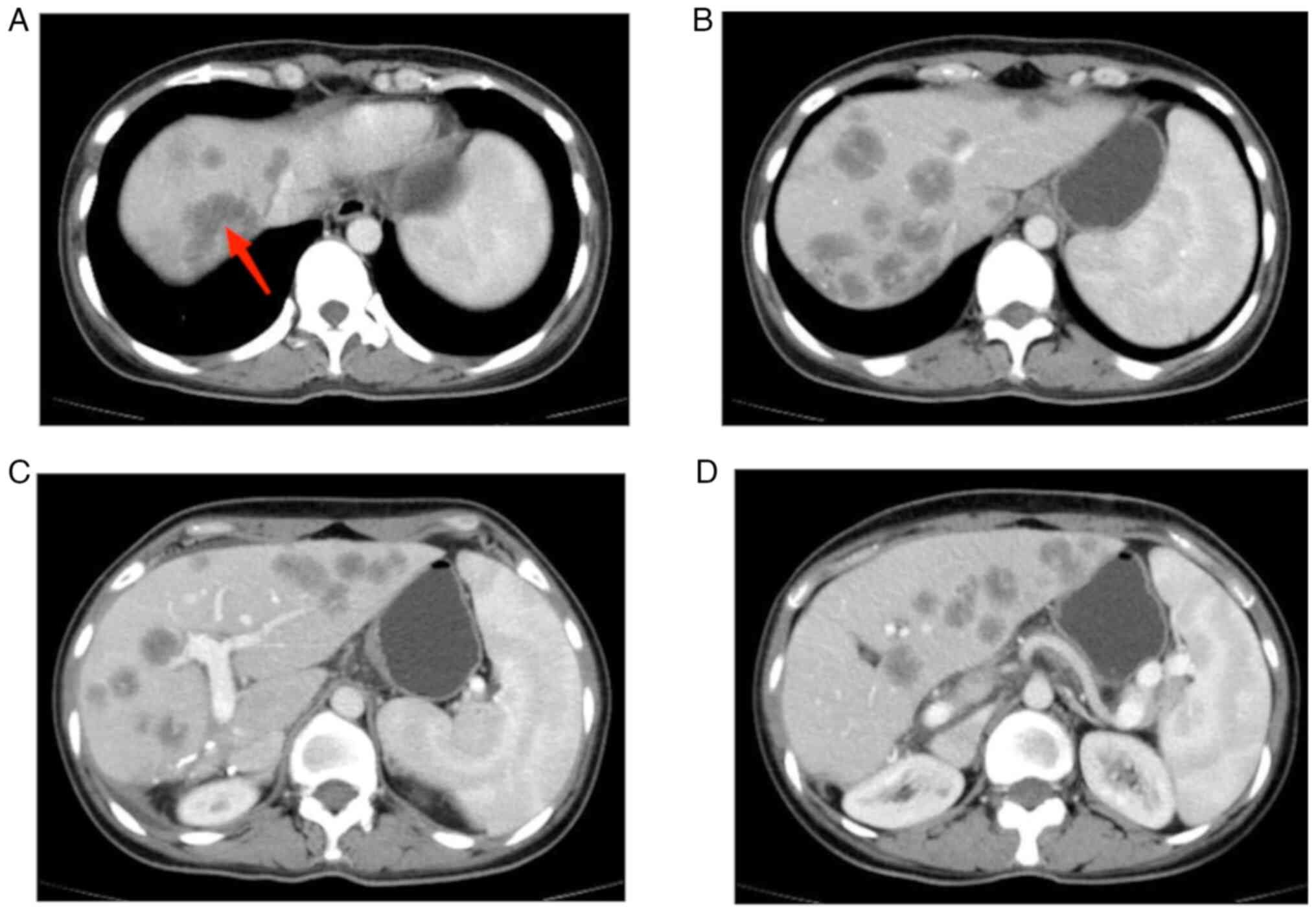
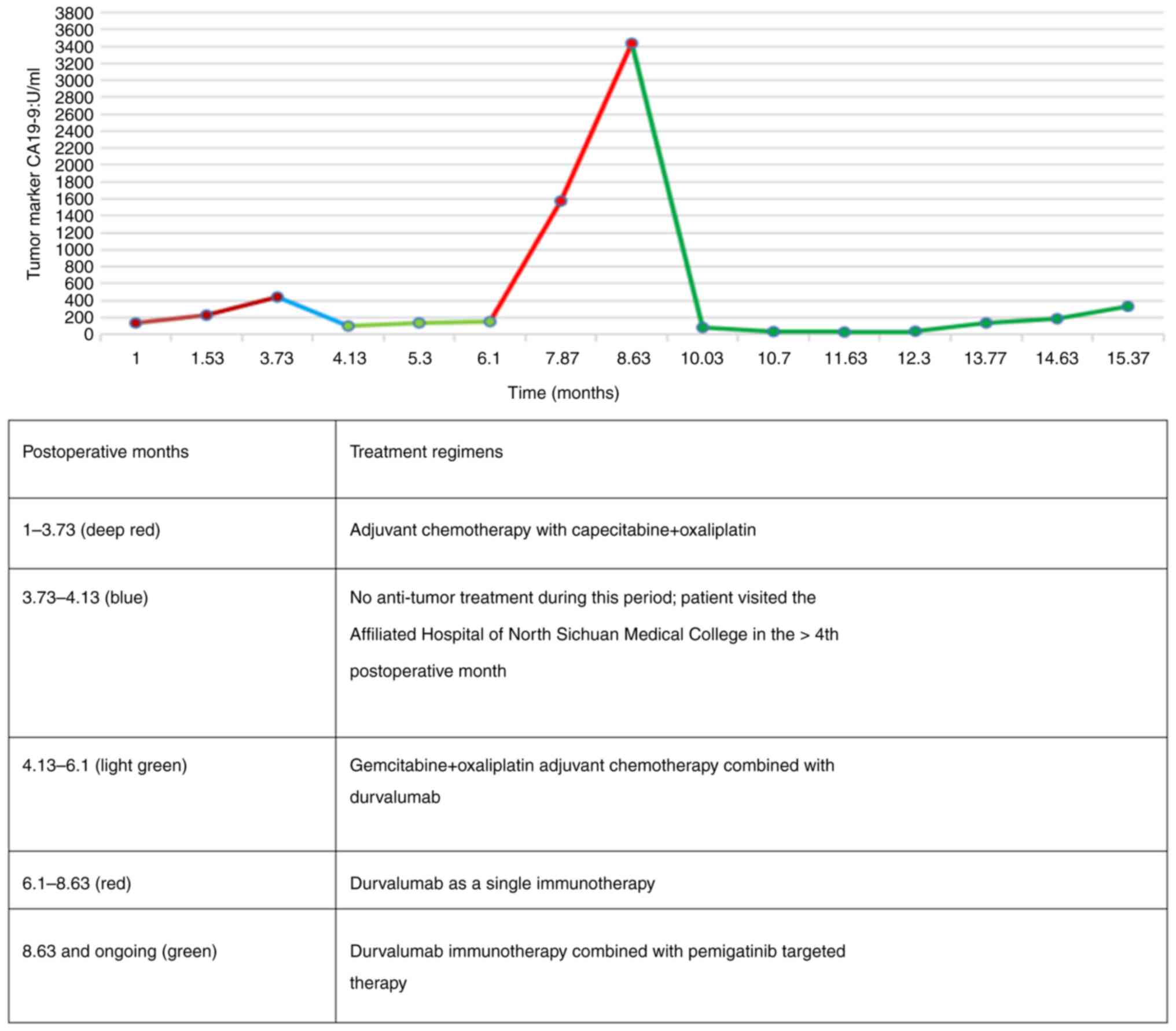
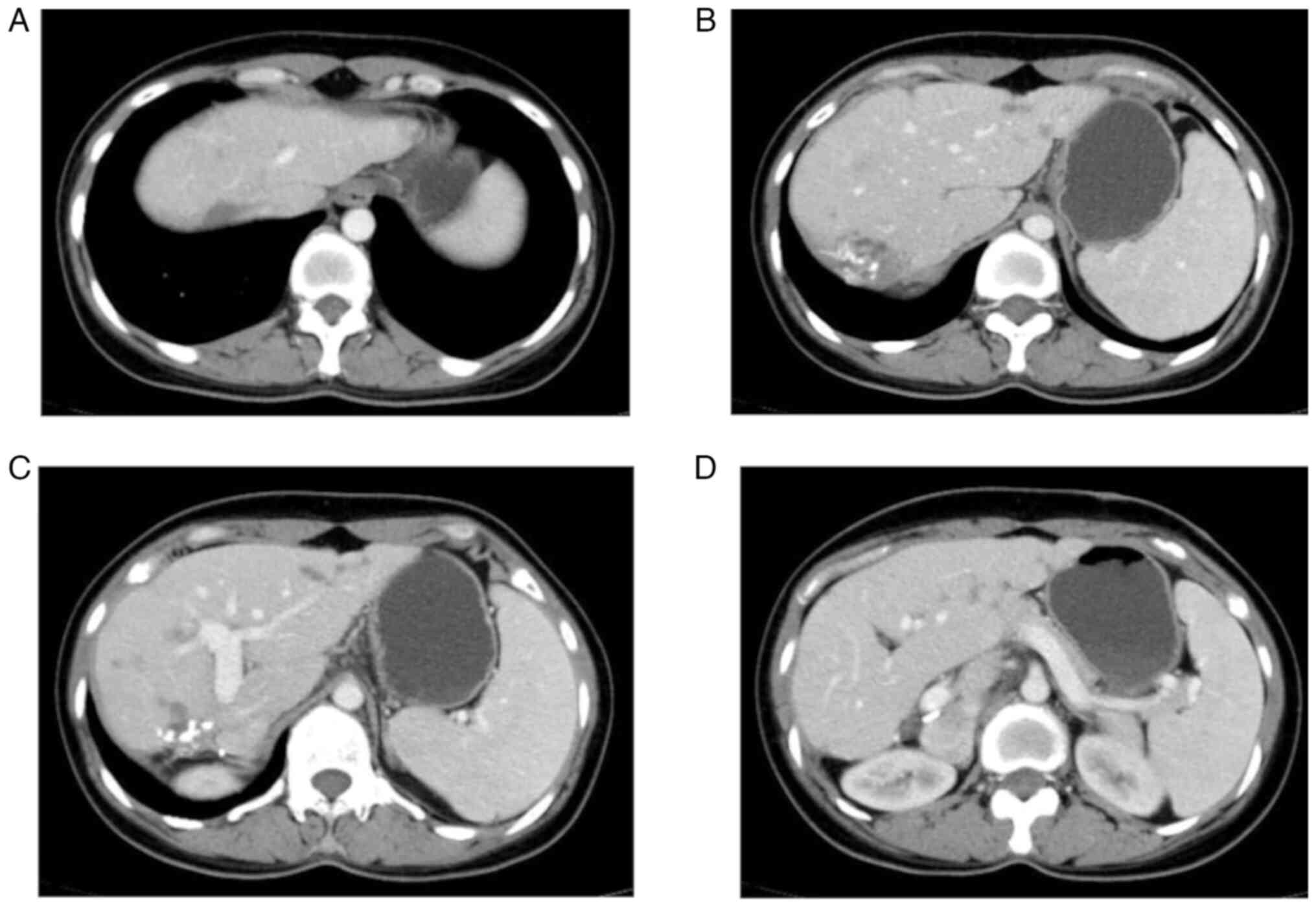
|
1
|
Kelley RK, Bridgewater J, Gores GJ and Zhu
AX: Systemic therapies for intrahepatic cholangiocarcinoma. J
Hepatol. 72:353–363. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Esnaola NF, Meyer JE, Karachristos A,
Maranki JL, Camp ER and Denlinger CS: Evaluation and management of
intrahepatic and extrahepatic cholangiocarcinoma. Cancer.
