Antioxidant curcumin induces oxidative stress to kill tumor cells (Review)
- Authors:
- Published online on: December 20, 2023 https://doi.org/10.3892/ol.2023.14200
- Article Number: 67
-
Copyright: © Hu et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Reactive oxygen species (ROS) are short-lived, highly electrophilic molecules. They are produced from secondary metabolites generated by a partial reduction of oxygen (1). When intracellular antioxidants are reduced or ROS accumulate excessively, an imbalance in the redox state occurs, which is referred to as oxidative stress. Under such conditions, excessive intracellular accumulation of ROS modifies redox-sensitive amino acid residues in regulatory proteins and alters the actions of proteins and enzymes. Protein kinases, transcription factors and the ubiquitin-proteasome system are vulnerable to excessive accumulation of ROS (2). Of note, when this occurs in tumor cells, it promotes tumor development. Stimulation of signaling pathways by ROS promotes the proliferation, migration and invasion of tumor cells, including in human breast, skin and liver cancers (3). Furthermore, oxidative stress caused by excessive accumulation of ROS can lead to genetic instability. Cell death modalities, such as apoptosis, autophagy, ferroptosis and pyroptosis, act as protective mechanisms to prevent the proliferation of damaged cells (4) and this is also seen in tumor cells. Thus, ROS effects are complex in tumor cells (5,6). Specifically, tumor cells maintain moderate-to-high ROS levels, namely, above the low cytostatic level and below the cytotoxic level, by enhancing their own antioxidant capacity. Therefore, the ROS level in tumor cells is subtoxic, which facilitates tumor cell progression, and ROS act as signaling molecules to increase the proliferation of tumor cells (7–10). However, under further oxidative stress, cancer cells are equally susceptible to excessive ROS (11), and increased ROS unbalance the redox response of cancer cells, ultimately leading to cellular senescence or death (12).
Curcumin is a plant polyphenol in the rhizome of turmeric and was classified as a third-generation cancer chemopreventive agent by the National Cancer Institute (13). Several studies have reported anticancer mechanisms mediated by curcumin through the induction of elevated ROS (14,15). This contradicts the antioxidant properties of curcumin, the potential reasons for which are discussed in the present review. Unfortunately, properties such as poor water solubility and low bioavailability limit the clinical application of curcumin (13). However, the clinical efficacy of curcumin has been enhanced by combining it with drugs, the introduction of nanocarriers and the development of curcumin derivatives, which have brought the clinical application of curcumin closer to reality (16–18). Therefore, the present study reviewed articles in which the effect of curcumin on tumors was evaluated, with a particular focus on its relationship with oxidative stress, to provide a reference for future studies.
Introduction to curcumin
Curcumin, also known as diferuloylmethane, has the chemical formula C21H20O6, a molecular weight of 368.38 g/mol and a symmetrical molecular structure (19). Curcumin has been commonly used as an aromatic and natural food coloring agent; however, its biological effects are gradually being elucidated. Curcumin has anti-inflammatory, antibacterial, hepatoprotective and anticancer properties (20–22), and its anticancer effects have been reported in several tumor types (Table I). In melanoma, curcumin has been reported to increase the ROS level and activate oxidative stress in the cysteine asparaginase pathway, which causes tumor cell death (23). Lysosome-associated membrane protein 3 belongs to the fourth transmembrane protein superfamily, which promotes tumor cell invasion and metastasis. It interacts with ubiquitin-specific peptidase 4 and is positively regulated by the latter. A recent study reported that curcumin downregulated ubiquitin-specific peptidase 4 to modulate lysosome-associated membrane protein 3, and thus inhibited the malignant progression of colorectal cancer cells (24). Furthermore, curcumin-induced accumulation of ROS in tumors to kill tumor cells has been noted in several studies (25,26), as discussed in the present review.
Curcumin is well tolerated by humans. For example, a study that evaluated the toxicity of curcumin in humans reported that subjects administered 8 mg/day curcumin (99.3% purity) did not develop toxicity (27). However, certain physical and chemical properties of curcumin limit its clinical application. For example, whilst its high lipophilicity assists in penetrating lipid structures, it is insoluble in water and has poor stability, resulting in low bioavailability (13). In a previous study, 440–2,200 mg curcumin extract was administered to patients with advanced colorectal cancer for 29 consecutive days, and curcumin was not detected in patient blood or urine. In several clinical trials, ≤12 g/day curcumin was administered orally, but the amount of curcumin detected in serum was <1% (27,28). To overcome these drawbacks, the use of curcumin in combination with other drugs to enhance its anticancer activity has been reported (29). In addition, the development of curcumin derivatives, the use of carriers or coverings such as chitosan, and nanosystems have been suggested to improve the bioavailability of curcumin (30). Thus, the potential for the clinical application of curcumin is increasing.
Curcumin causes oxidative stress in tumor cells and leads to cell death in different ways
Mechanism of action of curcumin against cancer via oxidative stress
ROS serve an essential role in the development of cancers as the microenvironment of cancer cells is involved in ROS homeostasis. High ROS levels have a cytotoxic effect on cancer cells, leading to malignant cell death and thus limiting cancer progression. Curcumin treatment of tumor cells induces high levels of ROS production and accumulation, resulting in a redox imbalance in tumor cells (31). As shown in Fig. 1, high ROS levels can cause mitochondrial damage, thereby triggering different types of cell death, such as apoptosis, autophagy, ferroptosis and pyroptosis.
Induction of mitochondrial oxidative stress
The main source of intracellular ROS is the respiratory chain of the inner mitochondrial membrane. ROS are continuously produced as a by-product of aerobic metabolism and are removed by cellular antioxidant mechanisms to maintain an essentially non-toxic level. Under normal physiological conditions, ROS act as specific molecular regulators of cellular signaling and function. A typical mode of ROS regulation is the reversible oxidation of target protein sulfhydryl groups to cystine to mediate biological effects. In addition, ROS-induced changes in the intracellular redox state affect cellular activities, including signaling, metabolism, growth and apoptosis (10,32,33). However, in tumors under inflammatory or stress conditions, the excess ROS produced cannot be adequately neutralized. These excess ROS propagate in the intercellular compartment and react with proteins and nucleic acids. They also react with polyunsaturated fatty acids to produce lipid hydroperoxides and unsaturated aldehydes, causing oxidative stress in mitochondria (34). Such an oxidative cascade affects several mitochondrial functions, such as biogenesis, ion homeostasis and antioxidant defense mechanisms (34). In non-neoplastic disease studies, curcumin is often considered a highly potent antioxidant that reduces oxidative damage to mitochondria. In a rat model of acute respiratory distress syndrome, curcumin treatment attenuated renal tubular epithelial and mitochondrial damage and reduced oxidative stress (35,36). Conversely, curcumin induced excessive ROS production in tumor cells and caused mitochondrial damage, which ultimately exerted its cancer-suppressive effects through certain cell death modalities (25). In addition, a study have reported that mitochondrial oxidative homeostasis can be disrupted by targeting mitochondria with curcumin. Curcumin-coated micelles effectively inhibit the progression of gastric cancer, and the potential therapeutic mechanism may be its effect on mitochondrial proteins to reduce the mitochondrial membrane potential and increase ROS to disrupt oxidative homeostasis (26). In addition to targeting curcumin to mitochondria through micelles, several other approaches may achieve this goal. For instance, the introduction of different nanocarriers and the development of curcumin targeting mitochondrial derivatives (37,38). Therefore, a therapeutic approach may be to induce oxidative stress in the mitochondria of tumor cells by targeting curcumin to mitochondria.
Curcumin induces apoptosis via ROS
Apoptosis is a form of programmed cell death that is mainly induced by damage to DNA and organelles, such as the mitochondria and endoplasmic reticulum (ER), via stimuli such as oxidative stress, chemotherapeutic drugs and ionizing radiation (39). The ability of curcumin to cause apoptosis has been reported in several types of tumor. After treatment of drug-resistant tumor cell lines, MCF7/TH, HCT116R and A549/ADR, with curcumin, ROS levels and expression of apoptotic markers, such as Bax and cytochrome c, were notably increased in the high-dose group compared to the low-dose group (25).
