Inhibitor of Wnt receptor 1 suppresses the effects of Wnt1, Wnt3a and β‑catenin on the proliferation and migration of C6 GSCs induced by low‑dose radiation
- Authors:
- Published online on: December 12, 2023 https://doi.org/10.3892/or.2023.8681
- Article Number: 22
-
Copyright: © Yan et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Gliomas are the most common malignant brain tumors in the central nervous system with multiple histologic subtypes and malignancy grades (1,2). Despite advances in treatment strategies that combine surgical resections with radiotherapy and chemotherapy (3,4), the results of combined therapies have not efficiently reduced the high mortality rate of glioma (5,6). For instance, malignant gliomas originating from astrocytoma are characterized by a dismal prognosis, frequent recurrence and a low five-year survival rate (3,5,7,8), and all of these factors have become major hurdles to the effective treatment of gliomas. Therefore, it is important to develop innovative and effective treatments for gliomas. Stem cells may be utilized to design novel treatments for malignant gliomas (9,10).
Accumulated studies have indicated that gliomas demonstrate considerable clonal evolution through sequential selection by the tumor microenvironment (11,12) and contain a population of glioma stem cells (GSCs) that exhibit stem-like characteristics, including maintained proliferation, self-renewal and differentiation (13,14). Increasing evidence has suggested that GSCs play critical roles in tumor initiation, progression, prognosis and resistance to conventional chemoradiotherapy (15–17). The remarkable oncogenic complexities of GSCs have generated overwhelming interest in improved defining and characterizing these cells and determining novel pathways driving glioma progression and recurrence that could lead to effective therapeutic targets.
Wnt/β-catenin signaling plays a key role in regulating the tumorigenesis and development of gliomas (18,19). The aberrant activation of Wnt/β-catenin signaling is involved in the chemo-radioresistance of glioma, while it is also crucial for the maintenance of GSCs (20). Activation of Wnt/β-catenin signaling was also reported to promote radioresistance in glioma cells. Cyclophilin A was shown to facilitate stemness, self-renewal and radioresistance of GSCs by activating Wnt/β-catenin signaling (21). In addition, Wnt/beta-catenin signaling is involved in regulating the proliferation and apoptosis of glioma cells (22).
In the present study, the expression of Wnt1, Wnt3a and β-catenin was examined and it was identified that they were upregulated in the proliferation and migration of C6 GSCs after low-dose radiation (LDR). Accordingly, it was deduced that activated Wnt/β-catenin signaling is closely associated with the proliferation and migration of GSCs. The results of pretreatment with Wnt receptor 1 (IWR1) indicated that IWR1 can specifically block or inhibit the proliferation and migration of C6 GSCs induced by LDR, which suggests that IWR1 is an effective radiosensitizer of GSCs that can be exploited and utilized.
Materials and methods
Cell culture
For culture preparation of C6 GSCs, C6 glioma cells were adherently cultured in a humidified incubator with 5% CO2/95% air at 37°C with DMEM/F12 1:1 medium (cat. no. 11330-032; Gibco; Thermo Fisher Scientific, Inc.) containing 10% fetal bovine serum (MilliporeSigma) after freeze thawing. A total of ~2 or 3 days later, living C6 glioma cells were dispersed completely with enzymatic digestion of trypsin solution (cat. no. T-4799; MilliporeSigma), and the cell suspension was centrifuged for 5 min at 518 × g. The supernatant was discarded, and the cell pellet was resuspended and seeded at a density of 0.5×105 cells in a T 25 cm2 culture flask in serum-free DMEM/F12 1:1 medium with 2% B27 (cat. no. 10587-010; Invitrogen; Thermo Fisher Scientific, Inc.), 20 ng/ml recombinant human epidermal growth factor (cat. no. 450-02; PeproTech, Inc.), and 20 ng/ml basic fibroblast growth factor (cat. no. 100-18B; PeproTech, Inc.). The suspension of cultured C6 clonal spheres gradually formed and grew with increasing cell numbers and was passaged for ~4 or 5 days. After the third passage, the clonal spheres were ready to conduct subsequent experiments.
BrdU incorporation and identification
For neurochemical identification of C6 GSC properties, a 5-bromodeoxyuridine (BrdU) incorporation experiment was performed in suspension-cultured C6 clonal spheres for ~3 or 4 days. BrdU (MilliporeSigma) dissolved in culture medium was added at 10 µM and incubated for 24–48 h. Then, the C6 clonal spheres were seeded on coverslips coated with poly-L-lysine (cat. no. P6282; MilliporeSigma) and incubated for 2–4 h in a humidified incubator with 5% CO2/95% air at 37°C. The C6 clonal spheres were fixed with 4% paraformaldehyde for 40 min at room temperature (RT). After being completely rinsed with 0.01 M PBS, the C6 clonal spheres were pretreated with 2 N HCl for 1 h at 37°C and boric acid buffer (pH 8.5) for 10 min at RT. BrdU-incorporated cells were visualized by an immunostaining protocol (23). Briefly, cells were incubated with blocking solution 0.3% Triton X-100 containing 1% donkey serum (cat. no. V11307; Invitrogen; Thermo Fisher scientific, Inc.) for 30 min and sequentially incubated with a primary antibody mixture of mouse anti-BrdU (1:500; cat. no. MAB3262F; Chemicon International; Thermo Fisher Scientific, Inc.) and goat anti-CD133 (1:200; cat. no. sc23797; Santa Cruz Biotechnology, Inc.) at 4°C overnight and a secondary antibody mixture of fluorescein-conjugated donkey anti-mouse IgG Alexa Fluor 488 (1:2,000; cat. no. R37114; Invitrogen; Thermo Fisher Scientific, Inc.) and rhodamine-conjugated donkey anti-goat IgG 594 (1:2,000; cat. no. 43R-GD001RD; Biosynth Ltd.) for 4 h at RT. The cell coverslips were mounted with Fluorescence-preserving VECTASHIELD mounting medium (cat. no. H-1000; Vector Laboratories, Inc.) and examined under a laser scanning confocal microscope (FV-1000; Olympus Corporation).
Low-dose ionizing radiation and IWR1 pretreatment
To determine the biological responses of C6 GSCs treated with low-dose ionizing radiation, cell experiments were designed with a control, 0.3, 0.3 Gy + IWR1 and 3.0 Gy radiation groups. Before low-dose ionizing radiation, C6 GSCs were pretreated with 10 µmol/l IWR1 (cat. no. S7086; Selleck Chemicals) for 15–30 min in the low-dose (0.3 Gy) + IWR1 group. The low-dose ionizing radiation experiments were conducted according to a previously described method (24). Ionizing radiation was performed with a 6 MV X-ray of a Varian 23 EX linear accelerator. A radiation dose of 0.3 Gy or 3.0 Gy was administered at a dose rate of 3.0 Gy/min. After they were subjected to radiation, C6 clonal spheres were sequentially cultured for 48 h and harvested to conduct flow cytometry and western blot experiments. A small number of clonal spheres were seeded on coverslips and incubated in serum-containing culture medium for 24 h to conduct the morphology and immunocytochemistry experiments.
Flow cytometric analysis
The harvested C6 clonal spheres were dispersed with Accutase (cat. no. A1110501; Gibco; Thermo Fisher Scientific, Inc.) for 5–8 min and mechanically dissociated into a single cell. Cell suspensions were adjusted to a cell density of 5×105/cm2 or 1×106/cm2. Flow cytometric analysis (CytoFLEX S; Beckman Coulter, Inc.) was performed to detect cell survival, necrosis and apoptosis by using an Annexin V/PI staining kit (BB-4101; BestBio Science). The distributions of cell survival, necrosis, early apoptosis and late apoptosis are shown with dot images and percentage data.
Immunocytochemistry
Adherent cells migrated from C6 clonal spheres and were fixed with 4% paraformaldehyde for 15 min at RT. The fixed cells were incubated at 4°C overnight with primary antibodies, including rabbit anti-γ-H2AX (1:2,000; cat. no. ab11174; Abcam), rabbit anti-Wnt1 (1:3,000; cat. no. ab15151; Abcam), rabbit anti-Wnt3a (1:2,000; cat. no. ab28472; Abcam) and rabbit anti-β-catenin (1:5,000; cat. no. C2206; MiliporeSigma). Next, the cell coverslips were incubated with the appropriate secondary antibodies donkey anti-rabbit IgG-Alexa Fluor 488 (1:3,000; cat. no. R37118; Invitrogen; Thermo Fisher Scientific, Inc.) and rhodamine-conjugated donkey anti-goat IgG 594 (1:2,000; cat. no. 43R-GD001RD; Biosynth Ltd.) for 2–4 h at RT. Hoechst 33342 (MilliporeSigma) was used to stain all nuclei. Cell coverslips were mounted onto slides and observed under a laser scanning confocal microscope.