122:1349–1369. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yamamoto M, Takasaki K, Otsubo T,
Katsuragawa H and Katagiri S: Recurrence after surgical resection
of intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Surg.
8:154–157. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Moris D, Palta M, Kim C, Allen PJ, Morse
MA and Lidsky ME: Advances in the treatment of intrahepatic
cholangiocarcinoma: An overview of the current and future
therapeutic landscape for clinicians. CA Cancer J Clin. 73:198–222.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
National Institutes of Health, . Common
Terminology Criteria for Adverse Events (CTCAE) version 5.
Department of Health and Human Services; National Cancer Institute,
Bethesda, ML: 2017
|
|
7
|
Spolverato G, Kim Y, Alexandrescu S,
Marques HP, Lamelas J, Aldrighetti L, Clark Gamblin T, Maithel SK,
Pulitano C, Bauer TW, et al: Management and outcomes of patients
with recurrent intrahepatic cholangiocarcinoma following previous
curative-intent surgical resection. Ann Surg Oncol. 23:235–243.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ke Q, Lin N, Deng M, Wang L, Zeng Y and
Liu J: The effect of adjuvant therapy for patients with
intrahepatic cholangiocarcinoma after surgical resection: A
systematic review and meta-analysis. PLoS One. 15:e02292922020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang ML, Ke ZY, Yin S, Liu CH and Huang Q:
The effect of adjuvant chemotherapy in resectable
cholangiocarcinoma: A meta-analysis and systematic review.
Hepatobiliary Pancreat Dis Int. 18:110–116. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Schweitzer N, Weber T, Kirstein MM,
Fischer M, Kratzel AM, Reineke-Plaaß T, Lehner F, Manns MP and
Vogel A: The effect of adjuvant chemotherapy in patients with
intrahepatic cholangiocarcinoma: A matched pair analysis. J Cancer
Res Clin Oncol. 143:1347–1355. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Reames BN, Bagante F, Ejaz A, Spolverato
G, Ruzzenente A, Weiss M, Alexandrescu S, Marques HP, Aldrighetti
L, Maithel SK, et al: Impact of adjuvant chemotherapy on survival
in patients with intrahepatic cholangiocarcinoma: A
multi-institutional analysis. HPB (Oxford). 19:901–909. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nakachi K, Ikeda M, Konishi M, Nomura S,
Katayama H, Kataoka T, Todaka A, Yanagimoto H, Morinaga S,
Kobayashi S, et al: Adjuvant S-1 compared with observation in
resected biliary tract cancer (JCOG1202, ASCOT): A multicentre,
open-label, randomised, controlled, phase 3 trial. Lancet.
401:195–203. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Edeline J, Benabdelghani M, Bertaut A,
Watelet J, Hammel P, Joly JP, Boudjema K, Fartoux L,
Bouhier-Leporrier K, Jouve JL, et al: Gemcitabine and oxaliplatin
chemotherapy or surveillance in resected biliary tract cancer
(PRODIGE 12-ACCORD 18-UNICANCER GI): A randomized phase III study.
J Clin Oncol. 37:658–667. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Primrose JN, Fox RP, Palmer DH, Malik HZ,
Prasad R, Mirza D, Anthony A, Corrie P, Falk S, Finch-Jones M, et
al: Capecitabine compared with observation in resected biliary
tract cancer (BILCAP): A randomised, controlled, multicentre, phase
3 study. Lancet. 20:663–673. 2019. View Article : Google Scholar
|
|
15
|
Oh DY, Lee KH, Lee DW, Yoon J, Kim TY,
Bang JH, Nam AR, Oh KS, Kim JM, Lee Y, et al: Gemcitabine and
cisplatin plus durvalumab with or without tremelimumab in
chemotherapy-naive patients with advanced biliary tract cancer: An
open-label, single-centre, phase 2 study. Lancet Gastroenterol
Hepatol. 7:522–532. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Doki Y, Ueno M, Hsu CH, Oh DY, Park K,
Yamamoto N, Ioka T, Hara H, Hayama M, Nii M, et al: Tolerability
and efficacy of durvalumab, either as monotherapy or in combination
with tremelimumab, in patients from Asia with advanced biliary
tract, esophageal, or head-and-neck cancer. Cancer Med.
