33)
IL-33 induces a type 2 immune response (65). Following sustained stimulation by damage signals, IL-33 is rapidly released from the nucleus of cells distributed in the pretumor period into the extracellular space, where it binds to the surface receptor tumorigenicity 2 (ST2) of innate lymphoid cells 2 (ILC2s) to activate the Th2 immune response (66). PSCs were the predominant cell type in CP fibroblastic tissue (43.8% of the total cells), and human PSCs were further characterized upon activation with an immune-activated genome enriched for IL-33 and IL-11 (20). IL-33-mediated activation of ILC2s directly contributes to PSCs proliferation and activation, ultimately leading to CP fibrosis (25). The IL-33-ST2 signaling axis in cancer cells promotes type 2 immune reactions, induces an immunosuppressive microenvironment and accelerates PDAC progression in the context of chronic inflammation accompanied by fibrosis or intratumoral fungi (67,68). Conversely, genetic deletion of IL-33, ST2 or MMP-9 has been shown to significantly block tumor metastasis in mouse and human fibroblast-rich PDAC (69). Moreover, blockade of PD-1 signaling by IL-33-activated ILC2s has a direct antitumor effect, and modulation of the IL-33-PD-1 axis demonstrates a more favorable prognosis in individuals with PDAC tumors (70). Furthermore, a recent study demonstrated that IL-6+ CAFs secrete IL-33 to induce CXCL3 secretion via macrophages. Notably, CXCL3 engages with its receptor CXCR2 on CAFs to convert them into myCAFs, and this IL-33-CXCL3-CXCR2 loop eventually promotes PDAC metastasis (71).
IL-33, the most responsive chromatin-activated tumor-forming effector, cooperates with mutant KRAS to generate a specific transcriptional program for tumor formation that contributes to epigenetic remodeling of early neoplasia and tumor transformation (72). Despite being expressed in only a fraction of KRAS-mutant pancreatic epithelial cells, IL-33 resulted in marked changes in the premalignant pancreatic cellular state and subsequently prevented the transition of the plasticized progenitor-like state to the PanIN populations (73). Among the downstream targets of oncogenic genes, IL-33 is the most altered cytokine (72,73). In conclusion, IL-33 is a vital target allele in damaged pancreatic tissue, acting as an immunotherapeutic enhancer in the PDAC stage while exerting opposite proinflammatory and fibrotic effects in precancerous tissue (72). IL-33 directly facilitates TGFβ-triggered differentiation of immunosuppressive Treg cells and IFNγ production in other solid tumors, thereby reducing immunotherapy efficacy (74). IL-33 is specifically elevated in human PDACs and is positively associated with tumor immunity in human patients with PDAC (71). However, the combined clinical effects of IL-33 require further investigation.
IL-6 family
The IL-6 family comprises cytokines with a similar structure and signaling mechanism as the subunit glycoprotein 130 kDa (GP130) (75). GP130 dimers are recruited through conjugation to the non-signaling α receptor (IL-6R) to form an IL-6/IL-6R/GP130 hexamer that initiates the intracellular signaling chain (75). During PanIN-PDAC progression, resident PSCs in the ECM secrete large amounts of inflammatory cytokines IL-6 and IL-11 (76). Bazedoxifene has been shown to have effective antitumor effects on pancreatic cancer via inhibition of the IL-6 (GP130/STAT3) pathway (clinical trial NCT04812808). Specifically, the JAK/STAT pathway activates JAK proteins in cells and phosphorylates the transcription factor STAT3, which translocates to the nucleus to regulate target gene expression (77). Although JAK/STAT signaling is the primary pathway for downstream activation of the IL-6 family of cytokines, the mitogen-activated protein kinase (MAPK) pathway can also undergo activation (78). IL-6 collaborates with the oncogene KRAS to activate the reactive oxygen species detoxification program downstream of the MAPK/ERK signaling pathway (79). For instance, a synergistic therapeutic combination with the CAF inhibitor nintedanib enhances PDAC chimeric antigen receptor-NK-mediated cytotoxicity via a reduction in CAF-released IL-6 (80). Clinically high levels of IL-6 in patients with PDAC are typically associated with large tumor volumes and distant metastases (81). Moreover, patients with unresectable and systemic metastatic PDAC have high IL-6 production (82). Thus, high levels of IL-6 indicate an accurate prognosis for adverse outcomes (82). In addition, the serum marker IL-6 is superior to C-reactive protein, carcinoembryonic antigen and carbohydrate antigen 19-9 for the diagnosis and prognosis of patients with PDAC (83). These phenomena clearly demonstrate that IL-6 is the intrinsic mechanism of PDAC development, recapitulating most of the hallmarks of cancer. However, it should be noted that IL-6 can be secreted from other compartments, such as immune cells and can affect PDAC growth and progression (31). Therefore, systemic depletion of IL-6 affects CAF-independent pathways in PDAC.
The IL-6 series of cytokines and their downstream mediators contributes to the initiation of PDAC. IL-6 release by aPSCs leads to the conversion of immature myeloid cells into myeloid-derived suppressor cells (MDSCs), which then inhibit the action of CD4+ T cells, CD8+ T cells and NK cells, thus forming an immunosuppressive environment for PDAC (22). Moreover, Nagathihalli et al (84) demonstrated that IL-6 secreted by PSCs causes marked fibrosis and catheterization of pancreatic tissue and that compromising the IL-6/Stat3 axis inhibits PanIN carcinogenesis. Several clinical trials targeting the IL-6/JAK/STAT3 pathway have been conducted (77). The chimeric mouse-human antibody siltuximab is the most widely developed anti-IL-6 clinical drug, but the highly heterogeneous nature of KRAS-mutated PDAC tumors and their autocrine IL-6 status may result in the clinical ineffectiveness of siltuximab against these tumors (85). Tocilizumab is an antibody against IL-6R that inhibits IL-6 signaling to significantly reduce the growth and recurrence of primary cancer (86). An early phase clinical trial on the safety and efficacy of tocilizumab in patients with PDAC is ongoing (clinical trial NCT02767557). Ruxolitinib is a clinically useful oral inhibitor of JAK and has been shown to inhibit tumor growth in several preclinical studies in mouse models of pancreatic cancer (87). However, ruxolitinib did not improve the survival rate of patients with advanced/metastatic pancreatic disease (87). The use of STAT3 as a possible inhibitor is challenging owing to its lack of enzyme activity (88). Nevertheless, a synthetic STAT3 inhibitor compound, AZD9150, is now in a phase 2 clinical trial (NCT02983578), but its clinical outcome is not yet known. Currently, IL-6 is a critical player in all stages of PDAC and is a potential therapeutic target.