Western blot analysis
The harvested cells were treated with RIPA lysis buffer (CW2334S; CWBIO) for 30 min (on ice), collected and centrifuged at 6,600 × g for 10–15 min at 4°C. The supernatant was collected, and the total protein concentration was estimated using a BCA assay. Total proteins (20–30 µg) were loaded in each well and separated by 10 and 12% SDS-polyacrylamide gel electrophoresis, transferred to PVDF membranes, blocked with 5% milk or bovine serum albumin (MilliporeSigma) at RT for 1 h, and incubated overnight at 4°C with the following primary antibodies: γ-H2AX (1:2,000), Wnt1 (1:3,000), Wnt3a (1:2,000) and β-catenin (1:5,000). The following day, they were incubated with HRP-conjugated mouse anti-rabbit IgG secondary antibody (1:3,000; cat. no. sc-2357; Santa Cruz Biotenchnology, Inc.) for 1 h at RT. The protein bands were visualized by incubating the membranes in Ultra High Sensitivity ECL substrate solution (cat. no. ab133409; Abcam). The bands were analyzed and documented using ImageJ 2.0 software (National Institutes of Health) and GraphPad Prism 5.0 software (Dotmatics).
Statistical analysis
All data are presented as the mean ± SEM. Statistical comparisons between or among groups were performed using Student's t-tests and one-way ANOVA followed by Dunnett's post hoc test. The data were collected from three independent experiments and analyzed using GraphPad Prism 5.0 software (Dotmatics). The difference was considered statistically significant when P<0.05.
Results
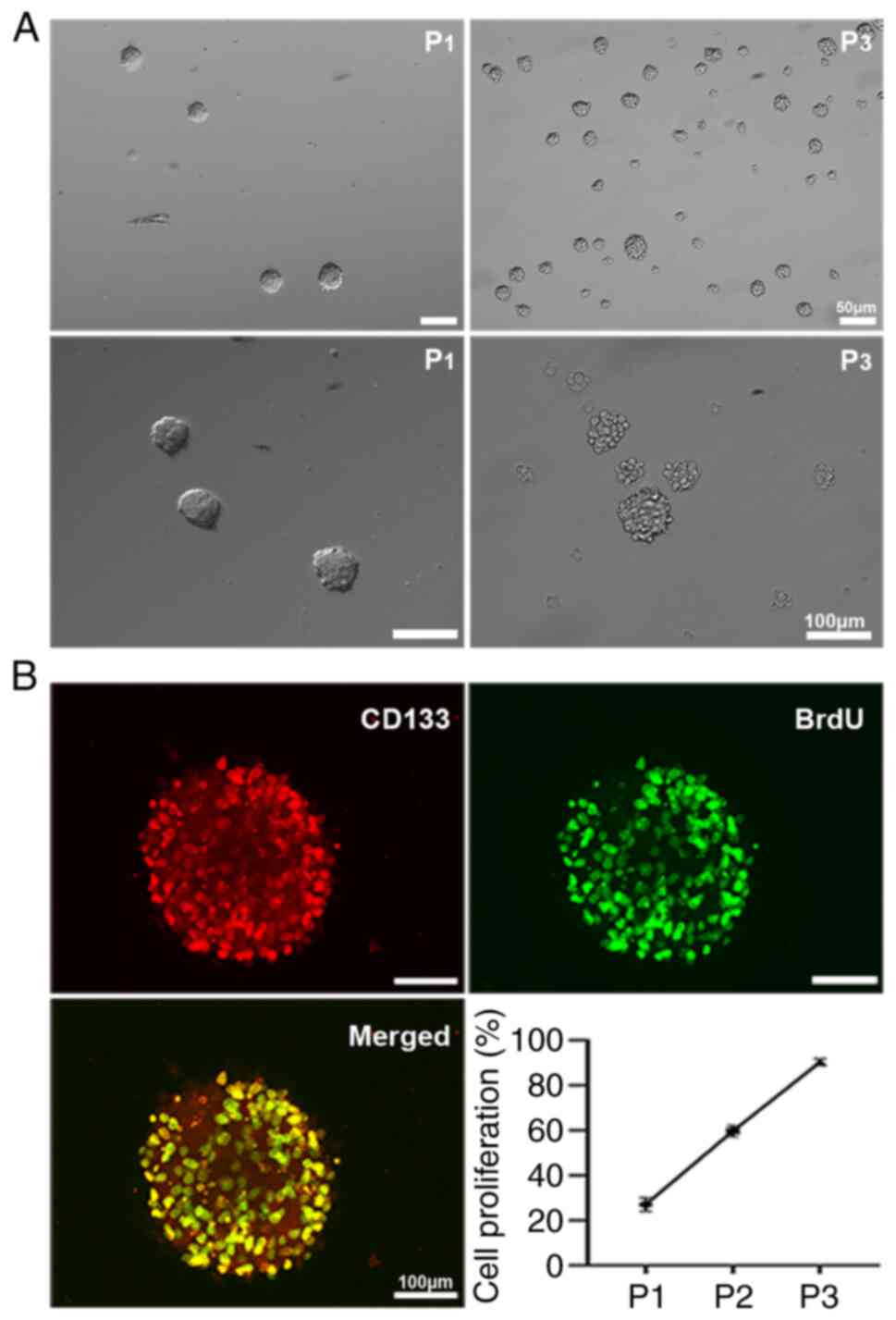
C6 GSCs show self-renewal and strong proliferation
The self-renewing and proliferative capacities of C6 GSCs were investigated in vitro. When third-passage C6 GSCs from C6 glioma cell lines were cultured in serum-free conditioned medium, it was found that non-adherent cultured C6 GSCs were large and rounded with strong light refraction. C6 GSCs gathered into clonal spheres and expanded rapidly. The passaged clonal spheres had the same self-renewing and proliferative abilities as the first-passage C6 GSCs (Fig. 1A). The clonal spheres were clearly observed and examined by double immunofluorescence staining for incorporation of the proliferation indicators BrdU and CD133 (a cancer stem cell marker), and the results revealed that both BrdU and CD133 were positively expressed on the cell nuclei. The statistical data indicated that the ratio of BrdU/CD133-positive cells reached 90% (Fig. 1B).