The mitochondrion-dependent pathway is a classical pathway that mediates apoptosis. Curcumin causes mitochondrial damage by promoting phosphorylation of ERK and JNK, resulting in the increased release of ROS and cytochrome c into the cytoplasm, thereby triggering a mitochondrion-dependent pathway of apoptosis. In addition, ER stress and mitochondrial dysfunction induce apoptosis (40). In the treatment of human papillary thyroid cancer with curcumin, both activating transcription factor (ATF) 6 and the ER stress marker C/EBP homologous protein (CHOP) were activated by curcumin and Ca2+-ATPase activity was also affected. This led to Ca2+ accumulation in the mitochondrial matrix, causing mitochondrial swelling, membrane potential changes and elevated ROS levels, which eventually resulted in mitochondrial rupture and the release of cytochrome c and other proapoptotic proteins into the cytoplasm (41,42).
It has been reported that 85% of patients with colon cancer have mutations or loss of function and expression of the Smad4 and p53 genes (43). Therapeutic agents that induce ROS-mediated apoptosis have been suggested to be potential agents for the treatment of Smad4- and p53-mutant colon cancer (44). Curcumin induced a marked increase in ROS levels in p53-mutated colon cancer HT-29 cells and induced apoptosis in colon cancer cells by altering the mitochondrial membrane potential (45). Another study reported the possible involvement of p53 in curcumin-mediated apoptosis of colon cancer cells by treating wild-type (HCT-116) and mutant (HT-29) p53 colon cancer cell lines with curcumin, but the underlying mechanism was not elucidated (46). Results from the aforementioned study indicate that under stress conditions such as tumorigenesis, p53 undergoes mitochondrial translocation and binds to superoxide dismutase (SOD), rendering it inactive and inducing ROS production, which triggers apoptosis. In turn, curcumin treatment further induces ROS generation, which again subjects p53 to oxidative stress conditions and further mitochondrial translocation, which serves a further role in promoting apoptosis. This may also explain why N-acetylcysteine (NAC) inhibited the loss of the mitochondrial membrane potential in HT-29 cells treated with both low and high concentrations of curcumin in the aforementioned study, whereas in HCT-116 cells, it only affected cells treated with low concentrations of curcumin.
Curcumin induces autophagy via ROS
Autophagy is another type of programmed cell death. In certain cases, autophagy phagocytoses cytoplasmic proteins or organelles and encapsulates them in vesicles for fusion with lysosomes to form autophagic lysosomes to degrade their encapsulated contents. During the process of degradation, autophagic lysosomes provide essential peptides and amino acids to cells (47,48). Inducing tumor cells to undergo autophagy to kill the tumor cells is a potential approach for clinical treatment and numerous studies have reported that curcumin induces autophagy in cancer cells. For instance, in ovarian cancer, a study demonstrated that curcumin induced autophagy via the inhibition of the AKT/mTOR/p70S6K signaling pathway, enhanced light chain (LC)3B-I/II expression and increased autophagy related 3 and Beclin1 expression in a concentration-dependent manner (49). ROS also induced cell damage and disrupted specific signaling pathways, leading to autophagic cell death, which is also known as type II cell death (50). Furthermore, a study evaluated the targets of curcumin in colon cancer and found that it serves a role in ROS generation and autophagy induction (51). Similarly, another study reported have reported that patients with colon cancer with high expression of heat shock protein 27 are more sensitive to curcumin (52). This may be due to the high expression of heat shock protein 27 increasing ROS levels and activating autophagy. Treatment with curcumin enhanced this effect. In addition, a study reported abnormal energy metabolism and accumulation of ROS in cervical cancer cells after curcumin treatment and further analysis of autophagy revealed that curcumin promoted the conversion of LC3 to LC3II and degradation of autophagosome markers. This process was blocked by the antioxidant NAC, indicating that curcumin induced autophagy by increasing ROS (53). Of note, curcumin derivatives have been reported to induce ROS production and activate autophagy. For instance, EF24 is a curcumin derivative with promising anticancer effects, and treatment of non-small lung cancer cells with EF24 was shown to result in a notable increase in ROS, autophagic markers and vesicles (54).
Curcumin triggers ferroptosis via ROS
Direct induction of cytotoxicity in cancer cells is the main goal of anticancer therapy. Unlike apoptosis, necrosis and autophagy, ferroptosis is the iron-dependent programmed cell death (55). It is characterized by intracellular iron accumulation, resulting in excessive ROS production, decreased glutathione (GSH) levels and lipid peroxidation (56). Heme oxygenase 1 (HO-1) catalyzes the degradation of heme to carbon monoxide, biliverdin and free iron (57). Its upregulation alters iron homeostasis and reduces tumor cell survival. Furthermore, oxidative stress induced by organic oxidants, such as tert-butyl hydroperoxide, have been reported to promote HO-1 translocation to the mitochondria, causing mitochondrial iron overload and ferroptosis in cardiomyocytes (58,59). Treatment of breast cancer M7 and 231 cell lines with curcumin has been reported to promote HO-1 expression, which in turn promoted heme degradation, leading to an increase in Fe2+ and ultimately an altered cellular iron distribution (60). Iron overload triggers the Fenton reaction, which increases ROS levels and causes peroxidation and oxidative damage to surrounding lipids and proteins (61). GSH peroxidase 4 (GPX4) acts as an antioxidant and counteracts lipid peroxidation (62). However, its expression is downregulated in curcumin-treated breast cancer cells, which eventually leads to ferroptosis (60). Furthermore, HO-1 is activated by nuclear factor erythroid 2-related factor 2 (Nrf2) in the nucleus to perform its antioxidant function (63). However, HO-1 is upregulated and elevates ROS and induces ferroptosis, suggesting a dual role for HO-1. In a study of breast cancer, curcumin induced elevated levels of lipid ROS, accumulation of lipid peroxide end product malondialdehyde and increased intracellular Fe2+ levels, ultimately enhancing the expression of solute carrier family 1 member 5 to induce ferroptosis in vitro and in vivo (64). Tang et al (65) assessed the specific role and potential mechanism of ferroptosis in treating non-small cell lung cancer with curcumin. Curcumin significantly triggered GSH depletion, lipid peroxidation and accumulation of ROS and iron in mice as well as in A549 and H1299 cells. In addition, pretreatment with the hemostatic inhibitor ferrostatin-1 or iron responsive element binding protein 2 knockdown notably reduced curcumin-induced siderosis in A549 and H1299 cells. Notably, in these cells, pretreatment with the autophagy inhibitor chloroquine or knockdown of the autophagy-related gene Beclin1 reduced curcumin-induced autophagy and subsequent ferroptosis, suggesting that activation of the autophagy pathway may also trigger ferroptosis in tumor cells.
Curcumin causes pyroptosis through ROS
Pyroptosis is a recently discovered form of programmed cell death that relies on the activation of inflammation-associated caspase-1, −4, −5 and −11, and apoptosis-associated caspase-3 (66). Activated caspase-3 cleaves gasdermin E (GSDME) proteins, releasing the active GSDME N-terminal and pore-forming structural domains, leading to the formation of non-selective pores in the cell membrane (67). A study has reported that cell swelling, membrane lysis, a large number of scorched vesicles and increased expression of GSDME-N appeared after curcumin treatment of hepatocellular carcinoma. In addition, ROS levels notably increased. Treatment with NAC inhibited pyroptosis induced by curcumin. These results suggest that curcumin induces pyroptosis by regulating ROS levels, but the exact mechanism has not been elucidated (68). Furthermore, a study reported on the design of a dicarbonyl curcumin analog (B2) of the curcumin β-dione structure (69). B2 treatment of lung cancer H146 cells increased the level of intracellular ROS and expression of caspase-3 in a concentration-dependent decrease, resulting in GSDME-N production. Thus, B2 exhibited antitumor activity through transition from apoptosis to pyroptosis. Combined with the possibility that high concentrations of ROS activate the ER stress-mediated cell death pathway, Wei et al (69) further assessed the transcription factors associated with ER stress pathways and reported that B2 increased the mRNA expression of binding immunoglobulin protein in H460 cells and thus the mRNA expression of ATF-4 and X-box binding protein (XBP-1). ATF-4 and XBP-1 entered the nucleus as transcription factors to regulate ER stress, resulting in increased protein expression of CHOP. The aforementioned studies indicate that the inhibitory effect of B2 on tumor cells may be achieved via the ROS-activated ER stress-mediated cell death pathway.
Combination of curcumin with anticancer drugs improves cancer inhibition
Curcumin is a pigment with a diketone structure. Curcumin-induced ROS is crucial for cancer cell death. In addition, curcumin mediates chemosensitization. However, its low solubility and poor stability limit its clinical application. To increase curcumin use, the combination of curcumin and drugs has been reported to sensitize drug-resistant cancer cells and enhance therapeutic effects (Table II) (70).