IL-10 family
The IL-10 family is classified based on structural similarity, common receptor use and downstream signaling. This family consists of nine members including IL-10 and IL-20 subfamily members IL-19, IL-20, IL-22, IL-24, IL-26, IL-28A, IL-28B and IL-29, which are categorized as type III interferons (IFNs) (89,90). All IL-10 family members preferentially bind to Janus kinase 1 and tyrosine kinase 2, and mediate differentiation through the downstream signaling JAK/STAT transcription factor pathway (89). IL-10 and IL-22 are the most widely studied family members in the pancreas (91,92).
IL-10
IL-10 cytokines are produced primarily by macrophages and T cell subsets (Th2 and Treg) in immune cells, and have been linked to the pathogenesis and development of autoimmune diseases and cancers (93). IL-10 predominates in inflammatory activity and wound recovery, releasing regenerative anti-inflammatory factors that suppress inflammation and promote favorable matrix remodeling and repair to alleviate organ impairment (94). IL-10 [source bone marrow, (BM)] knockout transplanted mice compared with wild-type mice exhibit substantially more fibrosis, inflammatory cell infiltration and BM-derived myofibroblasts, which emphasizes the crucial role of IL-10 in pancreatitis (95). However, whether IL-10 can be recovered by aPSCs requires further investigation (95).
The effect of IL-10 multipotency as an immunotherapeutic strategy has been investigated. A cetuximab-based IL-10 fusion protein exhibits powerful antitumor activity by blocking dendritic cell-mediated apoptosis of tumor-infiltrating CD8+ T cells (96). Conversely, IL-10 levels in the blood of patients with PDAC were 35-fold greater than the systemic concentrations, which may be associated with NK cells immune escape, a cytotoxic CD16hiCD57hi NK phenotype and impeded expression of cytotoxic T lymphocytes and IFNγ (91). PEGylated IL-10 treatment restored tumor-specific CD8+ T cell reactions and diminished tumor proliferation (97). However, a phase 3 trial of single pegilodecakin (PEGylated human IL-10) tumor immunotherapy revealed no evidence of improved survival in patients with PDAC (clinical trial NCT02923921). This may be the reason why IL-10 research in the field of pancreatic fibrosis has ended abruptly in recent years. However, it has also been shown that genetically engineered macrophages producing an IL-10-blocking antibody (αIL-10) can increase cancer cell death in human gastrointestinal tumors (98). A trial with a combination treatment is anticipated based on successful preclinical outcomes.
Interleukin-22 (IL-22)
IL-22 is mainly derived from CD4+ T cells (Th1, Th17 and Th22), NK cells and innate lymphoid cells, and plays an instrumental role in host defense, tissue repair and gastrointestinal tumor formation (99). The pancreas is one of the major targets of IL-22, as IL-22R is stably expressed in pancreatic epithelial cells and fibroblasts (100). IL-22 activates STAT-3 in coordination with the inflammatory response and affects the production of Reg survival genes, vascular endothelial growth factor (VEGF) and antiapoptotic protein Bcl-X (101,102). It also promotes repair and renewal signaling in impaired pancreatic vesicle cells, effectively protecting mice from induced acute pancreatitis or CP (103). However, IL-22 has not yet been investigated clinically (103,104). Conversely, with sustained increases in IL-22 expression and signaling imbalances, this protective effect can be easily manipulated into carcinogenesis (92). CD4+ T cell-derived IL-22 amplifies liver metastasis by promoting angiogenesis (105). The absence of PanIN and PDAC in IL-22 knockout mice confirms the importance of IL-22 in ductal differentiation (100). Moreover, STAT inhibitors prevent IL-22-induced ductal hyperplasia of the alveoli, the expression of stem-associated transcription factors and tumor regeneration during EMT (100). The aryl hydrocarbon receptor (AHR) in cigarette smoke dramatically increased pancreatic fibrosis in a mouse pancreatitis model (106). AHR is an area for the convergence of numerous cellular and environmental pathways, and the gut microbiota regulates IL-22 transcription and oncogenic IL-10 expression through AHR via tryptophan metabolites (107). Homologous IL-20RB includes immune-related genes in a prognostic model of pancreatic cancer, and IL-20 antagonists inhibit PD-L1 expression and prolong survival in PDAC (108,109). The IL-10 family is still a viable target in clinical trials, but it is possible that the collapse of clinical trials on IL-10 has put research projects in this category in jeopardy.
Interleukin-17 (IL-17) family
The IL-17 family comprises six subtypes of multifunctional cytokines (IL-17A to IL-17F) that perform diverse functions despite their amino acid sequence homology (110). Th17 cells preferentially produce IL-17A, IL-17F, IL-21 and IL-2 (110). IL-17A is active in epithelial cells and fibroblasts and is a characteristic hallmark of the proinflammatory cytokine Th17 cells, which participate in autoimmune, inflammatory and tumor pathogenesis, whereas IL-17F is mainly involved in mucosal host defense mechanisms (110,111). IL-17A signal-mediated tissue remodeling MMP may lead to ECM destruction and tissue lesions and is also capable of downregulating tissue inhibitors of metalloproteinases (111). IL-1β, TNFα, IL-6, IL-10 and IL-2 are typically induced following IL-17A stimulation of macrophages (112). The inflammatory and cancer paracrine factor regenerating islet-derived 3β stimulates IL-17RA, promotes cell proliferation, reduces susceptibility to apoptosis through coupling of the gp130-JAK2-pSTAT3 signaling pathway and initiates PanIN onset (113). IL-17A binds to IL-17RA/RC complexes to secrete cytokines to recruit neutrophils, which inhibits CD8+ T cells in the PDAC TME (114). Intestinal IL-17-IL-17RA signaling regulates microbes to promote barrier immunity and drive distant pancreatic tumor proliferation (115). Subsequently, IL-17A participates in the evolution of PanIN and promotes PDAC via the induction of iCAFs, which results in poor prognosis for patients (116,117). Ablation of IL-17A limits the immunosuppression of T cells to alter cytokine release by tumor fibroblasts (118). Thus, the inhibition of IL-17A may be a novel combination treatment (118). To assess the role of IL-17 therapy in tumor initiation and early tumor progression, an anti-IL-17 antibody was injected weekly to ensure inhibition of the IL-17A/IL-17RA axis throughout tumor development (119). However, there was no prolongation of overall survival in this curative model, which indicates that IL-17A may have a variable effect on PanIN and PDAC progression (119). Blocking the IL-17A/RA axis with antibodies alone is ineffective in preventing the development and progression of pancreatic cancer (119). Therefore, IL-17A is not suitable as primary monotherapy for pancreatic cancer in clinical practice. IL-17A is also a double-edged sword in the immune system (120). During tumorigenesis, IL-17A recruits MDSCs to suppress antitumor immunity, but it also activates STAT3 to induce IL-6 to promote tumor growth in vivo and upregulates oncogenes to promote tumor survival and angiogenesis (121). Additional studies are required to clarify the role of IL-17A in the progression of PanIN and PDAC.