11:2550–2560. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Graham RP, Barr Fritcher EG, Pestova E,
Schulz J, Sitailo LA, Vasmatzis G, Murphy SJ, McWilliams RR, Hart
SN, Halling KC, et al: Fibroblast growth factor receptor 2
translocations in intrahepatic cholangiocarcinoma. Hum Pathol.
45:1630–1638. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kipp BR, Voss JS, Kerr SE, Barr Fritcher
EG, Graham RP, Zhang L, Highsmith WE, Zhang J, Roberts LR, Gores GJ
and Halling KC: Isocitrate dehydrogenase 1 and 2 mutations in
cholangiocarcinoma. Hum Pathol. 43:1552–1558. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ross JS, Wang K, Gay L, Al-Rohil R, Rand
JV, Jones DM, Lee HJ, Sheehan CE, Otto GA, Palmer G, et al: New
routes to targeted therapy of intrahepatic cholangiocarcinomas
revealed by next-generation sequencing. Oncologist. 19:235–242.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zen Y: Intrahepatic cholangiocarcinoma:
Typical features, uncommon variants, and controversial related
entities. Hum Pathol. 132:197–207. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang XY, Zhu WW, Wang Z, Huang JB, Wang
SH, Bai FM, Li TE, Zhu Y, Zhao J, Yang X, et al: Driver mutations
of intrahepatic cholangiocarcinoma shape clinically relevant
genomic clusters with distinct molecular features and therapeutic
vulnerabilities. Theranostics. 12:260–276. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Abou-Alfa GK, Sahai V, Hollebecque A,
Vaccaro G, Melisi D, Al-Rajabi R, Paulson AS, Borad MJ, Gallinson
D, Murphy AG, et al: Pemigatinib for previously treated, locally
advanced or metastatic cholangiocarcinoma: A multicentre,
open-label, phase 2 study. Lancet Oncol. 21:671–684. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shi GM, Huang XY, Wen TF, Song TQ, Kuang
M, Mou HB, Bao LQ, Zhao HT, Zhao H, Feng XL, et al: Pemigatinib in
previously treated Chinese patients with locally advanced or
metastatic cholangiocarcinoma carrying FGFR2 fusions or
rearrangements: A phase II study. Cancer Med. 12:4137–4146. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim YS, Oh SY, Go SI, Kang JH, Park I,
Song HN, Ji JH, Hwang IG and Jang JS: The role of adjuvant therapy
after R0 resection for patients with intrahepatic and perihilar
cholangiocarcinomas. Cancer Chemother Pharmacol. 79:99–106. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim KS, Kim HY, Kim K, Yi NJ, Suh K and
Chie EK: Postoperative chemoradiotherapy for R1 resected
intrahepatic cholangiocarcinoma. J Liver Cancer. 18:115–120. 2018.
View Article : Google Scholar
|
|
26
|
Li T, Qin LX, Zhou J, Sun HC, Qiu SJ, Ye
QH, Wang L, Tang ZY and Fan J: Staging, prognostic factors and
adjuvant therapy of intrahepatic cholangiocarcinoma after curative
resection. Liver Int. 34:953–960. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wu ZF, Zhang HB, Yang N, Zhao WC, Fu Y and
Yang GS: Postoperative adjuvant transcatheter arterial
chemoembolisation improves survival of intrahepatic
cholangiocarcinoma patients with poor prognostic factors: Results
of a large monocentric series. Eur J Surg Oncol. 38:602–610. 2012.
View Article : Google Scholar : PubMed/NCBI
|