Polyphenols, such as curcumin, are a large family of organic compounds that contain a common ternary flavonoid ring system structure and polyphenolic units (70). These natural compounds are mainly found in plants and are beneficial to humans. For instance, certain polyphenols have been used to treat tumors, such as in one study where the treatment of breast cancer cells with resveratrol resulted in differential expression of several genes related to the cell cycle and induced S-phase arrest (71). Curcumin has also been reported to mediate the arrest of tumor cells in G2/M phase via p53 (17). However, polyphenols are also associated with negative outcomes, such as drug resistance induction. Nevertheless, studies have reported that combinations of certain polyphenols improved therapeutic effects on tumors, and both curcumin and resveratrol reduced cancer cell survival by upregulating ROS. Furthermore, the combination of curcumin and resveratrol notably increased ROS production compared with either agent alone (16,72). Furthermore, NAC treatment significantly reversed apoptosis induced by the combination of curcumin and resveratrol, and the expression of caspase-3, −8, and −9 was also reduced. These findings suggest that combination therapy with curcumin and resveratrol may induce apoptosis by activating caspases and ROS production. In addition, the combination of curcumin and resveratrol was reported to have activated ER stress, enhanced the activity of the protein kinase RNA-like ER kinase/eukaryotic initiation factor-2α/CHOP axis and induced apoptosis (73). The combination of the two increased poly (ADP-ribose) polymerase expression inhibited DNA damage, promoted apoptosis and reduced drug resistance in bladder cancer cells (74).
Paclitaxel (PTX), which is widely used for the treatment of lung cancer, induces cell death by disrupting the normal microtubule dynamics required for cell division and important interphase processes. In addition, PTX induces cell cycle arrest (75). However, PTX also has several toxic effects on normal cells, such as the induction of neurotoxicity and hematotoxicity (76). By measuring the amount of ROS produced when the lung cancer A549 and Calu-3 cell lines were co-treated with curcumin and PTX using a fluorescent probe, a study reported that the combination resulted in higher levels of ROS and showed stronger anticancer effects, such as the inhibition of cell proliferation, as well as induction of apoptosis and cell cycle arrest, compared with curcumin or PTX treatment alone (77). Of note, in the aforementioned study, curcumin also attenuated the toxic effects of PTX on normal cells. For instance, when the curcumin dose was higher, the combination was less cytotoxic to the normal lung Beas-2B cell line compared with the higher PTX dose group. This may indicate that curcumin cannot be internalized by normal cells (78,79) and therefore cannot induce the production of superoxide compounds or be toxic to normal cells. However, in tumor cells, curcumin has been reported to be internalized and induced the production of superoxide compounds (80), which, in conjunction with PTX, enhanced oxidative stress in tumor cells. In addition, as an antioxidant, curcumin in turn attenuated the oxidative stress caused by PTX in normal cells via several mechanisms, such as increasing the expression of glutathione and antioxidant proteins (81,82). Therefore, the combination of curcumin and PTX may enhance anticancer activity in cancer cells whilst protecting healthy cells from irreversible damage.
P-glycoprotein (P-gp) is the pump responsible for drug efflux and its high expression is associated with drug resistance in tumors (83). Attia et al (84) reported that combining curcumin and PTX reduced the activity and expression of P-gp and multidrug resistance 1 and increased PTX sensitivity in breast cancer. Furthermore, curcumin downregulated NF-κB, which increased the sensitivity of gastric cancer cells to PTX (85). Doxorubicin (Dox) is commonly used to treat malignant tumors (86–88). However, developing P-gp-mediated multidrug resistance during tumor chemotherapy severely reduces the therapeutic efficacy of Dox. Curcumin has been reported to have inhibited D-glutamine metabolism by decreasing the expression of ornithine decarboxylase. It also markedly inhibited the biosynthesis of spermine and spermidine, thereby reducing the oxidative stress capacity of SW620/AD300 cells and the ATP-dependent transport activity of P-gp. This increased the intracellular accumulation of Dox in drug-resistant cells and ultimately reversed the multidrug resistance of tumors (89). Finally, deguelin is a fish-like pigment in an African rhizome plant, which is used as an insecticide. Deguelin inhibits sperm function via the PI3K/AKT signaling pathway (90) and serves a role in cancer inhibition (91). For instance, deguelin has been reported to kill prostate cancer cells (92). Kocdor et al (93) reported that curcumin and deguelin were able to reduce the oxidative stress index and enhance the activity of superoxide dismutase.
Anticancer effects of curcumin derivatives are mediated via oxidative stress
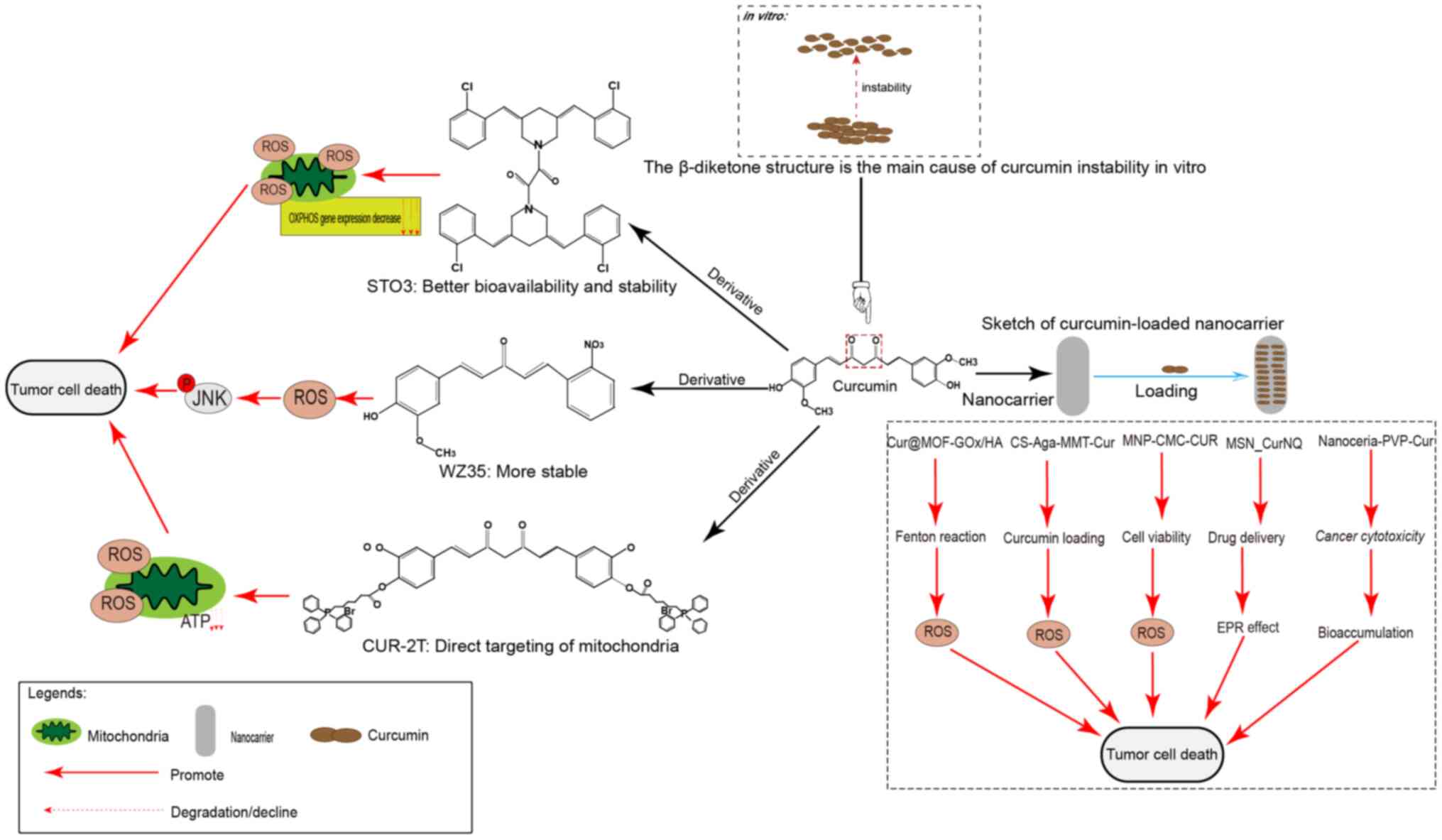
In addition to its use in combination with drugs, derivatives of curcumin are being developed to improve its pharmacological effects (Fig. 2). A study synthesized niacin (an essential vitamin necessary for normal cell function) with curcumin to produce a new curcumin derivative called nicotinic acid (17), which has greater selectivity for cancer cells than curcumin itself. As the curcumin derivative had an improved pharmacokinetic profile in vivo, it more strongly stimulated oxidative stress. Furthermore, β-diketones may be responsible for the instability and weak pharmacokinetic profile of curcumin in vitro. In a study, its β-diketone structure was deleted and a new curcumin derivative, WZ35, was constructed, which exhibited markedly improved chemical stability in vitro (94). In a follow-up study, WZ35 induced apoptosis in gastric cancer cells by increasing the ROS level, thereby causing ER stress and activating the JNK signaling pathway (95). Furthermore, WZ35 caused cell death in hepatocellular carcinoma and breast cancer cells via a similar mechanism (96,97). ST03, a recently synthesized curcumin derivative (98,99), has been reported to have improved bioavailability and stability, and to be detectable in plasma for up to 12 h. In a previous study, ST03 induced a peak in ROS within 1 h and a reduction in the transcription of mitochondrion DNA-encoded respiratory chain-associated protein genes after 48 h, which ultimately led to ovarian cancer cell death (99). A recent study synthesized novel mitochondrion-targeted curcumin derivatives by unilateral coupling of the curcumin phenolic hydroxyl group to triphenylphosphine through an ester bond or bilateral coupling (CUR-2T) (38). CUR-2T demonstrated a clear preferential selectivity for cancer cells and directly targeted mitochondria, resulting in disruption of the redox balance accompanied by increased ROS levels, decreased ATP levels and tumor cell cycle arrest.