Summary and overall conclusions
In conclusion, the interaction of PDAC with its TME components is becoming increasingly important. However, long-term fibrosis evolution and tumor immune cell imbalance result in an egregious TME (11). Currently, depletion of the ECM to remove the fibrous barrier has been proposed, but this approach has been found to be ineffective for PDAC treatment (122). Therefore, ameliorating the accumulation of the ECM in PDAC and targeting the ECM for immune cell modulation may have unexpected outcomes. Despite well-developed theories related to fibrosis or immune modulation in the pancreas, several questions remain to be addressed to translate mechanistic insights into new and effective therapeutic interventions. First, even with comprehensive combined systematic screening of fibrosis and immunomodulation with PSC and interleukins for PanIN and PDAC, there are limitations in extracting only gross information from the available study data. Utilizing scRNA-seq data for in-depth exploration of the mechanisms of PSC to CAF conversion remains a priority. Second, CAF-forming fibrosis interacts with immune cells in various ways, with the mode of action depending in part on the type of CAF under investigation. As markers for different CAF subgroups are not common to all CAF regulatory factors, personalized regimens are needed for the treatment of different stages of PDAC. Moreover, interleukin families affect the development of PDAC to varying extents, but a standard for interleukins to cause PDAC fibrosis directly by inducing PSC activation is lacking. Consequently, IL-1, IL-6, IL-10 and IL-17A are promising targets for the treatment of PDAC. Targeting the PanIN-PDAC process for immunity and fibrosis treatment is complex and requires further rigorous testing and validation of targets. The complex interactions between these elements and the development of specific therapeutic strategies require further elucidation.
Future research directions must focus on the stroma and immune cells in the TME of PDAC, both of which play critical roles in establishing structural and functional barriers to protect PDAC from external attack. Most studies investigating the interactions between PDAC and its ecotope have relied on traditional two-dimensional cell cultures, which may not accurately represent the complex three-dimensional microenvironment of PDAC in vivo and could be utilized in coculture modeling or organoid culture (123). Organoid technology has revolutionized in the field of precision medicine for treating PDAC (124). Concurrently, the emergence of high-resolution scRNA-seq technology provides an in-depth characterization of malignant cell types and increases the understanding of the heterogeneity and plasticity of PDAC in response to homeostatic and therapeutic perturbations (125).
In conclusion, pancreatic fibrosis and immunomodulation have been identified as key drivers in the development of PDAC and are major impediments to the efficacy of therapeutics for PDAC. With increasing research on PDAC-related signaling, it is anticipated that combination therapy regimens will be more successful, providing a more authoritative basis for drug development and clinical treatment, and thus improving the survival of patients with PDAC.
Acknowledgements
Not applicable.
Funding
The present study was supported by The National Natural Science Foundation of China (grant no. 82372686) and The Guangzhou Medical University Research Capacity Enhancement Program (grant no. 2024SRP192).
Availability of data and materials
The survival data of patients with pancreatic cancer generated in the present study may be found in the gene expression profiling interactive analysis 2 (GEPIA 2) using data from The Cancer Genome Atlas (http://gepia2.cancer-pku.cn/#survival).
Authors' contributions
HL and DL wrote the manuscript; KL acquired the data, YW and GZ analyzed and interpreted the data included in the review. KL, YW and GZ also contributed to the study design. LQ and KX conceived the original idea, corrected and finalized the manuscript and contributed to critical discussion. All authors read and approved the final version of the manuscript and checked and confirmed the authenticity of all the raw data.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Rahib L, Wehner MR, Matrisian LM and Nead KT: Estimated projection of US cancer incidence and death to 2040. JAMA Netw Open. 4:e2147082021. View Article : Google Scholar : PubMed/NCBI | |
|
Huang J, Lok V, Ngai CH, Zhang L, Yuan J, Lao XQ, Ng K, Chong C, Zheng ZJ and Wong MCS: Worldwide burden of, risk factors for, and trends in pancreatic cancer. Gastroenterology. 160:744–754. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Infante-Cossio P, Duran-Romero AJ, Castaño-Seiquer A, Martinez-De-Fuentes R and Pereyra-Rodriguez JJ: Estimated projection of oral cavity and oropharyngeal cancer deaths in Spain to 2044. BMC Oral Health. 22:4442022. View Article : Google Scholar : PubMed/NCBI | |
|
Viale PH: The American cancer society's facts & figures: 2020 Edition. J Adv Pract Oncol. 11:135–136. 2020.PubMed/NCBI | |
|
Blackford AL, Canto MI, Klein AP, Hruban RH and Goggins M: Recent trends in the incidence and survival of stage 1A pancreatic cancer: A surveillance, epidemiology, and end results analysis. J Natl Cancer Inst. 112:1162–1169. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Cao K, Xia Y, Yao J, Han X, Lambert L, Zhang T, Tang W, Jin G, Jiang H, Fang X, et al: Large-scale pancreatic cancer detection via non-contrast CT and deep learning. Nat Med. 29:3033–3043. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Zheng R, Zhang S, Zeng H, Wang S, Sun K, Chen R, Li L, Wei W and He J: Cancer incidence and mortality in China, 2016. J Nat Cancer Cent. 2:1–9. 2022. View Article : Google Scholar | |
|
Yachida S, Jones S, Bozic I, Antal T, Leary R, Fu B, Kamiyama M, Hruban RH, Eshleman JR, Nowak MA, et al: Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nature. 467:1114–1117. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Werba G, Weissinger D, Kawaler EA, Zhao E, Kalfakakou D, Dhara S, Wang L, Lim HB, Oh G, Jing X, et al: Single-cell RNA sequencing reveals the effects of chemotherapy on human pancreatic adenocarcinoma and its tumor microenvironment. Nat Commun. 14:7972023. View Article : Google Scholar : PubMed/NCBI | |
|
Du W, Xia X, Hu F and Yu J: Extracellular matrix remodeling in the tumor immunity. Front Immunol. 14:13406342024. View Article : Google Scholar : PubMed/NCBI | |