The characteristics of proliferative and migratory C6 GSCs pretreated with IWR1 after LDR
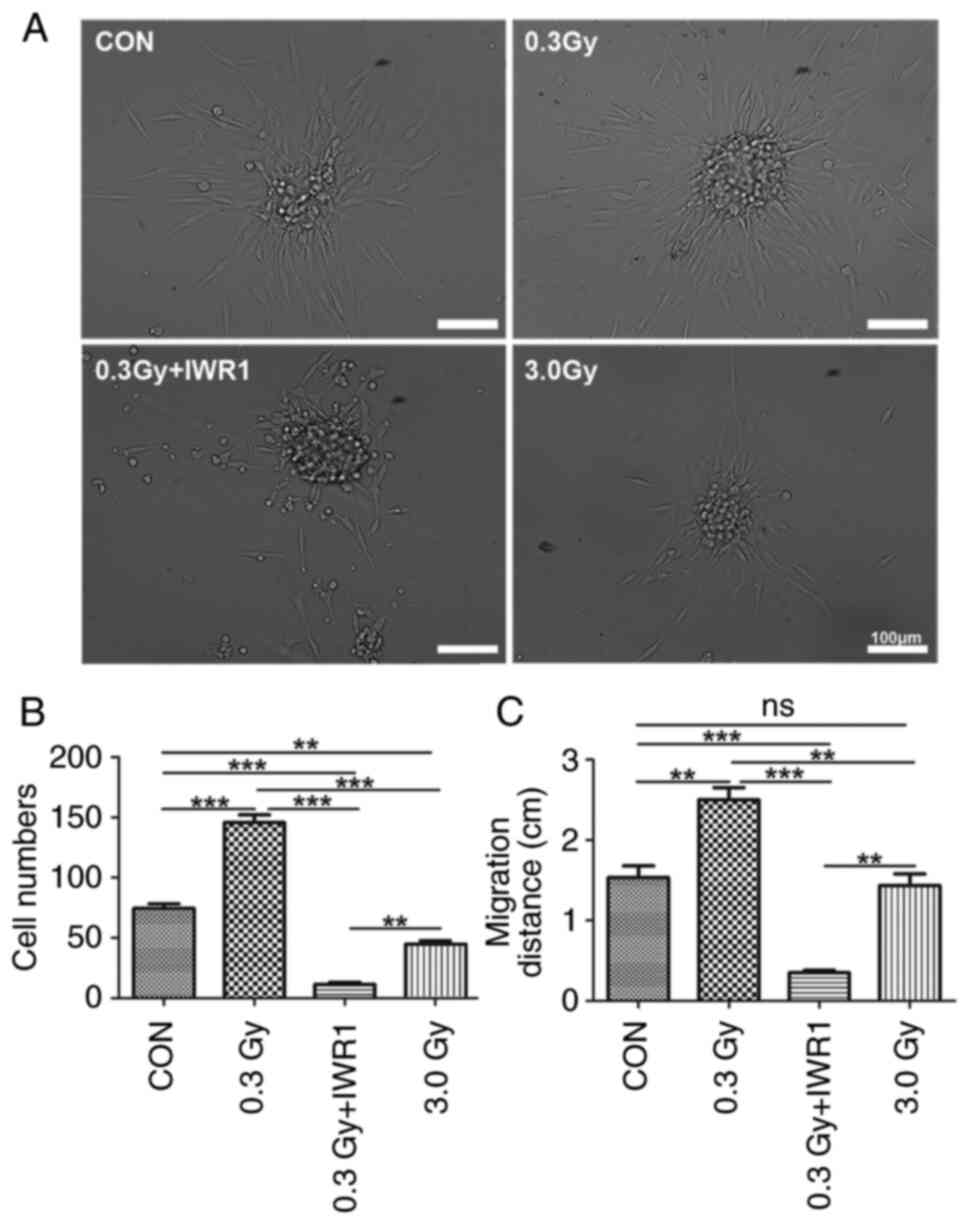
To observe C6 GSC proliferation and migration, the cells were treated with IWR1 after LDR for 48 h. The clonal spheres completely adhered to the coverslips at 4 h, and gradually, treated cells surrounding the clonal spheres in the 0.3 Gy group spread out over long distances, migrated and distributed completely for 24–48 h. However, for treated cells in the 3.0 Gy group, the cell protrusions shortened, and cell migration over a long distance remained stagnant. The proliferation of C6 GSCs pretreated with IWR1 after LDR significantly decreased, cell numbers markedly decreased, and the capability of C6 GSCs to form clonal spheres notably decreased compared with that of the control group (Fig. 2A). The statistical analysis demonstrated that the cell numbers of single clonal spheres and the migration distances of C6 GSCs in the treated groups were significantly different compared with those in the untreated group (Fig. 2B and C).
Enhanced cell survival and proliferation of C6 GSCs after LDR, which can be blocked by IWR1
The clonal spheres were subjected to LDR after pretreatment with 10 µmol/l IWR1 for 15–30 min and continued to be cultured for 48 h, after which they were collected and subjected to flow cytometric analysis for cell survival and apoptosis. The percentages of cell survival, necrosis, and early and late apoptosis are presented in the dot distribution image (Fig. 3A). Quantitative data revealed that the 0.3 Gy group exhibited increased cell survival and decreased apoptotic or necrotic cell death (P<0.001 vs. the control), while decreased cell survival and increased late apoptosis were observed in the 3.0 Gy group (P<0.05 or P<0.001 vs. the control). However, the percentages of cell survival and apoptotic and necrotic cells significantly decreased in the 0.3 Gy + IWR1 group (P<0.001 vs. 0.3 Gy). These results indicated that the enhanced cell survival and proliferation of C6 GSCs induced by LDR could be blocked by pretreatment with 10 µmol/l IWR1 (Fig. 3B).
High expression of γ-H2AX on C6 GSCs pretreated with IWR1 after LDR
To evaluate the effect of LDR, staining for γ-H2AX, a DNA damage marker, was performed. The fluorescence intensity of the γ-H2AX-positive cells was revealed after LDR. The results revealed a significant increase in the fluorescence intensity of γ-H2AX-positive cells after LDR (Fig. 4A). The fluorescence intensity of γ-H2AX-positive cells in the 0.3 and 3.0 Gy groups showed distinct differences (P<0.001 vs. the control). The fluorescence intensity of γ-H2AX-positive cells in the 3.0 Gy group demonstrated a clear difference (P<0.01 vs. 0.3 Gy). In addition, the fluorescence intensity of the γ-H2AX-positive cells in the 0.3 Gy + IWR1 group was different from that of the 0.3 Gy group (P<0.05) (Fig. 4B). Western blot analysis indicated that the expression of γ-H2AX protein was upregulated after LDR and that γ-H2AX high fluorescence intensity was positively related to radiation dose, which was not blocked by IWR1 (Fig. 4C and D).
Expression of Wnt1 in migrating C6 GSCs pretreated with IWR1 after LDR
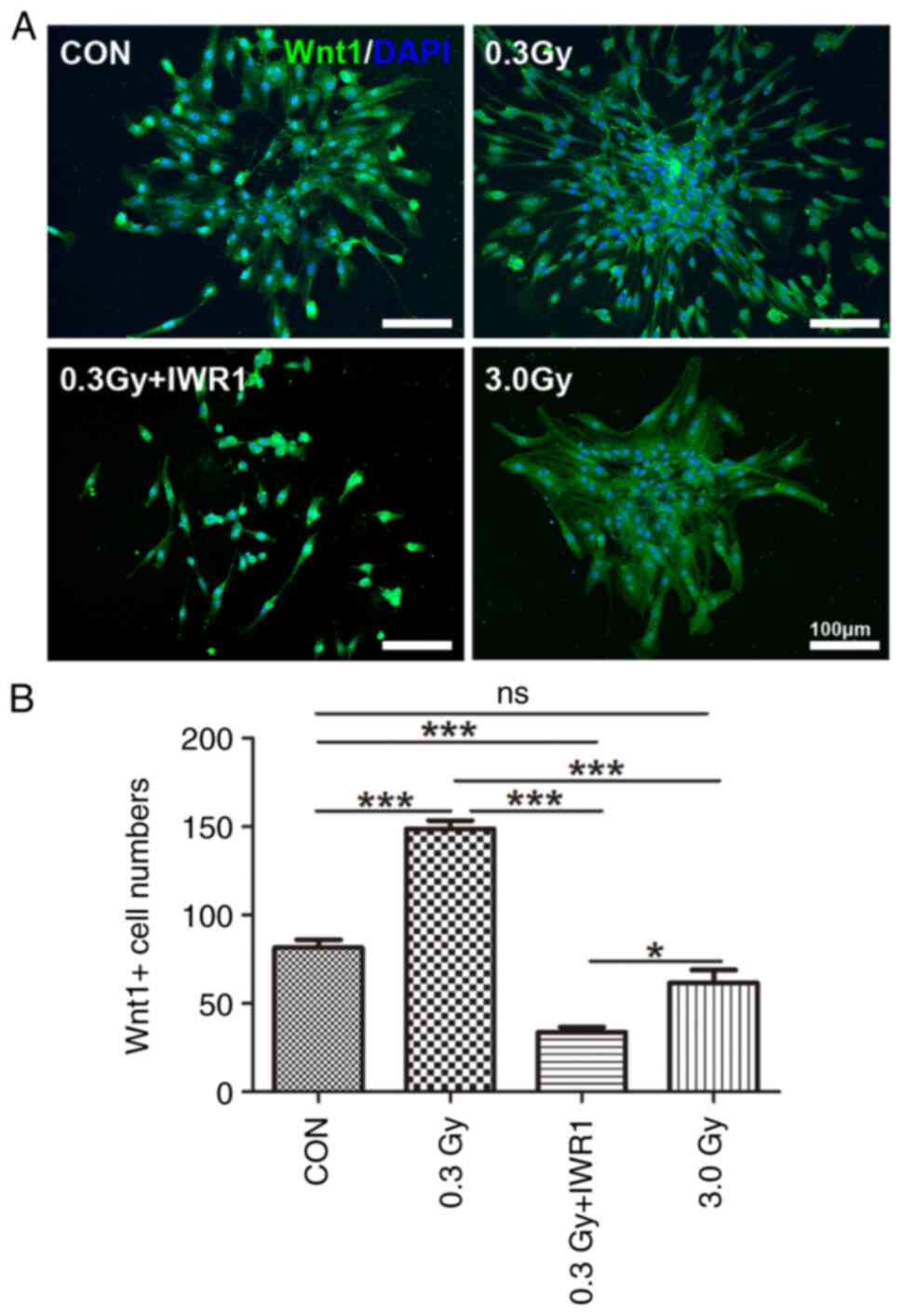
Wnt1-positive expression was detected in migratory C6 GSCs after LDR. The results revealed that Wnt1-positive cells were mainly found in the cytoplasm of C6 GSCs (Fig. 5A). Cell count data showed that the number of Wnt1-positive cells per clonal sphere significantly increased in the 0.3 Gy group (P<0.001 vs. the control), whereas the number of Wnt1-positive cells per clonal sphere significantly decreased in the 0.3 Gy + IWR1 group (P<0.001 vs. the control). The numbers of Wnt1-positive cells in the 0.3 Gy + IWR1 group demonstrated a notable difference in comparison with that of the 0.3 Gy group (P<0.001) (Fig. 5B).