The introduction of nanocarriers and loaders has greatly enhanced the loading and targeting ability of curcumin. A study constructed metal-organic frameworks (MOFs) modified with glucose oxidase (GOx). After loading with curcumin, it is further modified with tumor-targeting hyaluronic acid (HA) to obtain Cur@MOF-GOx/HA nano-enzymes. Exposure to GOx triggered tumor starvation and produced H2O2 to provide reactants for the MOF-mediated Fenton reaction to produce large amounts of ROS, whilst releasing curcumin to induce the autophagic death of tumor cells (18). In another study, a nanocomposite hydrogel platform for montmorillonite nanoparticles added to chitosan-agarose was constructed, increasing the loading capacity of curcumin from 63 to 76% and increasing the apoptotic rate. The delivery platform in the present study enhanced the curcumin load, sustained its release and increased its anticancer effects (100). In another study, magnetic nanoparticles containing curcumin with carboxymethyl chitosan (MNP-CMC-CUR) were designed and used to treat breast cancer MCF-7 and MDA-MB-231 cell lines and human fibroblasts, and their effect was compared to that of curcumin alone in MTT assays (101). It was observed that the IC50 of MCF-7 cells treated with MNP-CMC-CUR was notably reduced compared with that of curcumin itself without affecting the metabolic activity of normal cells. Furthermore, p53 and caspase-3 gene expression was markedly increased in MCF-7 cells treated with MNP-CMC-CUR. A study reported the cessation of G1-stage cancer cell growth after hyperthermia, as well as an increase in caspase-3 expression and ROS production. Furthermore, MNP-CMC-CUR improved drug effectiveness, and when combined with hyperthermia, the therapeutic effect was enhanced (102).
Mesoporous silica nanoparticles (MSNs) are a class of nanoparticles extensively studied for drug delivery applications. Curcumin and naphthoquinone (NQ) are novel therapeutic diagnostic molecules for cancer targeting, detection and treatment (103). A novel nanosystem has been developed using MSN_CurNQ that increases drug delivery of CurNQ by increasing the enhanced permeability and retention effect and sustained release (104). A study demonstrated that MSN_CurNQ treatment did not elicit any cytotoxicity in the fibroblast 3T3 cell line, but reduced the viability of cancer cells to <50%, indicating tumor-specific toxicity (103). Water- and alcohol-soluble cerium oxide-curcumin conjugates were obtained by co-evaporation with polyN-vinylpyrrolidone (PVP) (105), which induced oxidative stress under ultraviolet (UV) irradiation or hydrogen peroxide conditions. Nanoceramic PVP-curcumin (NPC) conjugates exhibited selective cytotoxicity. Unlike curcumin itself, NPC conjugates demonstrated photosensitivity in tumor cell cultures, whilst protecting untransformed cultures from the damaging effects of UV radiation and oxidative stress.
Curcumin combined with MOFs, nanocomposite hydrogel platforms, nanoparticles and nanosystems may enhance the sustained release of curcumin via several mechanisms of action to improve its targeting and release curve, increase the solubility of curcumin and toxicity in tumor cells, improve bioavailability and increase its anticancer effects.
Discussion
Cancer is a significant health issue, and although early diagnosis and targeted therapy have markedly reduced mortality, cancer drug resistance and drug side effects are still clinical problems that need to be resolved (106). Curcumin is a natural compound that has been used for the treatment of numerous types of diseases, such as Alzheimer's disease, fatty liver and cancer (53,107,108). Of note, curcumin has a dual role in oncological and non-oncologic diseases. Specifically, in non-neoplastic diseases, curcumin is a potent antioxidant that attenuates oxidative stress and mitochondrial damage (35). Conversely, in tumors, curcumin binds to several enzymes and increases ROS levels (23,24). These different effects may be the result of differences in dosage. For instance, in a previous study on curcumin treatment of drug-resistant tumor cells, a low dose of curcumin showed no effect on antioxidant proteins, whereas a high dose resulted in the inhibition of antioxidant proteins, thereby increasing ROS levels (25,109–112). Furthermore, mitochondria may be a potential target for high-dose curcumin. During tumorigenesis, mitochondria are often functionally and morphologically impaired, leading to aberrant changes in ROS levels, and high-dose curcumin treatment has been reported to exacerbate the effects on damaged mitochondria (42,113). In addition, compared with normal cells, proteins abnormally expressed in tumor cells, such as GSH and HO-1, may be targeted by curcumin to cause oxidative stress in tumor cells. The reasons for the dual action of curcumin need to be further explored.
Curcumin induces high levels of ROS accumulation, leading to an imbalance in the redox response. This results in mitochondrial and DNA damage and subsequent activation of the cell death pathway, providing possible approaches for cancer therapy. The MAPK signaling pathway consists of three distinct cascades, ERK, JNK and p38, which serve a role in cell growth, proliferation, motility and death (114); therefore, it may be an important pathway to target using curcumin to induce apoptosis in cancer cells. Curcumin promotes phosphorylation of ERK and JNK and induces ROS production, leading to mitochondrial damage and cytochrome C release, which activates caspase-related signaling pathways to cause apoptosis (115). In addition, it disrupts intracellular calcium homeostasis and activates ER stress, leading to apoptosis (42). Of note, changes in caspase-3 expression have also been reported to induce the production of GSDME-N and the formation of large amounts of pore, which ultimately lead to cell membrane rupture and pyroptosis (66). In terms of autophagy, curcumin has been reported to induce elevated ROS levels and the appearance of autophagy markers and autophagosomes, causing tumor cells to undergo autophagy. Nrf2 is a classical transcription factor that regulates the cellular oxidative stress response, which controls several cellular behaviors, such as cellular detoxification and oxidative stress, and induces the downstream production of a series of cytoprotective proteins to maintain the balance of the intracellular environment and prevent diseases (116). Curcumin achieves anticancer effects by regulating the expression of Nrf2 and its downstream target HO-1, inhibiting the expression of GPX4 and altering the accumulation of intracellular iron and inducing the Fenton reaction (60). In addition, the combination of curcumin with drugs, the introduction of curcumin derivatives and nanocarriers have markedly improved the pharmacokinetics of curcumin and enhanced its effects, including oxidative stress to kill tumor cells.