|
Chen K, Wang Q, Li M, Guo H, Liu W, Wang F, Tian X and Yang Y: Single-cell RNA-seq reveals dynamic change in tumor microenvironment during pancreatic ductal adenocarcinoma malignant progression. EBioMedicine. 66:1033152021. View Article : Google Scholar : PubMed/NCBI | |
|
Storz P and Crawford H: Carcinogenesis of pancreatic ductal adenocarcinoma. Gastroenterology. 158:2072–2081. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Wang S, Li Y, Xing C, Ding C, Zhang H, Chen L, You L, Dai M and Zhao Y: Tumor microenvironment in chemoresistance, metastasis and immunotherapy of pancreatic cancer. Am J Cancer Res. 10:1937–1953. 2020.PubMed/NCBI | |
|
Shi C, Washington MK, Chaturvedi R, Drosos Y, Revetta FL, Weaver CJ, Buzhardt E, Yull FE, Blackwell TS, Sosa-Pineda B, et al: Fibrogenesis in pancreatic cancer is a dynamic process regulated by macrophage-stellate cell interaction. Lab Invest. 94:409–421. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Briukhovetska D, Dörr J, Endres S, Libby P, Dinarello CA and Kobold S: Interleukins in cancer: From biology to therapy. Nature Rev Cancer. 21:481–499. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Kartsonaki C, Pang Y, Millwood I, Yang L, Guo Y, Walters R, Lv J, Hill M, Yu C, Chen Y, et al: Circulating proteins and risk of pancreatic cancer: A case-subcohort study among Chinese adults. Int J Epidemiol. 51:817–829. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Nie YJ, Wu SH, Xuan YH and Yan G: Role of IL-17 family cytokines in the progression of IPF from inflammation to fibrosis. Mil Med Res. 9:212022.PubMed/NCBI | |
|
Apte MV, Haber PS, Applegate TL, Norton ID, McCaughan GW, Korsten MA, Pirola RC and Wilson JS: Periacinar stellate shaped cells in rat pancreas: identification, isolation, and culture. Gut. 43:128–133. 1998. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou Y, Wang H, Zhou J, Qiu S, Cai T, Li H, Shen Z, Hu Y, Ding B, Luo M, et al: Vitamin A and its multi-effects on pancreas: Recent advances and prospects. Front Endocrinol (Lausanne). 12:6209412021. View Article : Google Scholar : PubMed/NCBI | |
|
Baron M, Veres A, Wolock SL, Faust AL, Gaujoux R, Vetere A, Ryu JH, Wagner BK, Shen-Orr SS, Klein AM, et al: A single-cell transcriptomic map of the human and mouse pancreas reveals inter- and intra-cell population structure. Cell Syst. 3:346–360.e4. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Ikejiri N: The vitamin A-storing cells in the human and rat pancreas. Kurume Med J. 37:67–81. 1990. View Article : Google Scholar : PubMed/NCBI | |
|
Ahmad RS, Eubank TD, Lukomski S and Boone BA: Immune cell modulation of the extracellular matrix contributes to the pathogenesis of pancreatic cancer. Biomolecules. 11:9012021. View Article : Google Scholar : PubMed/NCBI | |
|
Bazzichetto C, Conciatori F, Luchini C, Simionato F, Santoro R, Vaccaro V, Corbo V, Falcone I, Ferretti G, Cognetti F, et al: From genetic alterations to tumor microenvironment: The Ariadne's String in pancreatic cancer. Cells. 9:3092020. View Article : Google Scholar : PubMed/NCBI | |
|
Mews P, Phillips P, Fahmy R, Korsten M, Pirola R, Wilson J and Apte M: Pancreatic stellate cells respond to inflammatory cytokines: Potential role in chronic pancreatitis. Gut. 50:535–541. 2002. View Article : Google Scholar : PubMed/NCBI | |
|
Yang X, Chen J, Wang J, Ma S, Feng W, Wu Z, Guo Y, Zhou H, Mi W, Chen W, et al: Very-low-density lipoprotein receptor-enhanced lipid metabolism in pancreatic stellate cells promotes pancreatic fibrosis. Immunity. 55:1185–1199.e8. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Elyada E, Bolisetty M, Laise P, Flynn WF, Courtois ET, Burkhart RA, Teinor JA, Belleau P, Biffi G, Lucito MS, et al: Cross-species single-cell analysis of pancreatic ductal adenocarcinoma reveals antigen-presenting cancer-associated fibroblasts. Cancer Discov. 9:1102–1123. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Öhlund D, Handly-Santana A, Biffi G, Elyada E, Almeida AS, Ponz-Sarvise M, Corbo V, Oni TE, Hearn SA, Lee EJ, et al: Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J Exp Med. 214:579–596. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Schnittert J, Bansal R and Prakash J: Targeting pancreatic stellate cells in cancer. Trends Cancer. 5:128–142. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, McAndrews KM and Kalluri R: Clinical and therapeutic relevance of cancer-associated fibroblasts. Nat Rev Clin Oncol. 18:792–804. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, Kim J, Yang S, Wang H, Wu CJ, Sugimoto H, LeBleu VS and Kalluri R: Type I collagen deletion in αSMA+ myofibroblasts augments immune suppression and accelerates progression of pancreatic cancer. Cancer Cell. 39:548–565.e6. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
van Duijneveldt G, Griffin MDW and Putoczki TL: Emerging roles for the IL-6 family of cytokines in pancreatic cancer. Clin Sci (Lond). 134:2091–2115. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Biffi G, Oni TE, Spielman B, Hao Y, Elyada E, Park Y, Preall J and Tuveson DA: IL1-induced JAK/STAT signaling is antagonized by TGFβ to shape CAF heterogeneity in pancreatic ductal adenocarcinoma. Cancer Discov. 9:282–301. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Huber M, Brehm CU, Gress TM, Buchholz M, Alashkar Alhamwe B, von Strandmann EP, Slater EP, Bartsch JW, Bauer C and Lauth M: The immune microenvironment in pancreatic cancer. Int J Mol Sci. 21:73072020. View Article : Google Scholar : PubMed/NCBI | |
|
Miyai Y, Esaki N, Takahashi M and Enomoto A: Cancer-associated fibroblasts that restrain cancer progression: Hypotheses and perspectives. Cancer Sci. 111:1047–1057. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Opitz F, Haeberle L, Daum A and Esposito I: Tumor microenvironment in pancreatic intraepithelial neoplasia. Cancers (Basel). 13:61882021. View Article : Google Scholar : PubMed/NCBI | |
|
Carpenter ES, Elhossiny AM, Kadiyala P, Li J, McGue J, Griffith BD, Zhang Y, Edwards J, Nelson S, Lima F, et al: Analysis of donor pancreata defines the transcriptomic signature and microenvironment of early neoplastic lesions. Cancer Discov. 13:1324–1345. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Xue J, Sharma V, Hsieh MH, Chawla A, Murali R, Pandol SJ and Habtezion A: Alternatively activated macrophages promote pancreatic fibrosis in chronic pancreatitis. Nat Commun. 6:71582015. View Article : Google Scholar : PubMed/NCBI | |
|