Expression of Wnt3a in migratory C6 GSCs pretreated with IWR1 after LDR
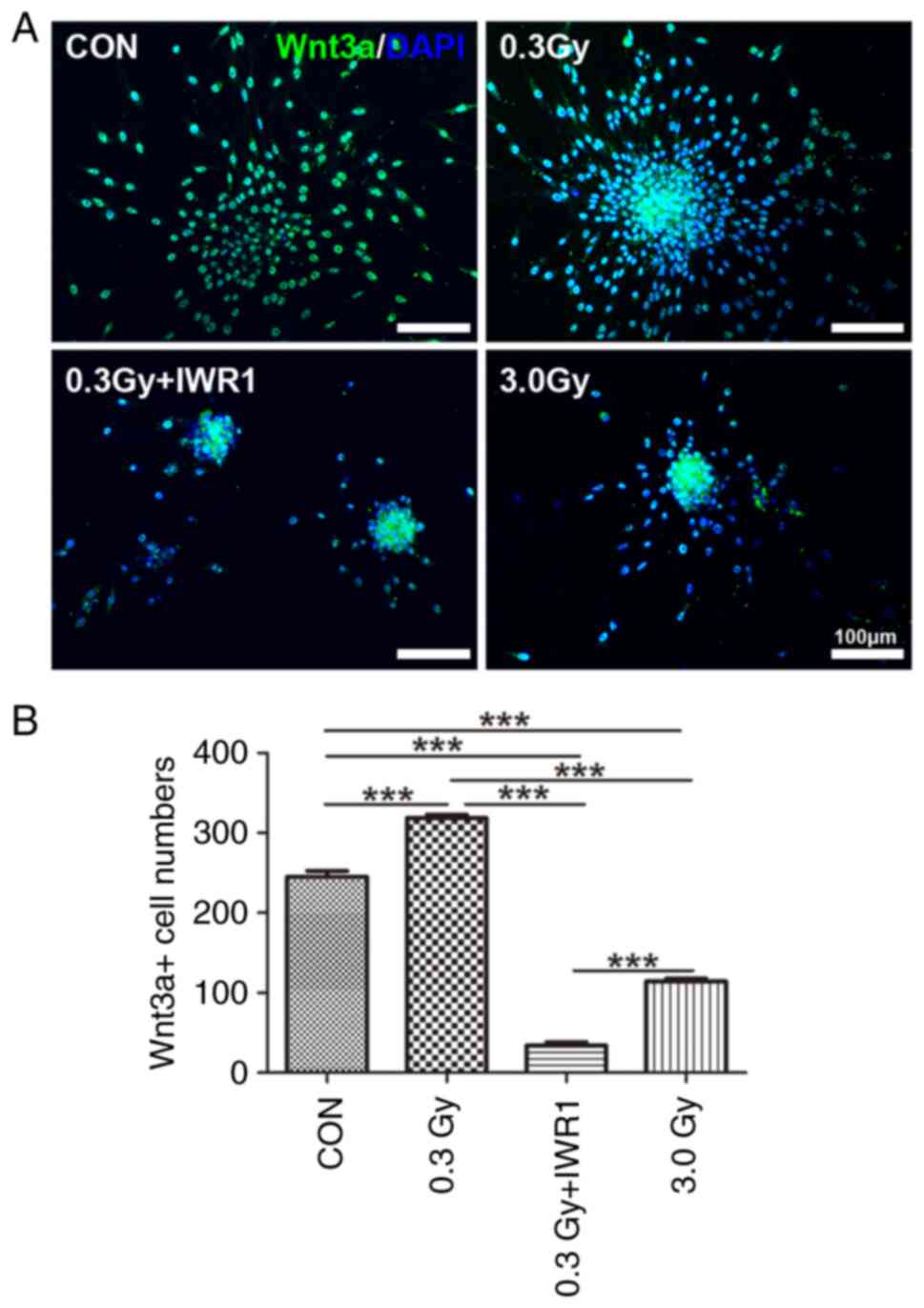
Wnt3a-positive expression was detected in migratory C6 GSCs after LDR. The results revealed that Wnt3a-positive cells were mainly found in the cell nucleus of C6 GSCs (Fig. 6A). Cell count data showed that the number of Wnt3a-positive cells per clonal sphere significantly increased in the 0.3 Gy group (P<0.001 vs. the control), whereas the number of Wnt3a-positive cells per clonal sphere significantly decreased in the 0.3 Gy + IWR1 group (P<0.001 vs. the control). The numbers of Wnt3a-positive cells in the 0.3 Gy + IWR1 group had a significant difference in comparison with that of the 0.3 Gy group (P<0.001). In addition, there were significant differences in the 3.0 Gy group compared with the control group (P<0.001) (Fig. 6B).
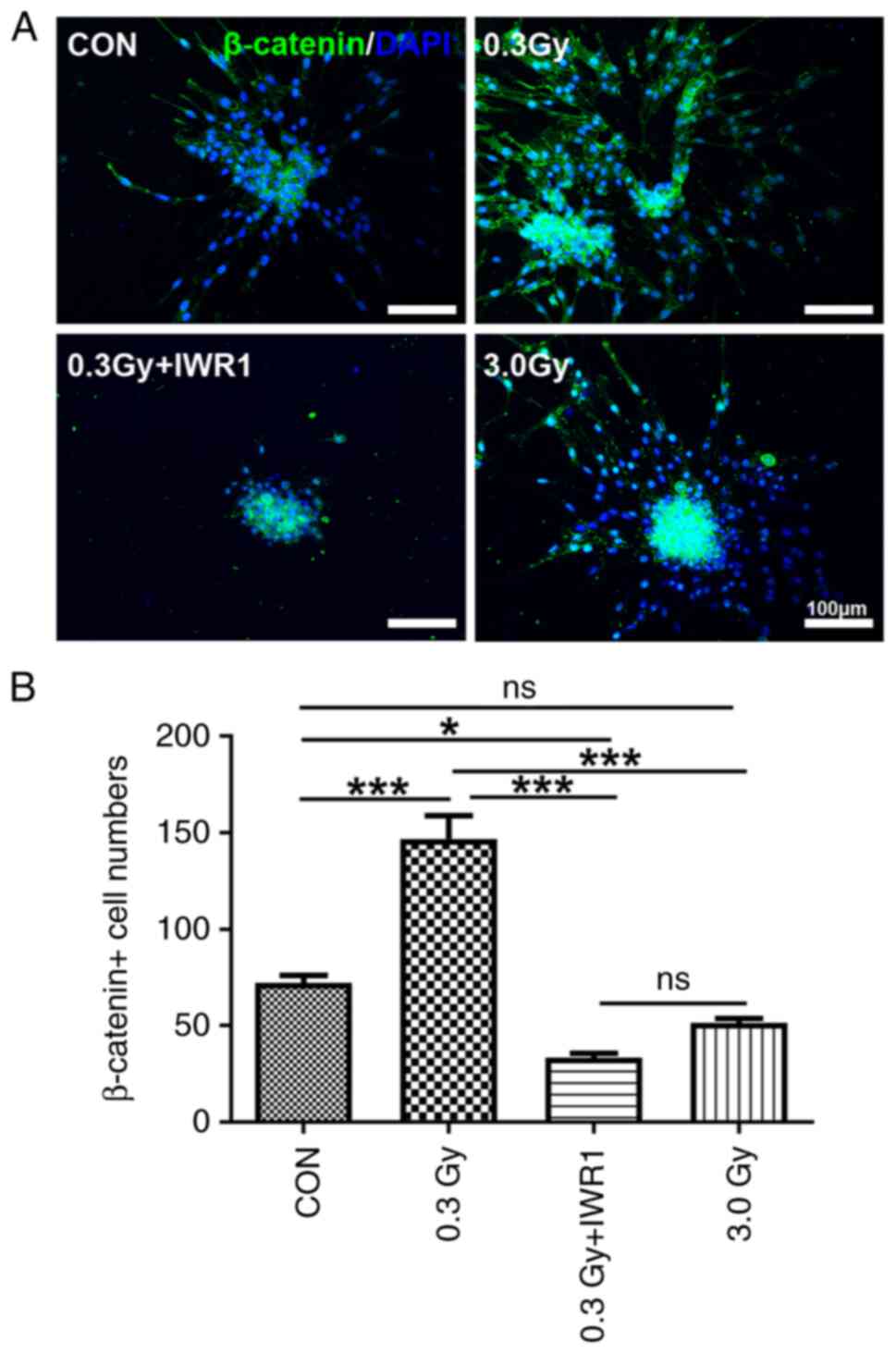
Expression of β-catenin in migratory C6 GSCs pretreated with IWR1 after LDR
The expression of β-catenin in migratory C6 GSCs was detected after LDR. The results demonstrated that β-catenin-positive cells were mainly found in the cytomembrane of C6 GSCs, which appeared as a string of beads (Fig. 7A). Cell count data revealed that the number of β-catenin-positive cells per clonal sphere significantly increased in the 0.3 Gy group (P<0.001 vs. the control), whereas the number of β-catenin-positive cells per clonal sphere significantly decreased in the 0.3 Gy + IWR1 group (P<0.05 vs. the control); the numbers of β-catenin-positive cells in the 0.3 Gy + IWR1 group had a significant difference in comparison with that of the 0.3 Gy group (P<0.001) (Fig. 7B).