Over the past two decades, the mechanisms by which curcumin inhibits several types of tumor have been gradually elucidated. In addition, research on curcumin derivatives and nanocarriers has been performed and its clinical therapeutic potential has been evaluated. However, research on curcumin needs to be further deepened in terms of the following aspects: i) Studies have reported that the cytotoxicity of curcumin nanoparticles is notably increased and water-soluble curcumin components demonstrate greater toxicity compared with curcumin itself (117,118). Therefore, the toxicity of nanoparticles and water-soluble curcumin needs to be reduced in the development of these substances; ii) clinical trials of nano-curcumin in tumor patients should be performed; iii) several formulations of curcumin have produced markedly different results in clinical trials. In a study by Sharma et al (119), patients experienced adverse reactions such as diarrhea at a dosage of 3.6 g curcumin, whereas no relevant therapeutic toxicity was observed at 8 g curcumin with 99.3% purity as described above. The difference between the two studies may be due to the different formulations of curcumin. Therefore, several formulations of curcumin should be assessed to reduce its clinical toxicity and enhance its pharmacokinetic effects; iv) the mechanism of action of curcumin in several tumor types should continue to be evaluated, with oxidative stress being the potential predominant mechanism.
In conclusion, curcumin may have the potential to become a cutting-edge drug for the treatment of tumors and other diseases. In-depth research on curcumin should be performed, as well as more clinical trials related to curcumin, to evaluate its anticancer activity.
Acknowledgements
Not applicable.
Funding
The present review was supported by the National Science Foundation of China (grant no. 31560312).
Availability of data and materials
Not applicable.
Authors' contributions
YH and LC designed and organized this manuscript. YH contributed the first draft of the manuscript, and LC refined and revised the contents, tables and figures. SD wrote the basic sections of the manuscript. KW and SL revised the manuscript. All authors read and approved the final manuscript. Data authentication does not apply.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Dizdaroglu M: Oxidatively induced DNA damage and its repair in cancer. Mutat Res Rev Mutat Res. 763:212–245. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Moldogazieva NT, Lutsenko SV and Terentiev AA: Reactive oxygen and nitrogen species-induced protein modifications: Implication in carcinogenesis and anticancer therapy. Cancer Res. 78:6040–6047. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Moloney JN and Cotter TG: ROS signalling in the biology of cancer. Semin Cell Dev Biol. 80:50–64. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Galadari S, Rahman A, Pallichankandy S and Thayyullathil F: Reactive oxygen species and cancer paradox: To promote or to suppress? Free Radic Biol Med. 104:144–164. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Frijhoff J, Winyard PG, Zarkovic N, Davies SS, Stocker R, Cheng D, Knight AR, Taylor EL, Oettrich J, Ruskovska T, et al: Clinical relevance of biomarkers of oxidative stress. Antioxid Redox Signal. 23:1144–1170. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Saikolappan S, Kumar B, Shishodia G, Koul S and Koul HK: Reactive oxygen species and cancer: A complex interaction. Cancer Lett. 452:132–143. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Saha SK, Lee SB, Won J, Choi HY, Kim K, Yang GM, Dayem AA and Cho SG: Correlation between oxidative stress, nutrition, and cancer initiation. Int J Mol Sci. 18:15442017. View Article : Google Scholar : PubMed/NCBI | |
|
Feno S, Butera G, Vecellio Reane D, Rizzuto R and Raffaello A: Crosstalk between calcium and ROS in pathophysiological conditions. Oxid Med Cell Longev. 2019:93240182019. View Article : Google Scholar : PubMed/NCBI | |
|
Mittler R: ROS Are Good. Trends Plant Sci. 22:11–19. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Yoneyama M, Kawada K, Gotoh Y, Shiba T and Ogita K: Endogenous reactive oxygen species are essential for proliferation of neural stem/progenitor cells. Neurochem Int. 56:740–746. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Trachootham D, Alexandre J and Huang P: Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nat Rev Drug Discov. 8:579–591. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Zoi V, Galani V, Lianos GD, Voulgaris S, Kyritsis AP and Alexiou GA: The role of curcumin in cancer treatment. Biomedicines. 9:10862021. View Article : Google Scholar : PubMed/NCBI | |
|
Nelson KM, Dahlin JL, Bisson J, Graham J, Pauli GF and Walters MA: The essential medicinal chemistry of curcumin. J Med Chem. 60:1620–1637. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Simon HU, Haj-Yehia A and Levi-Schaffer F: Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis. 5:415–418. 2000. View Article : Google Scholar : PubMed/NCBI | |
|
Abd El-Hack ME, El-Saadony MT, Swelum AA, Arif M, Abo Ghanima MM, Shukry M, Noreldin A, Taha AE and El-Tarabily KA: Curcumin, the active substance of turmeric: Its effects on health and ways to improve its bioavailability. J Sci Food Agric. 101:5747–5762. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Du Q, Hu B, An HM, Shen KP, Xu L, Deng S and Wei MM: Synergistic anticancer effects of curcumin and resveratrol in Hepa1-6 hepatocellular carcinoma cells. Oncol Rep. 29:1851–1858. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
He YC, He L, Khoshaba R, Lu FG, Cai C, Zhou FL, Liao DF and Cao D: Curcumin nicotinate selectively induces cancer cell apoptosis and cycle arrest through a P53-Mediated mechanism. Molecules. 24:41792019. View Article : Google Scholar : PubMed/NCBI | |
|
Yao H, Gong X, Geng M, Duan S, Qiao P, Sun F, Zhu Z and Du B: Cascade nanozymes based on the ‘butterfly effect’ for enhanced starvation therapy through the regulation of autophagy. Biomater Sci. 10:4008–4022. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Kotha RR and Luthria DL: Curcumin: Biological, pharmaceutical, nutraceutical, and analytical aspects. Molecules. 24:29302019. View Article : Google Scholar : PubMed/NCBI | |
|
Amalraj A, Pius A and Gopi S and Gopi S: Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives-A review. J Tradit Complement Med. 7:205–233. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Gopi S, Jacob J, Varma K, Jude S, Amalraj A, Arundhathy CA, George R, Sreeraj TR, Divya C, Kunnumakkara AB and Stohs SJ: Comparative oral absorption of curcumin in a natural turmeric matrix with two other curcumin formulations: An open-label Parallel-arm study. Phytother Res. 31:1883–1891. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Kunnumakkara AB, Bordoloi D, Padmavathi G, Monisha J, Roy NK, Prasad S and Aggarwal BB: Curcumin, the golden nutraceutical: Multitargeting for multiple chronic diseases. Br J Pharmacol. 174:1325–1348. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Manica D, Silva GBD, Silva APD, Marafon F, Maciel SFVO, Bagatini MD and Moreno M: Curcumin promotes apoptosis of human melanoma cells by caspase 3. Cell Biochem Funct. Oct 4–2023.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI | |
|
Wei H, Li X, Liu F, Li Y, Luo B, Huang X, Chen H, Wen B and Ma P: Curcumin inhibits the development of colorectal cancer via regulating the USP4/LAMP3 pathway. Naunyn Schmiedebergs Arch Pharmacol. Sep 20–2023.(Epub ahead of print). View Article : Google Scholar | |
|
Gabr SA, Elsaed WM, Eladl MA, El-Sherbiny M, Ebrahim HA, Asseri SM, Eltahir YAM, Elsherbiny N and Eldesoqui M: Curcumin modulates oxidative stress, fibrosis, and apoptosis in Drug-resistant cancer cell lines. Life (Basel). 12:14272022.PubMed/NCBI | |
|