Wu J, Zhang L, Shi J, He R, Yang W, Habtezion A, Niu N, Lu P and Xue J: Macrophage phenotypic switch orchestrates the inflammation and repair/regeneration following acute pancreatitis injury. EBioMedicine. 58:1029202020. View Article : Google Scholar : PubMed/NCBI | |
|
Hingorani SR: Epithelial and stromal co-evolution and complicity in pancreatic cancer. Nat Rev Cancer. 23:57–77. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Garlanda C and Mantovani A: Interleukin-1 in tumor progression, therapy, and prevention. Cancer Cell. 39:1023–1027. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Boersma B, Jiskoot W, Lowe P and Bourquin C: The interleukin-1 cytokine family members: Role in cancer pathogenesis and potential therapeutic applications in cancer immunotherapy. Cytokine Growth Factor Rev. 62:1–14. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Dinarello CA, Simon A and van der Meer JWM: Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 11:633–652. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Dinarello C: Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol Rev. 281:8–27. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Narros-Fernández P, Chomanahalli Basavarajappa S and Walsh PT: Interleukin-1 family cytokines at the crossroads of microbiome regulation in barrier health and disease. FEBS J. 291:1849–1869. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Tomimatsu S, Ichikura T and Mochizuki H: Significant correlation between expression of interleukin-1alpha and liver metastasis in gastric carcinoma. Cancer. 91:1272–1276. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Xue M, Zhu Y, Jiang Y, Han L, Shi M, Su R, Wang L, Xiong C, Wang C, Wang T, et al: Schwann cells regulate tumor cells and cancer-associated fibroblasts in the pancreatic ductal adenocarcinoma microenvironment. Nat Commun. 14:46002023. View Article : Google Scholar : PubMed/NCBI | |
|
Das S, Shapiro B, Vucic E, Vogt S and Bar-Sagi D: Tumor cell-derived IL1β promotes desmoplasia and immune suppression in pancreatic cancer. Cancer Res. 80:1088–1101. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Caronni N, La Terza F, Vittoria FM, Barbiera G, Mezzanzanica L, Cuzzola V, Barresi S, Pellegatta M, Canevazzi P, Dunsmore G, et al: IL-1β+ macrophages fuel pathogenic inflammation in pancreatic cancer. Nature. 623:415–422. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Herremans KD, Szymkiewicz DD, Riner AN, Bohan RP, Tushoski GW, Davidson AM, Lou X, Leong MC, Dean BD, Gerber M, et al: The interleukin-1 axis and the tumor immune microenvironment in pancreatic ductal adenocarcinoma. Neoplasia. 28:1007892022. View Article : Google Scholar : PubMed/NCBI | |
|
Chen L, Huang H, Zheng X, Li Y, Chen J, Tan B, Liu Y, Sun R, Xu B, Yang M, et al: IL1R2 increases regulatory T cell population in the tumor microenvironment by enhancing MHC-II expression on cancer-associated fibroblasts. J Immunother Cancer. 10:e0045852022. View Article : Google Scholar | |
|
Underwood PW, Gerber MN, Nguyen K, Delitto D, Han S, Thomas RM, Forsmark CE, Trevino JG, Gooding WE and Hughes SJ: Protein signatures and tissue diagnosis of pancreatic cancer. J Am Coll Surg. 230:26–36.e1. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Waldmann T: Cytokines in cancer immunotherapy. Cold Spring Hard Perspect Biol. 10:a0284722018. View Article : Google Scholar : PubMed/NCBI | |
|
Yasuda K, Nakanishi K and Tsutsui H: Interleukin-18 in health and disease. Int J Mol Sci. 20:6492019. View Article : Google Scholar : PubMed/NCBI | |
|
Kaplanski G: Interleukin-18: Biological properties and role in disease pathogenesis. Immunol Rev. 281:138–153. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Schneider A, Haas SL, Hildenbrand R, Siegmund S, Reinhard I, Nakovics H, Singer MV and Feick P: Enhanced expression of interleukin-18 in serum and pancreas of patients with chronic pancreatitis. World J Gastroentero. 12:6507–6514. 2006. View Article : Google Scholar | |
|
Manohar M, Verma AK, Venkateshaiah SU and Mishra A: Role of eosinophils in the initiation and progression of pancreatitis pathogenesis. Am J Physiol Gastrointest Liver Physiol. 314:G211–G222. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Li CX, Cui LH, Zhang LQ, Yang L, Zhuo YZ, Cui NQ and Zhang SK: Role of NLR family pyrin domain-containing 3 inflammasome in the activation of pancreatic stellate cells. Exp Cell Res. 404:1126342021. View Article : Google Scholar : PubMed/NCBI | |
|
Liu Y, Xu X, Lei W, Hou Y, Zhang Y, Tang R, Yang Z, Tian Y, Zhu Y, Wang C, et al: The NLRP3 inflammasome in fibrosis and aging: The known unknowns. Ageing Res Rev. 79:1016382022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou T, Damsky W, Weizman OE, McGeary MK, Hartmann KP, Rosen CE, Fischer S, Jackson R, Flavell RA, Wang J, et al: IL-18BP is a secreted immune checkpoint and barrier to IL-18 immunotherapy. Nature. 583:609–614. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Ahmed A, Klotz R, Köhler S, Giese N, Hackert T, Springfeld C, Jäger D and Halama N: Immune features of the peritumoral stroma in pancreatic ductal adenocarcinoma. Front Immunol. 13:9474072022. View Article : Google Scholar : PubMed/NCBI | |
|
Tarhini AA, Millward M, Mainwaring P, Kefford R, Logan T, Pavlick A, Kathman SJ, Laubscher KH, Dar MM and Kirkwood JM: A phase 2, randomized study of SB-485232, rhIL-18, in patients with previously untreated metastatic melanoma. Cancer. 115:859–868. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Kim SH, Eisenstein M, Reznikov L, Fantuzzi G, Novick D, Rubinstein M and Dinarello CA: Structural requirements of six naturally occurring isoforms of the IL-18 binding protein to inhibit IL-18. Proc Natl Acad Sci USA. 97:1190–1195. 2000. View Article : Google Scholar : PubMed/NCBI | |
|
Menachem A, Alteber Z, Cojocaru G, Fridman Kfir T, Blat D, Leiderman O, Galperin M, Sever L, Cohen N, Cohen K, et al: Unleashing natural IL18 activity using an anti-IL18BP blocker induces potent immune stimulation and antitumor effects. Cancer Immunol Res. 12:687–703. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Yang Y, Zhang ZX, Lian D, Haig A, Bhattacharjee R and Jevnikar AM: IL-37 inhibits IL-18-induced tubular epithelial cell expression of pro-inflammatory cytokines and renal ischemia-reperfusion injury. Kidney Int. 87:396–408. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Liew FY, Girard JP and Turnquist HR: Interleukin-33 in health and disease. Nat Rev Immunol. 16:676–689. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Larsen KM, Minaya MK, Vaish V and Peña MMO: The role of IL-33/ST2 pathway in tumorigenesis. Int J Mol Sci. 19:26762018. View Article : Google Scholar : PubMed/NCBI | |