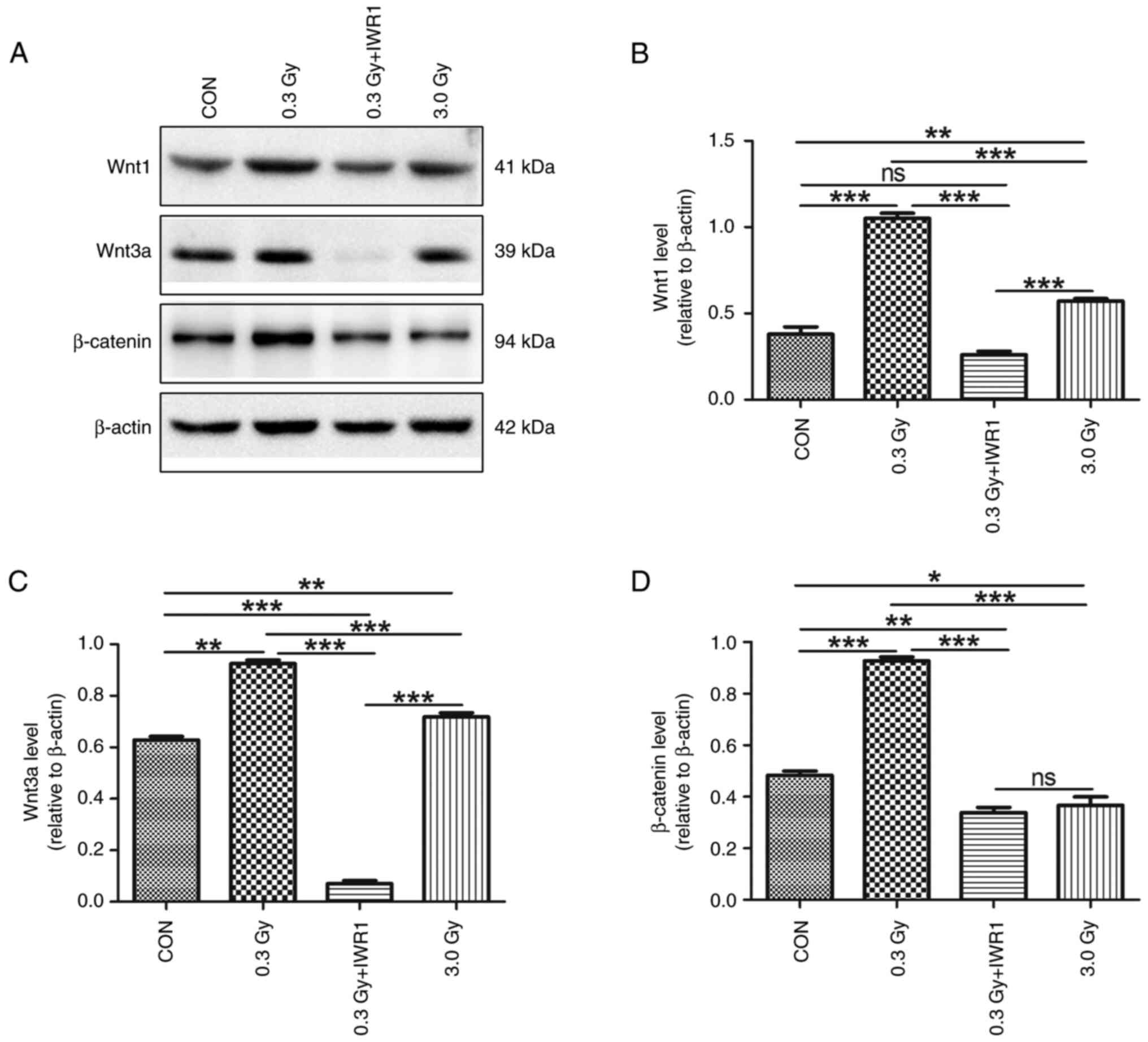
Migratory C6 GSCs demonstrate modulation of Wnt1, Wnt3a and β-catenin signaling activation after LDR
Western blotting and an inhibitor experiment were further performed to observe the involvement of Wnt/β-catenin signaling activation in C6 GSC migration ability or functional changes in C6 GSCs in response to LDR stimulation. Immunoblots to Wnt1, Wnt3a and β-catenin were detected in the control, 0.3, 0.3 Gy + IWR1 and 3.0 Gy radiation groups. Quantitative analysis revealed that the expression levels of Wnt1, Wnt3a and β-catenin increased in the 0.3 Gy group, indicating upregulation and activation of the Wnt/β-catenin signaling pathway in migrating C6 GSCs. The increased expression of Wnt1, Wnt3a and β-catenin was significantly diminished or almost completely blocked by pretreatment with 10 µmol/l IWR1. Furthermore, the expression levels of Wnt1, Wnt3a and β-catenin decreased in the 3.0 Gy group (Fig. 8A-D).
Discussion
Radiotherapy is frequently utilized in combination with surgical resection and chemotherapy for the treatment of brain tumors, but each of these therapy modalities has serious limitations. For instance, sensitivity to single radiation therapy can be strongly inhibited in the treatment of some gliomas. The efficacy of chemotherapeutic drugs can be greatly reduced due to poor penetrance of the blood-brain barrier, and the utility of surgery can be limited due to the position of tumors within the brain (25,26). At present, targeted regulation of stem cells can be adequately used for glioma therapy (10). C6 GSCs from the C6 glioma cell line form clonal spheres, which show self-renewing and proliferating multipotency, similar to cancer stem-like cells (27,28). in the present study, it was identified that C6 GSCs displayed proliferation and robust migration, and the rate of cell death and apoptosis significantly decreased after low-dose ionizing radiation (0.3 Gy). This effect was inhibited with higher-dose radiation (3.0 Gy). The level of γ-H2AX (a DNA damage marker) induced by ionizing radiation significantly improved with increasing radiation dose. Furthermore, the inhibitor of IWR1 can effectively suppress the function of low-dose ionizing radiation in the treatment of gliomas but has no relevance to the level of DNA damage.
Previously, for the proposed tumor stem cell hypothesis, CD133-positive tumor stem cells can be successfully isolated and cultured from glioma patients and their adjacent tissues, which provides favorable evidence for the existence of GSCs (29,30). Some studies have shown that cultured GSCs in vitro can proliferate and divide into cell spheres in serum-free medium, expressing Nestin and CD133 markers of neural stem cells. Analysis of the cell composition of the cell spheres found that GSCs also have the asymmetric division property of stem cells, and during the process of self-renewal, both BGSCs and non-BGSCs are generated. A small number of CD133-positive GSCs play an important role in the development and outcome of tumors (27,28). The results of the present study identified that GSCs isolated and cultured from the C6 glioma cell line grew as suspended cell spheres under the effect of stem cell conditioned medium and had strong proliferation and migration ability. It is suggested that GSCs with neural stem cell-like characteristics exist in the C6 glioma cell line, which supports the theory of neural stem cell origin to a large extent. After passage, C6 GSCs had favorable activity and could divide again and gather into cell spheres. BrdU incorporation and proliferation identification of P3 generation C6 GSCs demonstrated strong CD133 and BrdU positivity. Moreover, C6 GSCs proliferate rapidly and have numerous stem cells. This is consistent with the previously reported results (27,28). Therefore, in the present experiment, it was observed that passaged clonal spheres and first-passage C6 GSCs were not significantly different in morphology. Proliferation was identified by immunofluorescent staining and BrdU incorporation experiments, and CD133 and BrdU were strongly positive. All proved that the passaged clonal spheres had the same self-renewing and proliferative abilities as the first-passage C6 GSCs. The difference is that with increasing passage time, the number of tumor stem cells with self-renewal, proliferation and division abilities increases, as shown in Fig. 1A.