Lin X, Wang L, Zhao L, Zhu Z, Chen T, Chen S, Tao Y, Zeng T, Zhong Y, Sun H, et al: Curcumin micelles suppress gastric tumor cell growth by upregulating ROS generation, disrupting redox equilibrium and affecting mitochondrial bioenergetics. Food Funct. 11:4146–4159. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Sharma RA, McLelland HR, Hill KA, Ireson CR, Euden SA, Manson MM, Pirmohamed M, Marnett LJ, Gescher AJ and Steward WP: Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin Cancer Res. 7:1894–1900. 2001.PubMed/NCBI | |
|
Lao CD, Ruffin MT IV, Normolle D, Heath DD, Murray SI, Bailey JM, Boggs ME, Crowell J, Rock CL and Brenner DE: Dose escalation of a curcuminoid formulation. BMC Complement Altern Med. 6:102006. View Article : Google Scholar : PubMed/NCBI | |
|
Faião-Flores F, Suarez JA, Pardi PC and Maria DA: DM-1, sodium 4-[5-(4-hydroxy-3-methoxyphenyl)-3-oxo-penta-1,4-dienyl]-2-methoxy-phenolate: A curcumin analog with a synergic effect in combination with paclitaxel in breast cancer treatment. Tumour Biol. 33:775–785. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Stohs SJ, Chen O, Ray SD, Ji J, Bucci LR and Preuss HG: Highly bioavailable forms of curcumin and promising avenues for curcumin-based research and application: A review. Molecules. 25:13972020. View Article : Google Scholar : PubMed/NCBI | |
|
Yang ZJ, Huang SY, Zhou DD, Xiong RG, Zhao CN, Fang AP, Zhang YJ, Li HB and Zhu HL: Effects and mechanisms of curcumin for the prevention and management of cancers: An updated review. Antioxidants (Basel). 11:14812022. View Article : Google Scholar : PubMed/NCBI | |
|
Freyre-Fonseca V, Delgado-Buenrostro NL, Gutiérrez-Cirlos EB, Calderón-Torres CM, Cabellos-Avelar T, Sánchez-Pérez Y, Pinzón E, Torres I, Molina-Jijón E, Zazueta CP, et al: Titanium dioxide nanoparticles impair lung mitochondrial function. Toxicol Lett. 202:111–119. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Cremers CM and Jakob U: Oxidant sensing by reversible disulfide bond formation. J Biol Chem. 288:26489–26496. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Trujillo J, Granados-Castro LF, Zazueta C, Andérica-Romero AC, Chirino YI and Pedraza-Chaverrí J: Mitochondria as a target in the therapeutic properties of curcumin. Arch Pharm (Weinheim). 347:873–884. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Mortezaee K, Salehi E, Mirtavoos-Mahyari H, Motevaseli E, Najafi M, Farhood B, Rosengren RJ and Sahebkar A: Mechanisms of apoptosis modulation by curcumin: Implications for cancer therapy. J Cell Physiol. 234:12537–12550. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Yang M, Tian H, Shen P, Xu L, Liu H, Zhu J, Wang Q and Shi Y: Curcumin alleviates nuclear factor-κB/NOD-like receptor protein 3 mediated renal injury caused by acute respiratory distress syndrome through reducing mitochondrial oxidative stress. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 35:393–397. 2023.(In Chinese). PubMed/NCBI | |
|
Feng L, Wang Y, Bi Z, Wei Z, Zhang H and Zhang S: Single-Atom nanoenzyme-based autoluminescence system for cancer cell imaging and mitochondrial-targeted therapy. ACS Appl Bio Mater. 6:5086–5096. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Lu Z, Gao Z, Song H, Zhou Y, Yuan W, Wang X, Zhang L, Hong Y, Meng Y, Hu J, et al: Synthesis, biological evaluation and action mechanism study of new mitochondria-targeted curcumin derivative as potential antitumor drugs. Chem Biodivers. 20:e2023000862023. View Article : Google Scholar : PubMed/NCBI | |
|
Sarosiek K and Wood KC: Endogenous and imposed determinants of apoptotic vulnerabilities in cancer. Trends Cancer. 9:96–110. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, Tao Y, Hu K and Lu J: GRP78 inhibitor HA15 increases the effect of Bortezomib on eradicating multiple myeloma cells through triggering endoplasmic reticulum stress. Heliyon. 9:e198062023. View Article : Google Scholar : PubMed/NCBI | |
|
Liczbiński P, Michałowicz J and Bukowska B: Molecular mechanism of curcumin action in signaling pathways: Review of the latest research. Phytother Res. 34:1992–2005. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Cheng X, Xu S, Bao J and Yu HJM: Curcumin induces endoplasmic reticulum stress-associated apoptosis in human papillary thyroid carcinoma BCPAP cells via disruption of intracellular calcium homeostasis. Medicine (Baltimore). 97:e110952018. View Article : Google Scholar : PubMed/NCBI | |
|
Goswami RS, Patel KP, Singh RR, Meric-Bernstam F, Kopetz ES, Subbiah V, Alvarez RH, Davies MA, Jabbar KJ, Roy-Chowdhuri S, et al: Hotspot mutation panel testing reveals clonal evolution in a study of 265 paired primary and metastatic tumors. Clin Cancer Res. 21:2644–2651. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Hail N Jr: Mitochondrial reactive oxygen species affect sensitivity to curcumin-induced apoptosis. Free Radic Biol Med. 44:1382–1393. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Agarwal A, Kasinathan A, Ganesan R, Balasubramanian A, Bhaskaran J, Suresh S, Srinivasan R, Aravind KB and Sivalingam N: Curcumin induces apoptosis and cell cycle arrest via the activation of reactive oxygen species-independent mitochondrial apoptotic pathway in Smad4 and p53 mutated colon adenocarcinoma HT29 cells. Nutr Res. 51:67–81. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Sritharan S and Sivalingam N: Curcumin induced apoptosis is mediated through oxidative stress in mutated p53 and wild type p53 colon adenocarcinoma cell lines. J Biochem Mol Toxicol. 35:e226162021. View Article : Google Scholar : PubMed/NCBI | |
|
Onorati AV, Dyczynski M, Ojha R and Amaravadi RK: Targeting autophagy in cancer. Cancer. 124:3307–3318. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Ashrafizadeh M, Zarrabi A, Orouei S, Kiavash Hushmandi, Hakimi A, Amirhossein Zabolian, Daneshi S, Samarghandian S, Baradaran B and Najafi M: MicroRNA-mediated autophagy regulation in cancer therapy: The role in chemoresistance/chemosensitivity. Eur J Pharmacol. 892:1736602021. View Article : Google Scholar : PubMed/NCBI | |
|
Liu LD, Pang YX, Zhao XR, Li R, Jin CJ, Xue J, Dong RY and Liu PS: Curcumin induces apoptotic cell death and protective autophagy by inhibiting AKT/mTOR/p70S6K pathway in human ovarian cancer cells. Arch Gynecol Obstet. 299:1627–1639. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Mortezaee K, Parwaie W, Motevaseli E, Mirtavoos-Mahyari H, Musa AE, Shabeeb D, Esmaely F, Najafi M and Farhood B: Targets for improving tumor response to radiotherapy. Int Immunopharmacol. 76:1058472019. View Article : Google Scholar : PubMed/NCBI | |
|
Wang J, Zhang J, Zhang CJ, Wong YK, Lim TK, Hua ZC, Liu B, Tannenbaum SR, Shen HM and Lin Q: In situ proteomic profiling of curcumin targets in HCT116 colon cancer cell line. Sci Rep. 6:221462016. View Article : Google Scholar : PubMed/NCBI | |
|
Liang HH, Huang CY, Chou CW, Makondi PT, Huang MT, Wei PL and Chang YJ: Heat shock protein 27 influences the anti-cancer effect of curcumin in colon cancer cells through ROS production and autophagy activation. Life Sci. 209:43–51. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Wang T, Wu X, Al Rudaisat M, Song Y and Cheng H: Curcumin induces G2/M arrest and triggers autophagy, ROS generation and cell senescence in cervical cancer cells. J Cancer. 11:6704–6715. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Chang M, Shang M, Yuan F, Guo W and Wang C: EF24 exerts cytotoxicity against NSCLC via inducing ROS accumulation. Cancer Cell Int. 21:5312021. View Article : Google Scholar : PubMed/NCBI | |
|
Mou Y, Wang J, Wu J, He D, Zhang C, Duan C and Li B: Ferroptosis, a new form of cell death: Opportunities and challenges in cancer. J Hematol Oncol. 12:342019. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Li XM, Shi XH, Ye K, Fu XL, Wang X, Guo SM, Ma JQ, Xu FF, Sun HM, et al: Sorafenib triggers ferroptosis via inhibition of HBXIP/SCD axis in hepatocellular carcinoma. Acta Pharmacol Sin. 44:622–634. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Tang X, Li Y, Zhao J, Liang L, Zhang K, Zhang X, Yu H and Du H: Heme oxygenase-1 increases intracellular iron storage and suppresses inflammatory response of macrophages by inhibiting M1 polarization. Metallomics. 15:mfad0622023. View Article : Google Scholar : PubMed/NCBI | |