|
Park JH, Ameri AH, Dempsey KE, Conrad DN, Kem M, Mino-Kenudson M and Demehri S: Nuclear IL-33/SMAD signaling axis promotes cancer development in chronic inflammation. EMBO J. 40:e1061512021. View Article : Google Scholar : PubMed/NCBI | |
|
Alam A, Levanduski E, Denz P, Villavicencio HS, Bhatta M, Alhorebi L, Zhang Y, Gomez EC, Morreale B, Senchanthisai S, et al: Fungal mycobiome drives IL-33 secretion and type 2 immunity in pancreatic cancer. Cancer Cell. 40:153–167.e11. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Andersson P, Yang Y, Hosaka K, Zhang Y, Fischer C, Braun H, Liu S, Yu G, Liu S, Beyaert R, et al: Molecular mechanisms of IL-33-mediated stromal interactions in cancer metastasis. JCI insight. 3:e1223752018. View Article : Google Scholar : PubMed/NCBI | |
|
Moral JA, Leung J, Rojas LA, Ruan J, Zhao J, Sethna Z, Ramnarain A, Gasmi B, Gururajan M, Redmond D, et al: ILC2s amplify PD-1 blockade by activating tissue-specific cancer immunity. Nature. 579:130–135. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Sun X, He X, Zhang Y, Hosaka K, Andersson P, Wu J, Wu J, Jing X, Du Q, Hui X, et al: Inflammatory cell-derived CXCL3 promotes pancreatic cancer metastasis through a novel myofibroblast-hijacked cancer escape mechanism. Gut. 71:129–147. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Alonso-Curbelo D, Ho YJ, Burdziak C, Maag JLV, Morris JP IV, Chandwani R, Chen HA, Tsanov KM, Barriga FM, Luan W, et al: A gene-environment-induced epigenetic program initiates tumorigenesis. Nature. 590:642–648. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Burdziak C, Alonso-Curbelo D, Walle T, Reyes J, Barriga FM, Haviv D, Xie Y, Zhao Z, Zhao CJ, Chen HA, et al: Epigenetic plasticity cooperates with cell-cell interactions to direct pancreatic tumorigenesis. Science. 380:eadd53272023. View Article : Google Scholar : PubMed/NCBI | |
|
Hatzioannou A, Banos A, Sakelaropoulos T, Fedonidis C, Vidali MS, Köhne M, Händler K, Boon L, Henriques A, Koliaraki V, et al: An intrinsic role of IL-33 in Treg cell-mediated tumor immunoevasion. Nat Immunol. 21:75–85. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Martínez-Pérez C, Kay C, Meehan J, Gray M, Dixon JM and Turnbull AK: The IL6-like cytokine family: Role and biomarker potential in breast cancer. J Pers Med. 11:10732021. View Article : Google Scholar : PubMed/NCBI | |
|
Shi Y, Gao W, Lytle NK, Huang P, Yuan X, Dann AM, Ridinger-Saison M, DelGiorno KE, Antal CE, Liang G, et al: Targeting LIF-mediated paracrine interaction for pancreatic cancer therapy and monitoring. Nature. 569:131–135. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Johnson DE, O'Keefe RA and Grandis JR: Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 15:234–248. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Müller-Newen G and Schaper F: Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J. 374:1–20. 2003. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang Y, Yan W, Collins MA, Bednar F, Rakshit S, Zetter BR, Stanger BZ, Chung I, Rhim AD and di Magliano MP: Interleukin-6 is required for pancreatic cancer progression by promoting MAPK signaling activation and oxidative stress resistance. Cancer Res. 73:6359–6374. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Lee YE, Go GY, Koh EY, Yoon HN, Seo M, Hong SM, Jeong JH, Kim JC, Cho D, Kim TS, et al: Synergistic therapeutic combination with a CAF inhibitor enhances CAR-NK-mediated cytotoxicity via reduction of CAF-released IL-6. J Immunother Cancer. 11:e0061302023. View Article : Google Scholar : PubMed/NCBI | |
|
Ramsey ML, Talbert E, Ahn D, Bekaii-Saab T, Badi N, Bloomston PM, Conwell DL, Cruz-Monserrate Z, Dillhoff M, Farren MR, et al: Circulating interleukin-6 is associated with disease progression, but not cachexia in pancreatic cancer. Pancreatology. 19:80–87. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Ebrahimi B, Tucker SL, Li D, Abbruzzese JL and Kurzrock R: Cytokines in pancreatic carcinoma: Correlation with phenotypic characteristics and prognosis. Cancer. 101:2727–2736. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Kumari N, Dwarakanath BS, Das A and Bhatt AN: Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol. 37:11553–11572. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Nagathihalli NS, Castellanos JA, VanSaun MN, Dai X, Ambrose M, Guo Q, Xiong Y and Merchant NB: Pancreatic stellate cell secreted IL-6 stimulates STAT3 dependent invasiveness of pancreatic intraepithelial neoplasia and cancer cells. Oncotarget. 7:65982–65992. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Angevin E, Tabernero J, Elez E, Cohen SJ, Bahleda R, van Laethem JL, Ottensmeier C, Lopez-Martin JA, Clive S, Joly F, et al: A phase I/II, multiple-dose, dose-escalation study of siltuximab, an anti-interleukin-6 monoclonal antibody, in patients with advanced solid tumors. Clin Cancer Res. 20:2192–2204. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Goumas FA, Holmer R, Egberts JH, Gontarewicz A, Heneweer C, Geisen U, Hauser C, Mende MM, Legler K, Röcken C, et al: Inhibition of IL-6 signaling significantly reduces primary tumor growth and recurrencies in orthotopic xenograft models of pancreatic cancer. Int J Cancer. 137:1035–1046. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Hurwitz H, Van Cutsem E, Bendell J, Hidalgo M, Li CP, Salvo MG, Macarulla T, Sahai V, Sama A, Greeno E, et al: Ruxolitinib + capecitabine in advanced/metastatic pancreatic cancer after disease progression/intolerance to first-line therapy: JANUS 1 and 2 randomized phase III studies. Invest New Drugs. 36:683–695. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Wong ALA, Hirpara JL, Pervaiz S, Eu JQ, Sethi G and Goh BC: Do STAT3 inhibitors have potential in the future for cancer therapy? Expert Opin Investig Drugs. 26:883–887. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Ouyang W, Rutz S, Crellin NK, Valdez PA and Hymowitz SG: Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol. 29:71–109. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Lazear HM, Schoggins JW and Diamond MS: Shared and distinct functions of type I and type III interferons. Immunity. 50:907–923. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Marcon F, Zuo J, Pearce H, Nicol S, Margielewska-Davies S, Farhat M, Mahon B, Middleton G, Brown R, Roberts KJ and Moss P: NK cells in pancreatic cancer demonstrate impaired cytotoxicity and a regulatory IL-10 phenotype. Oncoimmunology. 9:18454242020. View Article : Google Scholar : PubMed/NCBI | |