Radiation neurobiological studies have shown that mature neurons in the brain have a high tolerance to ionizing radiation. Higher-dose ionizing radiation (up to 10.0 Gy) will not cause obvious damage to neuronal structure, but neural stem cells are very sensitive to ionizing radiation, and a large dose of ionizing radiation can eliminate more than 90% of neural stem cells or cause cell apoptosis (31–33). However, low-dose ionizing radiation promotes the proliferation of mesenchymal stem cells or neural stem cells (34,24), and the sensitivity of glioma-initiating cells to low-dose ionizing radiation increases, which promotes cell survival (35). By analyzing the dose-response relationship of low-dose ionizing radiation, it was found that stem cells were more sensitive to LDR stimulation, and the maximum protective response occurred at a dose of 0.3 Gy, while the protective response could not be induced at doses greater than 0.5 Gy (24,33,36). Studies have confirmed that low-dose ionizing radiation stimulates stem cells at 0.01–0.1 Gy low-dose irradiation, and a single dose of 0.01 Gy for four consecutive times or a dose of 0.1 Gy irradiation can stimulate the proliferation of stem cells (37). In the present study, regarding the sensitivity of C6 GSCs to ionizing radiation, the findings showed that the proliferation and migration of C6 GSCs in the 0.3 Gy group could be promoted. The proliferation of C6 GSCs was inhibited when the radiation dose reached 3.0 Gy, and cell migration was evidently decreased. Thus, a dose of 0.3 Gy could induce a protective response, but a dose of 3.0 Gy could not induce a protective response. These results suggested that low-dose ionizing radiation (0.3 Gy) may promote the proliferation and migration of GSCs, which is consistent with the aforementioned results. Of course, at present, radiotherapy has become an important medical treatment method, especially in the treatment of malignant tumors, and high-dose ionizing radiation is often used in tumor radiotherapy. However, the effects of ionizing radiation on the organism, tissues and cells are complex and variable and have harmful and adaptive effects or excitatory effects. Therefore, these issues need to be further studied.
Previous research confirmed that ionizing radiation activated the response of oxidative stress, which directly caused DNA damage (38,39). In fact, γ-H2AX, which is produced by phosphorylation of histone H2AX at S139, is a very early step in the DNA damage response and an essential signaling molecule for the recruitment and retention of DNA damage response complexes at the site of damage (40). The present findings indicated that ionizing radiation initially increased DNA damage, as indicated by a similar level of γ-H2AX (the DNA damage marker) fluorescence intensity in C6 GSCs. However, the level of γ-H2AX fluorescence intensity was positively related to the increase in radiation dose. In the current experiment, the level of γ-H2AX significantly improved with increasing radiation dose (3.0 Gy). Furthermore, the level of γ-H2AX in the 0.3 Gy group significantly differed from that of γ-H2AX in the 0.3 Gy with IWR1 group (P<0.05). It was analyzed how IWR1 can effectively inhibit the cell numbers of C6 GSCs after low-dose ionizing radiation, which has no relevance to radiation dose. The upregulation of DNA repair enzymes in conjunction with γ-H2AX results suggested a vital role for DNA repair in the survival and proliferation of C6 GSCs, and it is necessary to further investigate the regulation of specific mechanisms.
The Wnt/β-catenin signaling pathway plays a major role in cell survival, apoptosis and proliferation, as well as in migration and invasion in various cancer types (41–43). Studies have shown that Wnt3a molecules promote the occurrence and development of glioma by activating Wnt/β-catenin signaling (44). Wnt5a could be involved in the process of brain invasion and enhance the survival rate of the host in a mouse transplantation model of glioblastoma (45). The overexpression of Wnt2, β-catenin and Wnt5a is observed in gliomas, while cell proliferation and invasion are obviously inhibited, and apoptotic cell death is induced in glioma cells (46). The expression of β-catenin in glioma tissues was significantly higher than that in normal tissues (47). Other studies have also confirmed that Wnt/β-catenin interacts with multiple signaling pathways to inhibit the proliferation, apoptosis or survival of glioma cells (22,48). The findings of the present study identified that the expression of Wnt1, Wnt3a and β-catenin molecules on C6 GSCs was significantly upregulated after low-dose ionizing radiation (0.3 Gy). The expression levels of Wnt1, Wnt3a and β-catenin were inhibited when the radiation dose reached 3.0 Gy. However, the levels of Wnt1, Wnt3a and β-catenin in the 0.3 Gy + IWR1 group were partly and completely inhibited (P<0.001 vs. the 0.3 Gy group). This finding indicated that Wnt/β-catenin signaling modulates the proliferation and migration of C6 GSCs induced by LDR.
At the beginning of the present experiment, the effect of different factors was tested. Concentration of IWR1 (0, 0.1, 1, 5 and 10 µM) on suspending-cultured GSC spheres, the same treatment methods were previously reported (24); all of cell spheres were first, mixed before treatment, and then treated in separate bottles to ensure the consistency of the control group. When the suspended GSC spheres were cultured for 4–5 days, different concentrations of IWR1 were added to the medium and cultured for 24–48 h. During this period, it was observed under the microscope that there was no significant difference in cell size between 0.1 and 1.0 µM IWR1 group and Con group. The cells exhibited strong refraction, clear liquid and several scattered single cells. The number and proliferation rate of cell spheres in 5.0 µM IWR1 group slowed, and there were multiple scattered single cells in the fluid. In the 10.0 µM IWR1 group, the number and proliferation rate of cell spheres were significantly reduced, the ability of cell agglomeration was sharply decreased, and there were more scattered, sheets or groups of single cells in the liquid. Flow cytometric analysis showed that the proportion of living cells in the 10.0 µM IWR1 group decreased to <50%, and the proportion of apoptosis and cell necrosis increased greatly. Moreover, after 48 h of continuous culture in the 10.0 µM IWR1 group, the number of cell spheres observed under the microscope was small, the number of cells in each cell sphere was also small, and the ability of cells gathering the spheres was almost lost. There were numerous single cells in the liquid, and numerous cells disassembled and fragmented. After cell centrifugation and resuspension, there were few cells in the field of vision under the microscope. Cells were collected, but the number of cells was too small to meet the minimum number of cells needed for flow cytometry and could not be detected. Therefore, in the follow-up experiment, the results of the 10.0 µM IWR1 group were not shown without LDR as a control. However, according to the present experimental data and reference to other studies (49,50), it is feasible that the current experimental results and purposes are not affected.
In addition, according to the dose-effect relationship of LDR stimulation and the inhibitor experiment, the present study indicated that the Wnt/β-catenin signaling pathway plays a key role in regulating the occurrence and development of glioma (51,52). Therefore, the current study observed the effects of different doses of radiation stimulation on the proliferation and migration of C6 GSCs, as well as the changes in Wnt1, Wnt3a and β-catenin levels before and after LDR. Before and after radiation, the treatment of C6 GSCs was pretreated with 10.0 µM IWR1 (the proportion of living cells was reduced to <50%), and its effect on the protective response induced by 0.3 Gy radiation dose was observed, which will provide an important experimental basis for revealing the regulatory mechanism of LDR technology for the prevention and treatment of brain glioma.