|
Giorgi G, Mascaró M, Gandini N, Rabassa ME, Coló GP, Arévalo J, Curino AC, Facchinetti MM and Roque ME: Iron cycle disruption by heme oxygenase-1 activation leads to a reduced breast cancer cell survival. Biochim Biophys Acta Mol Basis Dis. 1869:1666212023. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Y, Guo X, Zeng Y, Mo X, Hong S, He H, Li J, Fatima S and Liu Q: Oxidative stress induces mitochondrial iron overload and ferroptotic cell death. Sci Rep. 13:155152023. View Article : Google Scholar : PubMed/NCBI | |
|
Li R, Zhang J, Zhou Y, Gao Q, Wang R, Fu Y, Zheng L and Yu H: Transcriptome investigation and in vitro verification of curcumin-induced HO-1 as a feature of ferroptosis in breast cancer cells. Oxid Med Cell Longev. 2020:34698402020. View Article : Google Scholar : PubMed/NCBI | |
|
Suttner DM and Dennery PA: Reversal of HO-1 related cytoprotection with increased expression is due to reactive iron. FASEB J. 13:1800–1809. 1999. View Article : Google Scholar : PubMed/NCBI | |
|
McBean GJ: The transsulfuration pathway: A source of cysteine for glutathione in astrocytes. Amino Acids. 42:199–205. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou XL, Zhu CY, Wu ZG, Guo X and Zou W: The oncoprotein HBXIP competitively binds KEAP1 to activate NRF2 and enhance breast cancer cell growth and metastasis. Oncogene. 38:4028–4046. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Cao X, Li Y, Wang Y, Yu T, Zhu C, Zhang X and Guan J: Curcumin suppresses tumorigenesis by ferroptosis in breast cancer. PLoS One. 17:e02613702022. View Article : Google Scholar : PubMed/NCBI | |
|
Tang X, Ding H, Liang M, Chen X, Yan Y, Wan N, Chen Q, Zhang J and Cao J: Curcumin induces ferroptosis in non-small-cell lung cancer via activating autophagy. Thorac Cancer. 12:1219–1230. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Shi J, Gao W and Shao F: Pyroptosis: Gasdermin-Mediated programmed necrotic cell death. Trends Biochem Sci. 42:245–254. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Frank D and Vince JE: Pyroptosis versus necroptosis: Similarities, differences, and crosstalk. Cell Death Differ. 26:99–114. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Liang WF, Gong YX, Li HF, Sun FL, Li WL, Chen DQ, Xie DP, Ren CX, Guo XY, Wang ZY, et al: Curcumin activates ROS signaling to promote pyroptosis in hepatocellular carcinoma HepG2 cells. In Vivo. 35:249–257. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Wei T, Zheng Z, Wei X, Liu Y, Li W, Fang B, Yun D, Dong Z, Yi B, Li W, et al: Rational design, synthesis, and pharmacological characterisation of dicarbonyl curcuminoid analogues with improved stability against lung cancer via ROS and ER stress mediated cell apoptosis and pyroptosis. J Enzyme Inhib Med Chem. 37:2357–2369. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Li AN, Li S, Zhang YJ, Xu XR, Chen YM and Li HB: Resources and biological activities of natural polyphenols. Nutrients. 6:6020–6047. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Wu H, Chen L, Zhu F, Han X, Sun L and Chen K: The cytotoxicity effect of resveratrol: Cell cycle arrest and induced apoptosis of breast cancer 4T1 cells. Toxins (Basel). 11:7312019. View Article : Google Scholar : PubMed/NCBI | |
|
Tanaka T, Aoki R and Terasaki M: Potential chemopreventive effects of dietary combination of phytochemicals against cancer development. Pharmaceuticals (Basel). 16:15912023. View Article : Google Scholar : PubMed/NCBI | |
|
Arena A, Romeo MA, Benedetti R, Masuelli L, Bei R, Gilardini Montani MS and Cirone M: New insights into curcumin- and resveratrol-mediated anti-cancer effects. Pharmaceuticals (Basel). 14:10682021. View Article : Google Scholar : PubMed/NCBI | |
|
Cho CJ, Yang CW, Wu CL, Ho JY, Yu CP, Wu ST and Yu DS: The modulation study of multiple drug resistance in bladder cancer by curcumin and resveratrol. Oncol Lett. 18:6869–6876. 2019.PubMed/NCBI | |
|
Xia RL, Lu Y, Zhu LN, Zhang SF, Zhao FK and Fu CY: Different regulatory pathways are involved in the proliferative inhibition of two types of leukemia cell lines induced by paclitaxel. Oncol Rep. 30:1853–1859. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Rosière R, Van Woensel M, Mathieu V, Langer I, Mathivet T, Vermeersch M, Amighi K and Wauthoz N: Development and evaluation of well-tolerated and tumor-penetrating polymeric micelle-based dry powders for inhaled anti-cancer chemotherapy. Int J Pharm. 501:148–159. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Lee WH, Loo CY, Traini D and Young PM: Development and evaluation of paclitaxel and curcumin dry powder for inhalation lung cancer treatment. Pharmaceutics. 13:92020. View Article : Google Scholar : PubMed/NCBI | |
|
Lee WH, Bebawy M, Loo CY, Luk F, Mason RS and Rohanizadeh R: Fabrication of curcumin micellar nanoparticles with enhanced anti-cancer activity. J Biomed Nanotechnol. 11:1093–1105. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Lee WH, Loo CY, Ong HX, Traini D, Young PM and Rohanizadeh R: Synthesis and characterization of inhalable flavonoid nanoparticle for lung cancer cell targeting. J Biomed Nanotechnol. 12:371–386. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Syng-Ai C, Kumari AL and Khar A: Effect of curcumin on normal and tumor cells: Role of glutathione and bcl-2. Mol Cancer Ther. 3:1101–1108. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Tapia E, Zatarain-Barrón ZL, Hernández-Pando R, Zarco-Márquez G, Molina-Jijón E, Cristóbal-García M, Santamaría J and Pedraza-Chaverri J: Curcumin reverses glomerular hemodynamic alterations and oxidant stress in 5/6 nephrectomized rats. Phytomedicine. 20:359–366. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Serafini MM, Catanzaro M, Fagiani F, Simoni E, Caporaso R, Dacrema M, Romanoni I, Govoni S, Racchi M, Daglia M, et al: Modulation of Keap1/Nrf2/ARE signaling pathway by Curcuma- and Garlic-Derived Hybrids. Front Pharmacol. 10:15972020. View Article : Google Scholar : PubMed/NCBI | |
|
Waghray D and Zhang Q: Inhibit or evade multidrug resistance P-Glycoprotein in cancer treatment. J Med Chem. 61:5108–5121. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Attia YM, El-Kersh DM, Ammar RA, Adel A, Khalil A, Walid H, Eskander K, Hamdy M, Reda N, Mohsen NE, et al: Inhibition of aldehyde dehydrogenase-1 and p-glycoprotein-mediated multidrug resistance by curcumin and vitamin D3 increases sensitivity to paclitaxel in breast cancer. Chem Biol Interact. 315:1088652020. View Article : Google Scholar : PubMed/NCBI | |
|
Ebrahimifar M, Hasanzadegan Roudsari M, Kazemi SM, Ebrahimi Shahmabadi H, Kanaani L, Alavi SA and Izadi Vasfi M: Enhancing effects of curcumin on cytotoxicity of paclitaxel, methotrexate and vincristine in gastric cancer cells. Asian Pac J Cancer Prev. 18:65–68. 2017.PubMed/NCBI | |
|
Zhang N, Gao M, Wang Z, Zhang J, Cui W, Li J, Zhu X, Zhang H, Yang DH and Xu X: Curcumin reverses doxorubicin resistance in colon cancer cells at the metabolic level. J Pharm Biomed Anal. 201:1141292021. View Article : Google Scholar : PubMed/NCBI | |
|
Firouzi Amoodizaj F, Baghaeifar S, Taheri E, Farhoudi Sefidan Jadid M, Safi M, Seyyed Sani N, Hajazimian S, Isazadeh A and Shanehbandi D: Enhanced anticancer potency of doxorubicin in combination with curcumin in gastric adenocarcinoma. J Biochem Mol Toxicol. 34:e224862020. View Article : Google Scholar : PubMed/NCBI | |
|
Dhandapani KM, Mahesh VB and Brann DW: Curcumin suppresses growth and chemoresistance of human glioblastoma cells via AP-1 and NFkappaB transcription factors. J Neurochem. 102:522–538. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Abbaspour H and Afshar AS: Curcumin inhibits the expression of ornithine decarboxylase and adenosine deaminase genes in MCF-7 human breast cancer cells. Arch Biol Sci. 70:639–645. 2018. View Article : Google Scholar | |