|
Xuan X, Tian Z, Zhang M, Zhou J, Gao W, Zhang Y, Zhang Y, Lei B, Ni B, Wu Y and Fan W: Diverse effects of interleukin-22 on pancreatic diseases. Pancreatology. 18:231–237. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Zhao Y, Chen J, Andreatta M, Feng B, Xie YQ, Wenes M, Wang Y, Gao M, Hu X, Romero P, et al: IL-10-expressing CAR T cells resist dysfunction and mediate durable clearance of solid tumors and metastases. Nat Biotechnol. Jan 2–2024.(Epub ahead of print). View Article : Google Scholar | |
|
Ip WKE, Hoshi N, Shouval DS, Snapper S and Medzhitov R: Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science. 356:513–519. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Lin WR, Lim SN, Yen TH and Alison MR: The influence of bone marrow-secreted IL-10 in a mouse model of cerulein-induced pancreatic fibrosis. Biomed Res Int. 2016:46015322016. View Article : Google Scholar : PubMed/NCBI | |
|
Qiao J, Liu Z, Dong C, Luan Y, Zhang A, Moore C, Fu K, Peng J, Wang Y, Ren Z, et al: Targeting tumors with IL-10 prevents dendritic cell-mediated CD8+ T cell apoptosis. Cancer Cell. 35:901–915.e4. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Naing A, Infante JR, Papadopoulos KP, Chan IH, Shen C, Ratti NP, Rojo B, Autio KA, Wong DJ, Patel MR, et al: PEGylated IL-10 (pegilodecakin) induces systemic immune activation, CD8+ T cell invigoration and polyclonal T cell expansion in cancer patients. Cancer Cell. 34:775–791.e3. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Labadie KP, Kreuser SA, Brempelis KJ, Daniel SK, Jiang X, Sullivan KM, Utria AF, Kenerson HL, Kim TS, Crane CA and Pillarisetty VG: Production of an interleukin-10 blocking antibody by genetically engineered macrophages increases cancer cell death in human gastrointestinal tumor slice cultures. Cancer Gene Ther. 30:1227–1233. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Perusina Lanfranca M, Lin Y, Fang J, Zou W and Frankel T: Biological and pathological activities of interleukin-22. J Mol Med (Berl). 94:523–534. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Perusina Lanfranca M, Zhang Y, Girgis A, Kasselman S, Lazarus J, Kryczek I, Delrosario L, Rhim A, Koneva L, Sartor M, et al: Interleukin 22 signaling regulates acinar cell plasticity to promote pancreatic tumor development in mice. Gastroenterology. 158:1417–1432.e11. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Curd LM, Favors SE and Gregg RK: Pro-tumour activity of interleukin-22 in HPAFII human pancreatic cancer cells. Clin Exp Immunol. 168:192–199. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Arshad T, Mansur F, Palek R, Manzoor S and Liska V: A double edged sword role of interleukin-22 in wound healing and tissue regeneration. Front Immunol. 11:21482020. View Article : Google Scholar : PubMed/NCBI | |
|
Feng D, Park O, Radaeva S, Wang H, Yin S, Kong X, Zheng M, Zakhari S, Kolls JK and Gao B: Interleukin-22 ameliorates cerulein-induced pancreatitis in mice by inhibiting the autophagic pathway. Int J Biol Sci. 8:249–257. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Yang H, Cao R, Zhou F, Wang B, Xu Q, Li R, Zhang C and Xu H: The role of Interleukin-22 in severe acute pancreatitis. Mol Med. 30:602024. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang T, Wahib R, Zazara DE, Lücke J, Shiri AM, Kempski J, Zhao L, Agalioti T, Machicote AP, Giannou O, et al: CD4+ T cell-derived IL-22 enhances liver metastasis by promoting angiogenesis. Oncoimmunology. 12:22696342023. View Article : Google Scholar : PubMed/NCBI | |
|
Xue J, Zhao Q, Sharma V, Nguyen LP, Lee YN, Pham KL, Edderkaoui M, Pandol SJ, Park W and Habtezion A: Aryl hydrocarbon receptor ligands in cigarette smoke induce production of interleukin-22 to promote pancreatic fibrosis in models of chronic pancreatitis. Gastroenterology. 151:1206–1217. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, Zecchi R, D'Angelo C, Massi-Benedetti C, Fallarino F, et al: Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity. 39:372–385. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Liu B, Fu T, He P, Du C and Xu K: Construction of a five-gene prognostic model based on immune-related genes for the prediction of survival in pancreatic cancer. Biosci Rep. 41:BSR202043012021. View Article : Google Scholar : PubMed/NCBI | |
|
Lu SW, Pan HC, Hsu YH, Chang KC, Wu LW, Chen WY and Chang MS: IL-20 antagonist suppresses PD-L1 expression and prolongs survival in pancreatic cancer models. Nat Commun. 11:46112020. View Article : Google Scholar : PubMed/NCBI | |
|
McGeachy MJ, Cua DJ and Gaffen SL: The IL-17 family of cytokines in health and disease. Immunity. 50:892–906. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Meehan EV and Wang K: Interleukin-17 family cytokines in metabolic disorders and cancer. Genes (Basel). 13:16432022. View Article : Google Scholar : PubMed/NCBI | |
|
Jovanovic DV, Di Battista JA, Martel-Pelletier J, Jolicoeur FC, He Y, Zhang M, Mineau F and Pelletier JP: IL-17 stimulates the production and expression of proinflammatory cytokines, IL-beta and TNF-alpha, by human macrophages. J Immunol. 160:3513–3521. 1998. View Article : Google Scholar : PubMed/NCBI | |
|
Loncle C, Bonjoch L, Folch-Puy E, Lopez-Millan MB, Lac S, Molejon MI, Chuluyan E, Cordelier P, Dubus P, Lomberk G, et al: IL17 functions through the novel REG3β-JAK2-STAT3 inflammatory pathway to promote the transition from chronic pancreatitis to pancreatic cancer. Cancer Res. 75:4852–4862. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Z, Qiao S, Yang L, Sun M, Li B, Lu A and Li F: Mechanistic insights into the roles of the IL-17/IL-17R families in pancreatic cancer. Int J Mol Sci. 24:135392023. View Article : Google Scholar : PubMed/NCBI | |
|
Chandra V, Li L, Le Roux O, Zhang Y, Howell RM, Rupani DN, Baydogan S, Miller HD, Riquelme E, Petrosino J, et al: Gut epithelial Interleukin-17 receptor A signaling can modulate distant tumors growth through microbial regulation. Cancer Cell. 42:85–100.e6. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Hu F, Guo F, Zhu Y, Zhou Q, Li T, Xiang H and Shang D: IL-17 in pancreatic disease: Pathogenesis and pharmacotherapy. Am J Cancer Res. 10:3551–3564. 2020.PubMed/NCBI | |
|