The results of cell survival and apoptosis using flow cytometric analysis showed that LDR stimulation remarkably attenuated cell necrosis and apoptosis and increased the proliferation and survival of C6 GSCs in the 0.3 Gy group, but these effects were reversed due to the inhibition of Wnt/β-catenin signaling molecules. Of course, it is known that cell necrosis is the passive death caused by pathology, which is pathological cell death. Apoptosis is the programmed death of cells, which is widespread in the development of organisms. Of course, the scope of the two is different; apoptosis is mostly observed in a single scattered cell, while cell necrosis is observed in large tissues or groups of cells. However, the cell membrane remains intact until apoptotic bodies are formed. Cell necrosis breaks the cell membrane, and the cell swells and disintegrates, forming cell fragments. In the present experiment, although no specific marker for cell necrosis and apoptosis was used to identify cells before and after an IWR1 pretreatment experiment, the growth of cell spheres before and after treatment could also be preliminarily evaluated. For example, GSC cell spheres in the 0.3 and 3.0 Gy groups and 0.3 and 0.3 Gy + IWR1 groups were compared and observed under an inverted phase contrast microscope (data not shown). The results revealed that the cell spheres in the 0.3 Gy group had a strong aggregation ability, rapid proliferation rate, increased cell number, large and round cell bodies and strong refraction. In the center of the larger cell ball, a hypotrophic zone was formed. Under the microscope, the color was dark, the cells were necrotic and disintegrated, and some cell fragments were found in the medium. In the 3.0 Gy group, the cell ball aggregation ability was weak, the proliferation rate decreased, the number of cells decreased, the cell membrane structure was complete, the cell body was large and round, and there was strong refraction. In addition, the cells in the 0.3 Gy + IWR1 group were observed, and the number of cells was sharply reduced, with numerous cell disintegrating fragments in the liquid, and cells with complete cell membrane structure and dark refraction were also visible. Further analysis by flow cytometry demonstrated that the proportion of apoptotic cells increased in the 3.0 Gy group, and the proportions of necrotic and apoptotic cells increased in the 0.3 Gy + IWR1 group. The results are consistent with those observed under a phase contrast microscope.
Overall, in the present experiment, it was confirmed that C6 GSCs generated from the C6 glioma cell line can form cell spheres, which have self-renewing and migratory capabilities. With LDR intervention, cell survival and cell apoptosis could be changed, and these effects were completely and partly blocked by pretreatment with IWR1. Therefore, the current results also revealed that Wnt/β-catenin signaling may modulate the proliferation and migration of C6 GSCs induced by LDR. Of course, ionizing radiation stimulation can promote the proliferation, survival, invasion and apoptosis of glioma cells within a certain dose range, and the mechanism may be related to the crosstalk of multiple signaling molecules via the activation of Wnt/β-catenin signaling (53–55). Therefore, according to individual differences, how to define the scope of radiation dose and develop a specific radiation scheme, how to achieve the best protective response using a large dose of shock therapy or multiple small doses of staging treatment, and how to further study the relationship between radiation-sensitive effects and activated Wnt signaling will become a research topic in the future.
In conclusion, these data indicated that Wnt1, Wnt3a and β-catenin are upregulated in GSCs and correlated with low-dose ionizing radiation. Moreover, Wnt3a could behave as an oncogene that was completely blocked by pretreatment with IWR1 after LDR, which suggested that activating Wnt/β-catenin signaling in glioma cells is a potential target for an effective radiosensitizer of GSCs to exploit and utilize.
Acknowledgements
The authors would like to thank Professor Liang-Wei Chen (Institute of Neurosciences of the Fourth Military Medical University) for his help with various aspects of the present study.
Funding
The present study was supported by the Ningxia University Scientific Research Project (grant no. NGY2018-68), the National Basic Research Program of Ningxia (grant nos. 2022AAC03153 and 2022AAC03184) and the National Basic Research Program of China (grant no. 31760279).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
YXD and HYC conceived the study design. YYC and YY performed the experiments. YXD and YYC analyzed the data. YRL, XWJ and YML provided technical advice and/or helped with data analysis. All authors read and approved the final version of the manuscript. YXD, YYC and YY confirm the authenticity of all the raw data.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, Wolinsky Y, Kruchko C and Barnholtz-Sloan J: CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol. 16 (Suppl 4):iv1–iv63. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW and Kleihues P: The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 114:97–109. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Stupp R, Mason WP, Van Den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, et al: Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Stupp R, Taillibert S, Kanner A, Read W, Steinberg D, Lhermitte B, Toms S, Idbaih A, Ahluwalia MS, Fink K, et al: Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: A randomized clinical trial. JAMA. 318:2306–2316. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Gallego O: Nonsurgical treatment of recurrent glioblastoma. Curr Oncol. 22:e273–e281. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Prados MD, Byron SA, Tran NL, Phillips JJ, Molinaro AM, Ligon KL, Wen PY, Kuhn JG, Mellinghoff IK, de Groot JF, et al: Toward precision medicine in glioblastoma: The promise and the challenges. Neuro Oncol. 17:1051–1063. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Rudà R, Pellerino A, Magisstrello M, Franchino F, Pinessi L and Soffietti R: Molecularly based management of gliomas in clinical practice. Neurol Sci. 36:1551–1557. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Hollon TC, Pandian B, Urias E, Save AV, Adapa AR, Srinivasan S, Jairath NK, Farooq Z, Marie T, Al-Holou WN, et al: Rapid, label-free detection of diffuse glioma recurrence using intraoperative stimulated Raman histology and deep neural networks. Neuro Oncol. 23:144–155. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Sundar SJ, Hsieh JK, Manjila S, Lathia JD and Sloan A: The role of cancer stem cells in glioblastoma. Neurosurg Focus. 37:E62014. View Article : Google Scholar : PubMed/NCBI | |
|
Ruiz-Garcia H, Alvarado-Estrada K, Krishnan S, Quinones-Hinojosa A and Trifiletti DM: Nanoparticles for stem cell therapy bioengineering in glioma. Front Bioeng Biotechnol. 8:5583752020. View Article : Google Scholar : PubMed/NCBI | |
|
Nowell PC: The clonal evolution of tumor cell populations. Science. 194:23–28. 1976. View Article : Google Scholar : PubMed/NCBI | |
|
Bao S, Wu Q, Li Z, Sathornsumetee S, Wang H, McLendon RE, Hjelmeland AB and Rich JN: Targeting cancer stem cells through L1CAM suppresses glioma growth. Cancer Res. 68:6043–6048. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A, De Vitis S, Fiocco R, Foroni C, Dimeco F and Vescovi A: Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res. 64:7011–7021. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Chen R, Nishimura MC, Bumbaca SM, Kharbanda S, Forrest WF, Kasman IM, Greve JM, Soriano RH, Gilmour LL, Rivers CS, et al: A hierarchy of self-renewing tumor-initiating cell types in glioblastoma. Cancer Cell. 17:362–375. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Chen J, Li Y, Yu TS, McKay RM, Burns DK, Kernie SG and Parada LF: A restricted cell population propagates glioblastoma growth after chemotherapy. Nature. 488:522–526. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Eyler CE, Foo WC, LaFiura KM, McLendon RE, Hjelmeland AB and Rich JN: Brain cancer stem cells display preferential sensitivity to Akt inhibition. Stem Cells. 26:3027–3036. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Hoang-Minh LB, Siebzehnrubl FA, Yang C, Suzuki-Hatano S, Dajac K, Loche T, Andrews N, Schmoll Massari M, Patel J, Amin K, et al: Infiltrative and drug-resistant slow-cycling cells support metabolic heterogeneity in glioblastoma. EMBO J. 37:e987722018. View Article : Google Scholar : PubMed/NCBI | |
|
Li DX, Fei XR, Dong YF, Cheng CD, Yang Y, Deng XF, Huang HL, Niu WX, Zhou CX, Xia CY and Niu CS: The long non-coding RNA CRNDE acts as a ceRNA and promotes glioma malignancy by preventing miR-136-5p-mediated downregulation of Bcl-2 and Wnt2. Oncotarget. 8:88163–88178. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Shi Y, Wang Y, Luan W, Wang P, Tao T, Zhang J, Qian J, Liu N and You Y: Long non-coding RNA H19 promotes glioma cell invasion by deriving miR-675. PLo S One. 9:e862952014. View Article : Google Scholar : PubMed/NCBI | |