|
Lee WJ, Jo JH, Jang SI, Jung EJ, Hwang JM, Bae JW, Ha JJ, Kim DH and Kwon WS: The natural flavonoid compound deguelin suppresses sperm (Sus Scrofa) functions through abnormal activation of the PI3K/AKT pathway. Reprod Toxicol. 120:1084262023. View Article : Google Scholar : PubMed/NCBI | |
|
Lin ZY, Yun QZ, Wu L, Zhang TW and Yao TZ: Pharmacological basis and new insights of deguelin concerning its anticancer effects. Pharmacol Res. 174:1059352021. View Article : Google Scholar : PubMed/NCBI | |
|
Russell DA, Bridges HR, Serreli R, Kidd SL, Mateu N, Osberger TJ, Sore HF, Hirst J and Spring DR: Hydroxylated rotenoids selectively inhibit the proliferation of prostate cancer cells. J Nat Prod. 83:1829–1845. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Kocdor MA, Cengiz H, Ates H and Kocdor H: Inhibition of cancer stem-like phenotype by curcumin and deguelin in CAL-62 anaplastic thyroid cancer cells. Anticancer Agents Med Chem. 19:1887–1898. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Liang G, Shao L, Wang Y, Zhao C, Chu Y, Xiao J, Zhao Y, Li X and Yang S: Exploration and synthesis of curcumin analogues with improved structural stability both in vitro and in vivo as cytotoxic agents. Bioorg Med Chem. 17:2623–2631. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Zou P, Zhang J, Xia Y, Kanchana K, Guo G, Chen W, Huang Y, Wang Z, Yang S and Liang G: ROS generation mediates the anti-cancer effects of WZ35 via activating JNK and ER stress apoptotic pathways in gastric cancer. Oncotarget. 6:5860–5876. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Wang L, Han L, Tao Z, Zhu Z, Han L, Yang Z, Wang H, Dai D, Wu L, Yuan Z and Chen T: The curcumin derivative WZ35 activates ROS-dependent JNK to suppress hepatocellular carcinoma metastasis. Food Funct. 9:2970–2978. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Wang L, Wang C, Tao Z, Zhao L, Zhu Z, Wu W, He Y, Chen H, Zheng B, Huang X, et al: Curcumin derivative WZ35 inhibits tumor cell growth via ROS-YAP-JNK signaling pathway in breast cancer. J Exp Clin Cancer Res. 38:4602019. View Article : Google Scholar : PubMed/NCBI | |
|
Koroth J, Nirgude S, Tiwari S, Gopalakrishnan V, Mahadeva R, Kumar S, Karki SS and Choudhary B: Investigation of anti-cancer and migrastatic properties of novel curcumin derivatives on breast and ovarian cancer cell lines. BMC Complement Altern Med. 19:2732019. View Article : Google Scholar : PubMed/NCBI | |
|
Koroth J, Mahadeva R, Ravindran F, Parashar TR, Teja V, Karki SS and Choudhary B: Curcumin derivative 1, 2-bis [(3E, 5E)-3, 5-bis [(2-chlorophenyl) methylene]-4-oxo-1-piperidyl] ethane-1, 2-dione (ST03) induces mitochondria mediated apoptosis in ovarian cancer cells and inhibits tumor progression in EAC mouse model. Transl Oncol. 15:1012802022. View Article : Google Scholar : PubMed/NCBI | |
|
Haseli S, Pourmadadi M, Samadi A, Yazdian F, Abdouss M, Rashedi H and Navaei-Nigjeh M: A novel pH-responsive nanoniosomal emulsion for sustained release of curcumin from a chitosan-based nanocarrier: Emphasis on the concurrent improvement of loading, sustained release, and apoptosis induction. Biotechnol Prog. 38:e32802022. View Article : Google Scholar : PubMed/NCBI | |
|
Hou CH, Lin FL, Hou SM and Liu JF: Hyperthermia induces apoptosis through endoplasmic reticulum and reactive oxygen species in human osteosarcoma cells. Int J Mol Sci. 15:17380–17395. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Pazouki N, Irani S, Olov N, Atyabi SM and Bagheri-Khoulenjani S: Fe3O4 nanoparticles coated with carboxymethyl chitosan containing curcumin in combination with hyperthermia induced apoptosis in breast cancer cells. Prog Biomater. 11:43–54. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Freidus LG, Kumar P, Marimuthu T, Pradeep P and Choonara YE: Theranostic Mesoporous Silica Nanoparticles Loaded With a Curcumin-Naphthoquinone Conjugate for Potential Cancer Intervention. Front Mol Biosci. 8:6707922021. View Article : Google Scholar : PubMed/NCBI | |
|
Freidus LG, Kumar P, Marimuthu T, Pradeep P, Pillay V and Choonara YE: Synthesis and Properties of CurNQ for the theranostic application in ovarian cancer intervention. Molecules. 25:44712020. View Article : Google Scholar : PubMed/NCBI | |
|
Zholobak NM, Shcherbakov AB, Ivanova OS, Reukov V, Baranchikov AE and Ivanov VK: Nanoceria-curcumin conjugate: Synthesis and selective cytotoxicity against cancer cells under oxidative stress conditions. J Photochem Photobiol B. 209:1119212020. View Article : Google Scholar : PubMed/NCBI | |
|
Haider T, Pandey V, Banjare N, Gupta PN and Soni V: Drug resistance in cancer: Mechanisms and tackling strategies. Pharmacol Rep. 72:1125–1151. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Li L, Wang F, Jia X, Yao L and Liu Y: Research mechanism and progress of the natural compound curcumin in treating Alzheimer´s disease. Mini Rev Med Chem. Oct 30–2023.(Epub ahead of print). View Article : Google Scholar | |
|
Molani-Gol R, Dehghani A and Rafraf M: Effects of curcumin/turmeric supplementation on the liver enzymes, lipid profiles, glycemic index, and anthropometric indices in non-alcoholic fatty liver patients: An umbrella meta-analysis. Phytother Res. Nov 2–2023.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI | |
|
Wu WS, Wu JR and Hu CT: Signal cross talks for sustained MAPK activation and cell migration: The potential role of reactive oxygen species. Cancer Metastasis Rev. 27:303–314. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Das L and Vinayak M: Long term effect of curcumin in restoration of tumour suppressor p53 and phase-II antioxidant enzymes via activation of Nrf2 signalling and modulation of inflammation in prevention of cancer. PLoS One. 10:e01240002015. View Article : Google Scholar : PubMed/NCBI | |
|
Birben E, Sahiner UM, Sackesen C, Erzurum S and Kalayci O: Oxidative stress and antioxidant defense. World Allergy Organ J. 5:9–19. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Lin X, Bai D, Wei Z, Zhang Y, Huang Y, Deng H and Huang X: Curcumin attenuates oxidative stress in RAW264.7 cells by increasing the activity of antioxidant enzymes and activating the Nrf2-Keap1 pathway. PLoS One. 14:e02167112019. View Article : Google Scholar : PubMed/NCBI | |
|
Ghosh P, Vidal C, Dey S and Zhang L: Mitochondria targeting as an effective strategy for cancer therapy. Int J Mol Sci. 21:33632020. View Article : Google Scholar : PubMed/NCBI | |
|
Guo YJ, Pan WW, Liu SB, Shen ZF, Xu Y and Hu LL: ERK/MAPK signalling pathway and tumorigenesis. Exp Ther Med. 19:1997–2007. 2020.PubMed/NCBI | |
|
Wu MF, Huang YH, Chiu LY, Cherng SH, Sheu GT and Yang TY: Curcumin induces apoptosis of chemoresistant lung cancer cells via ROS-Regulated p38 MAPK phosphorylation. Int J Mol Sci. 23:82482022. View Article : Google Scholar : PubMed/NCBI | |
|
He F, Antonucci L and Karin M: NRF2 as a regulator of cell metabolism and inflammation in cancer. Carcinogenesis. 41:405–416. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Karthikeyan A, Senthil N and Min T: Nanocurcumin: A promising candidate for therapeutic applications. Front Pharmacol. 11:4872020. View Article : Google Scholar : PubMed/NCBI | |
|
Safavy A, Raisch KP, Mantena S, Sanford LL, Sham SW, Krishna NR and Bonner JA: Design and development of water-soluble curcumin conjugates as potential anticancer agents. J Med Chem. 50:6284–6288. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Sharma RA, Euden SA, Platton SL, Cooke DN, Shafayat A, Hewitt HR, Marczylo TH, Morgan B, Hemingway D, Plummer SM, et al: Phase I clinical trial of oral curcumin: biomarkers of systemic activity and compliance. Clin Cancer Res. 10:6847–6854. 2004. View Article : Google Scholar : PubMed/NCBI |