Picard FSR, Lutz V, Brichkina A, Neuhaus F, Ruckenbrod T, Hupfer A, Raifer H, Klein M, Bopp T, Pfefferle PI, et al: IL-17A-producing CD8+ T cells promote PDAC via induction of inflammatory cancer-associated fibroblasts. Gut. 72:1510–1522. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Mucciolo G, Curcio C, Roux C, Li WY, Capello M, Curto R, Chiarle R, Giordano D, Satolli MA, Lawlor R, et al: IL17A critically shapes the transcriptional program of fibroblasts in pancreatic cancer and switches on their protumorigenic functions. Proc Natl Acad Sci USA. 118:e20203951182021. View Article : Google Scholar : PubMed/NCBI | |
|
Li J, Betzler C, Lohneis P, Popp MC, Qin J, Kalinski T, Wartmann T, Bruns CJ, Zhao Y and Popp FC: The IL-17A/IL-17RA axis is not related to overall survival and cancer stem cell modulation in pancreatic cancer. Int J Mol Sci. 21:22152020. View Article : Google Scholar : PubMed/NCBI | |
|
Qian X, Chen H, Wu X, Hu L, Huang Q and Jin Y: Interleukin-17 acts as double-edged sword in anti-tumor immunity and tumorigenesis. Cytokine. 89:34–44. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Wang J, Zhang Y, Yin K, Xu P, Tian J, Ma J, Tian X, Wang Y, Tang X, Xu H and Wang S: IL-17A weakens the antitumor immuity by inhibiting apoptosis of MDSCs in Lewis lung carcinoma bearing mice. Oncotarget. 8:4814–4825. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
McAndrews KM, Chen Y, Darpolor JK, Zheng X, Yang S, Carstens JL, Li B, Wang H, Miyake T, Correa de Sampaio P, et al: Identification of functional heterogeneity of carcinoma-associated fibroblasts with distinct IL6-mediated therapy resistance in pancreatic cancer. Cancer Discov. 12:1580–1597. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Ware MJ, Keshishian V, Law JJ, Ho JC, Favela CA, Rees P, Smith B, Mohammad S, Hwang RF, Rajapakshe K, et al: Generation of an in vitro 3D PDAC stroma rich spheroid model. Biomaterials. 108:129–142. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang L, Qin J, Dai Y, Zhao S, Zhan Q, Cui P, Ren L, Wang X, Zhang R, Gao C, et al: Prospective observational study on biomarkers of response in pancreatic ductal adenocarcinoma. Nat Med. 30:749–761. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Bärthel S, Falcomatà C, Rad R, Theis FJ and Saur D: Single-cell profiling to explore pancreatic cancer heterogeneity, plasticity and response to therapy. Nat Cancer. 4:454–467. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Ayars M, O'Sullivan E, Macgregor-Das A, Shindo K, Kim H, Borges M, Yu J, Hruban RH and Goggins M: IL2RG, identified as overexpressed by RNA-seq profiling of pancreatic intraepithelial neoplasia, mediates pancreatic cancer growth. Oncotarget. 8:83370–83383. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Hulst SPL: Zur kenntnis der Genese des Adenokarzinoms und Karzinoms des Pankreas. Virchows Arch. 180:288–316. 1905. View Article : Google Scholar | |
|
Dougan M, Ingram JR, Jeong HJ, Mosaheb MM, Bruck PT, Ali L, Pishesha N, Blomberg O, Tyler PM, Servos MM, et al: Targeting cytokine therapy to the pancreatic tumor microenvironment using PD-L1-specific VHHs. Cancer Immunol Res. 6:389–401. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Ahmed A, Köhler S, Klotz R, Giese N, Lasitschka F, Hackert T, Springfeld C, Zörnig I, Jäger D and Halama N: Peripheral blood and tissue assessment highlights differential tumor-circulatory gradients of IL2 and MIF with prognostic significance in resectable pancreatic ductal adenocarcinoma. Oncoimmunology. 10:19621352021. View Article : Google Scholar : PubMed/NCBI | |
|
Mayer P, Linnebacher A, Glennemeier-Marke H, Marnet N, Bergmann F, Hackert T, Klauss M, Poth T and Gaida MM: The microarchitecture of pancreatic cancer as measured by diffusion-weighted magnetic resonance imaging is altered by T cells with a tumor promoting Th17 phenotype. Int J Mol Sci. 21:3462020. View Article : Google Scholar : PubMed/NCBI | |
|
Linnebacher A, Mayer P, Marnet N, Bergmann F, Herpel E, Revia S, Yin L, Liu L, Hackert T, Giese T, et al: Interleukin 21 receptor/ligand interaction is linked to disease progression in pancreatic cancer. Cells. 8:11042019. View Article : Google Scholar : PubMed/NCBI | |
|
Zaidi N, Quezada SA, Kuroiwa JMY, Zhang L, Jaffee EM, Steinman RM and Wang B: Anti-CTLA-4 synergizes with dendritic cell-targeted vaccine to promote IL-3-dependent CD4+ effector T cell infiltration into murine pancreatic tumors. Ann N Y Acad Sci. 1445:62–73. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Savid-Frontera C, Viano ME, Baez NS, Lidon NL, Fontaine Q, Young HA, Vimeux L, Donnadieu E and Rodriguez-Galan MC: Exploring the immunomodulatory role of virtual memory CD8+ T cells: Role of IFN gamma in tumor growth control. Front Immunol. 13:9710012022. View Article : Google Scholar : PubMed/NCBI | |
|
Hussain SM, Reed LF, Krasnick BA, Miranda-Carboni G, Fields RC, Bi Y, Elahi A, Ajidahun A, Dickson PV, Deneve JL, et al: IL23 and TGF-ß diminish macrophage associated metastasis in pancreatic carcinoma. Sci Rep. 8:58082018. View Article : Google Scholar : PubMed/NCBI | |
|
Mirlekar B, Michaud D, Lee SJ, Kren NP, Harris C, Greene K, Goldman EC, Gupta GP, Fields RC, Hawkins WG, et al: B cell-derived IL35 drives STAT3-dependent CD8+ T-cell exclusion in pancreatic cancer. Cancer Immunol Res. 8:292–308. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Liou GY, Bastea L, Fleming A, Döppler H, Edenfield BH, Dawson DW, Zhang L, Bardeesy N and Storz P: The presence of interleukin-13 at pancreatic ADM/PanIN lesions alters macrophage populations and mediates pancreatic tumorigenesis. Cell Rep. 19:1322–1333. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Shi J, Shen X, Kang Q, Yang X, Denzinger M, Kornmann M and Traub B: Loss of interleukin-13-receptor-alpha-1 induces apoptosis and promotes EMT in pancreatic cancer. Int J Mol Sci. 23:36592022. View Article : Google Scholar : PubMed/NCBI | |
|
Fujisawa T, Shimamura T, Goto K, Nakagawa R, Muroyama R, Ino Y, Horiuchi H, Endo I, Maeda S, Harihara Y, et al: A novel role of interleukin 13 receptor alpha2 in perineural invasion and its association with poor prognosis of patients with pancreatic ductal adenocarcinoma. Cancers (Basel). 12:12942020. View Article : Google Scholar : PubMed/NCBI | |
|
Arnoletti JP, Reza J, Rosales A, Monreal A, Fanaian N, Whisner S, Srivastava M, Rivera-Otero J, Yu G, Phanstiel Iv O, et al: Pancreatic ductal adenocarcinoma (PDAC) circulating tumor cells influence myeloid cell differentiation to support their survival and immunoresistance in portal vein circulation. PLoS One. 17:e02657252022. View Article : Google Scholar : PubMed/NCBI |