|
Lee Y, Lee JK, Ahn SH, Lee J and Nam DH: WNT signaling in glioblastoma and therapeutic opportunities. Lab Invest. 96:137–150. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Wang G, Shen J, Sun J, Jiang Z, Fan J, Wang H, Yu S, Long Y, Liu Y, Bao H, et al: Cyclophilin A maintains glioma-initiating cell stemness by regulating Wnt/β-catenin signaling. Clin Cancer Res. 23:6640–6649. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Gao L, Chen B, Li J, Yang F, Cen X, Liao Z and Long X: Wnt/β-catenin signaling pathway inhibits the proliferation and apoptosis of U87 glioma cells via different mechanisms. PLoS One. 12:e01813462017. View Article : Google Scholar : PubMed/NCBI | |
|
Kee N, Sivalingam S, Boonstra R and Wojtowicz JM: The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods. 115:97–105. 2002. View Article : Google Scholar : PubMed/NCBI | |
|
Wei LC, Ding YX, Liu YH, Duan L, Bai Y, Shi M and Chen LW: Low-dose radiation stimulates Wnt/β-catenin signaling, neural stem cell proliferation and neurogenesis of the mouse hippocampus in vitro and in vivo. Curr Alzheimer Res. 9:278–289. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Bylicky MA, Mueller GP and Day RM: Radiation resistance of normal human astrocytes: The role of non-homologous end joining DNA repair activity. J Radiat Res. 60:37–50. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Eom HS, Park HR, Jo SK, Kim YS, Moon C, Kim SH and Jung U: Ionizing radiation induces altered neuronal differentiation by mGluR1 through PI3K-STAT3 signaling in C17.2 mouse neural stem-like cells. PLoS One. 11:e01475382016. View Article : Google Scholar : PubMed/NCBI | |
|
Kondo T, Setoguchi T and Taga T: Persistence of a small subpopulation of cancer stem-like cells in the C6 glioma cell line. Proc Natl Acad Sci USA. 101:781–786. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD and Dirks PB: Identification of human brain tumour initiating cells. Nature. 432:396–401. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Stiles CD and Rowitch DH: Glioma stem cells: A midterm exam. Neuron. 58:832–846. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Fan X, Salford LG and Widegren B: Gliomal stem cells: Evidence and Limitation. Semin Cancer Bio. 17:214–218. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Mizumatsu S, Monje ML, Morhardt DR, Rola R, Palmer TD and Fike JR: Extreme sensitivity of adult neurogenesis to low doses of X-irradiation. Cancer Res. 63:4021–4027. 2003.PubMed/NCBI | |
|
Zhang W, Wang PJ, Li P, Li MH and Gao XL: Effects of low-dose X-ray on morphology of neuron and expression of microtubule associated protein-2 in hippocampus of young rats. Zhonghua Yi Xue Za Zhi. 92:336–340. 2012.(In Chinese). PubMed/NCBI | |
|
Ding M, Zhang E, He R, Wang X, Li R, Wang W and Yi Q: The radiation dose-regulated AND gate genetic circuit, a novel targeted and real-time monitoring strategy for cancer gene therapy. Cancer Gene Ther. 19:382–392. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Liang X, So YH, Cui J, Ma K, Xu X, Zhao Y, Cai L and Li W: The low-dose ionizing radiation stimulates cell proliferation via activation of the MAPK/ERK pathway in rat cultured mesenchymal stem cells. J Radiat Res. 52:380–386. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Lim YC, Roberts TL, Day BW, Stringer BW, Kozlov S, Fazry S, Bruce ZC, Ensbey KS, Walker DG, Boyd AW and Lavin MF: Increased sensitivity to ionizing radiation by targeting the homologous recombination pathway in glioma initiating cells. Mol Oncol. 8:1603–1615. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Kang JO, Kim SK, Hong SE, Lee TH and Kim CJ: Low dose radiation overcomes diabetes-induced suppression of hippocampal neuronal cell proliferation in rats. J Korean Med Sci. 21:500–505. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Tapio S: Pathology and biology of radiation-induced cardiac disease. J Radiat Res. 57:439–448. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Basso E, Regazzo G, Fiore M, Palma V, Traversi G, Testa A, Degrassi F and Cozzi R: Resveratrol affects DNA damage induced by ionizing radiation in human lymphocytes in vitro. Mutat Res Genet Toxicol Environ Mutagen. 806:40–46. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Djuzenova CS, Elsner I, Katzer A, Worschech E, Distel LV, Flentje M and Polat B: Radiosensitivity in breast cancer assessed by the histone γ-H2AX and 53BP1 foci. Radiat Oncol. 8:982013. View Article : Google Scholar : PubMed/NCBI | |
|
Ji J, Zhang Y, Redon CE, Reinhold WC, Chen AP, Fogli LK, Holbeck SL, Parchment RE, Hollingshead M, Tomaszewski JE, et al: Phosphorylated fraction of H2AX as a measurement for DNA damage in cancer cells and potential applications of a novel assay. PLoS One. 12:e01715822017. View Article : Google Scholar : PubMed/NCBI | |
|
Kahlert UD, Suwala AK, Koch K, Natsumeda M, Orr BA, Hayashi M, Maciaczyk J and Eberhart CG: Pharmacologic Wnt inhibition reduces proliferation, survival, and clonogenicity of glioblastoma cells. J Neuropathol Exp Neurol. 74:889–900. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Qi Y, Guo L, Liu Y, Zhao T, Liu X and Zhang Y: Sevoflurane limits glioma progression by regulating cell proliferation, apoptosis, migration, and invasion via miR-218-5p/DEK/β-catenin axis in glioma. Cancer Manag Res. 13:2057–2069. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Z, Chen G and Zhao H: FDPS promotes glioma growth and macrophage recruitment by regulating CCL20 via Wnt/β-catenin signalling pathway. J Cell Mol Med. 24:9055–9066. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Kaur N, Chettiar S, Rathod S, Rath P, Muzumdar D, Shaikh ML and Shiras A: Wnt3a mediated activation of Wnt/β-catenin signaling promotes tumor progression in glioblastoma. Mol Cell Neurosci. 54:44–57. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Li H, Tong F, Meng R, Peng L, Wang J, Zhang R and Dong X: E2F1-mediated repression of WNT5A expression promotes brain metastasis dependent on the ERK1/2 pathway in EGFR-mutant non-small cell lung cancer. Cell Mol Life Sci. 78:2877–2891. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Pu P, Zhang Z, Kang C, Jiang R, Jia Z, Wang G and Jiang H: Downregulation of Wnt2 and beta-catenin by siRNA suppresses malignant glioma cell growth. Cancer Gene Ther. 16:351–361. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Du L, Lee JH, Jiang H, Wang C, Wang S, Zheng Z, Shao F, Xu D, Xia Y, Li J, et al: β-Catenin induces transcriptional expression of PD-L1 to promote glioblastoma immune evasion. J Exp Med. 217:e201911152020. View Article : Google Scholar : PubMed/NCBI | |
|
hang J, Cai H, Sun L, Zhan P, Chen M, Zhang F, Ran Y and Wan J: LGR5, a novel functional glioma stem cell marker, promotes EMT by activating the Wnt/β-catenin pathway and predicts poor survival of glioma patients. J Exp Clin Cancer Res. 37:2252018. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang X, Chen L, Wang Y, Ding Y, Peng Z, Duan L, Ju G, Ren Y and Wang X: Macrophage migration inhibitory factor promotes proliferation and neuronal differentiation of neural stem/precursor cells through Wnt/β-catenin signal pathway. Int J Biol Sci. 9:1108–1120. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Chen B, Dodge ME, Tang W, Lu J, Ma Z, Fan CW, Wei S, Hao W, Kilgore J, Williams NS, et al: Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat Chem Biol. 5:100–107. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Kierulf-Vieira KS, Sandberg CJ, Grieg Z, Günther CC, Langmoen IA and Vik-Mo EO: Wnt inhibition is dysregulated in gliomas and its re-establishment inhibits proliferation and tumor sphere formation. Exp Cell Res. 340:53–61. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Griveau A, Seano G, Shelton SJ, Kupp R, Jahangiri A, Obernier K, Krishnan S, Lindberg OR, Yuen TJ, Tien AC, et al: A glial signature and Wnt7 signaling regulate glioma-vascular interactions and tumor microenvironment. Cancer Cell. 33:874–889.e7. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Feng F, Zhao Z, Zhou Y, Cheng Y, Wu X and Heng X: CUX1 facilitates the development of oncogenic properties via activating Wnt/β-catenin signaling pathway in glioma. Front Mol Biosci. 8:7050082021. View Article : Google Scholar : PubMed/NCBI | |
|
Dong Z, Zhou L, Han N, Zhang M and Lyu X: Wnt/β-catenin pathway involvement in ionizing radiation-induced invasion of U87 glioblastoma cells. Strahlenther Onkol. 191:672–680. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Osuka S, Zhu D, Zhang Z, Li C, Stackhouse CT, Sampetrean O, Olson JJ, Gillespie GY, Saya H, Willey CD and Van Meir EG: N-cadherin upregulation mediates adaptive radioresistance in glioblastoma. J Clin Invest. 131:e1360982021. View Article : Google Scholar : PubMed/NCBI |