Cellular senescence in metastatic prostate cancer: A therapeutic opportunity or challenge (Review)
- Authors:
- Published online on: July 8, 2024 https://doi.org/10.3892/mmr.2024.13286
- Article Number: 162
-
Copyright: © Jin et al. This is an open access article distributed under the terms of Creative Commons Attribution License.
Abstract
Introduction
Prostate cancer (PCa) is a type of malignant tumor that originates in the epithelium of the prostate gland. According to the 2023 American Cancer Society report, PCa is ranked as the second most common type of cancer in men worldwide and is a leading cause of cancer-related death in the male population (1,2). Although radical prostatectomy is an effective strategy in treating early-stage PCa, a number of patients with PCa present with distant metastases at diagnosis (3). Surgical removal of the tumor, or chemical and surgical castration, are the standard treatments for patients with advanced PCa. However, a large proportion of patients with PCa that are initially responsive to endocrine therapy progress to chemoresistant PCa (CRPC) in 18–24 months (4). Patients with CRPC typically develop metastases; bone and lymph node metastases are the most common types of metastases in these patients (5). Furthermore, a number of patients with metastatic PCa present with resistance to established treatment modalities, notably androgen deprivation therapy (ADT), which results in suboptimal therapeutic outcomes (6). Therefore, understanding the molecular mechanisms involved in the transition from localized to metastatic PCa is crucial for the development of more effective treatments for patients with metastatic PCa.
A large proportion of the available PCa treatments are associated with the induction of cellular senescence. Senescence, while effectively arresting the cell cycle, has a dual role in tumor treatment. Although senescence imparts a steady state of cell cycle arrest, it also increases the potential of tumor cell invasion and lymphangiogenesis, which promotes PCa metastasis (7). This phenomenon is suggested to be associated with the secretome produced by senescent cells, which includes cytokines, chemokines, inflammatory factors and proteases, collectively referred to as the senescence-associated secretory phenotype (SASP) (8). The SASP can promote tumor cell epithelial-mesenchymal transition (EMT) and angiogenesis, which are both pivotal in tumor metastasis (9). It is considered a challenge to effectively harness the antitumor effects of the SASP, while avoiding its potential pro-tumor effects in current drug development. However, few studies have summarized the role of senescence and its associated phenotype in promoting PCa metastasis and its relevance to therapeutic mechanisms (10–12). In the present review, the complex relationship between PCa metastasis, the SASP and the potential of senescence-targeting therapies as an opportunity for treatment of metastatic PCa is discussed, with the aim of advancing research on senotherapeutic drugs.
Cellular senescence
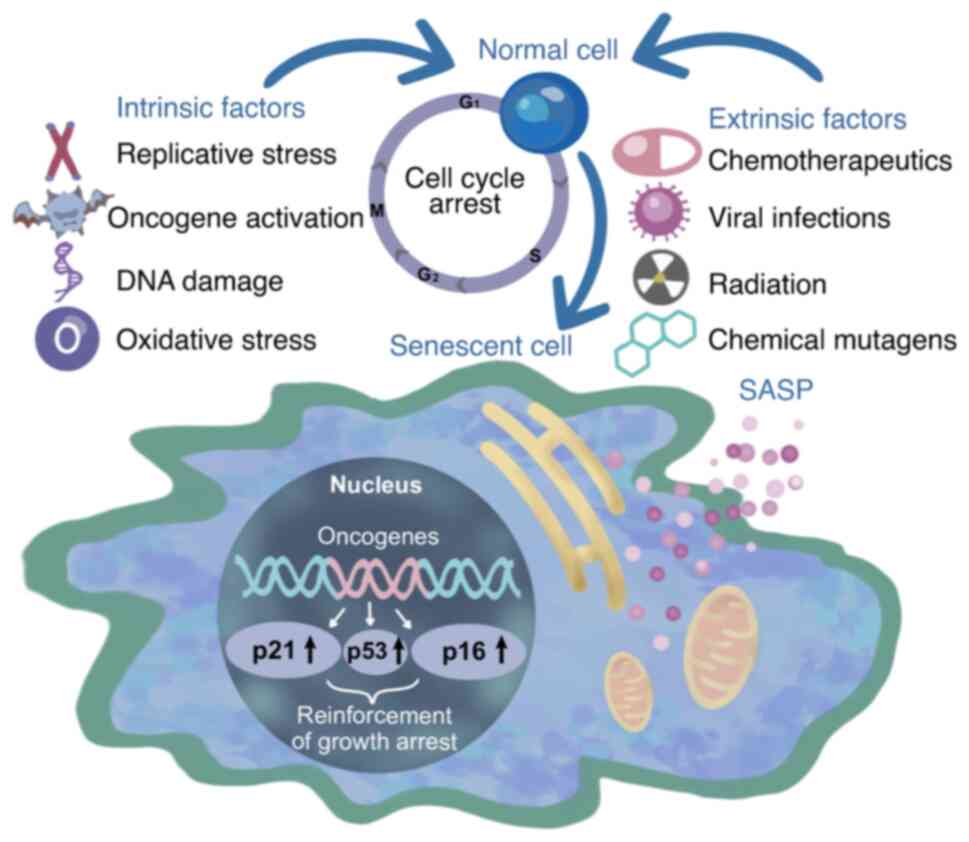
Under in vitro culture conditions, healthy human cells have a short lifespan and inevitably undergo restricted proliferation, even under optimal culture conditions (13). This restriction in proliferation, known as the Hayflick limit, describes the phenomenon whereby normal human cells possess limited capacity for in vitro division, commonly referred to as cellular senescence (14). Cellular senescence can be induced by various factors, which includes intrinsic factors such as activation of oncogenes, oxidative stress, genotoxic stress and mitochondrial dysfunction, as well as extrinsic factors such as radiation and chemotherapeutic drugs (15). These factors instigate cellular senescence by inducing DNA damage and activation of the p53/p21 or p16 signaling axis, which results in cell cycle arrest (16). Cellular senescence demonstrates a dual role in PCa treatment, with both beneficial and detrimental effects. In terms of antitumor effects, cellular senescence is regarded as an anticancer phenomenon as senescent tumor cells are generally considered to be rare in malignant tumor tissues (17). This is attributed to senescent cells being in a stable state of cell cycle arrest, which can restrict the unchecked proliferation of tumor cells, induce a senescent state in neighboring cells and recruit immune cells to inhibit tumor growth (18). By contrast, in terms of the pro-tumorigenic effects of senescence, the complexity of the SASP components produced by the senescent microenvironment contributes to adverse effects. A number of the cytokines produced by the SASP can stimulate tumor cell proliferation, invasion and metastasis, promote tumor angiogenesis and interfere with tumor immunity (Fig. 1) (19).
The SASP
Recent studies have suggested that the induction of cellular senescence as a treatment for PCa may yield certain benefits to patients, such as improved immune surveillance and inhibition of tumor growth (11,20). However, ~1 year following senescence-inducing treatment, patients with PCa were reported to develop resistance to the treatment and progress to CRPC (21). These contradictory outcomes can be attributed to the SASP.
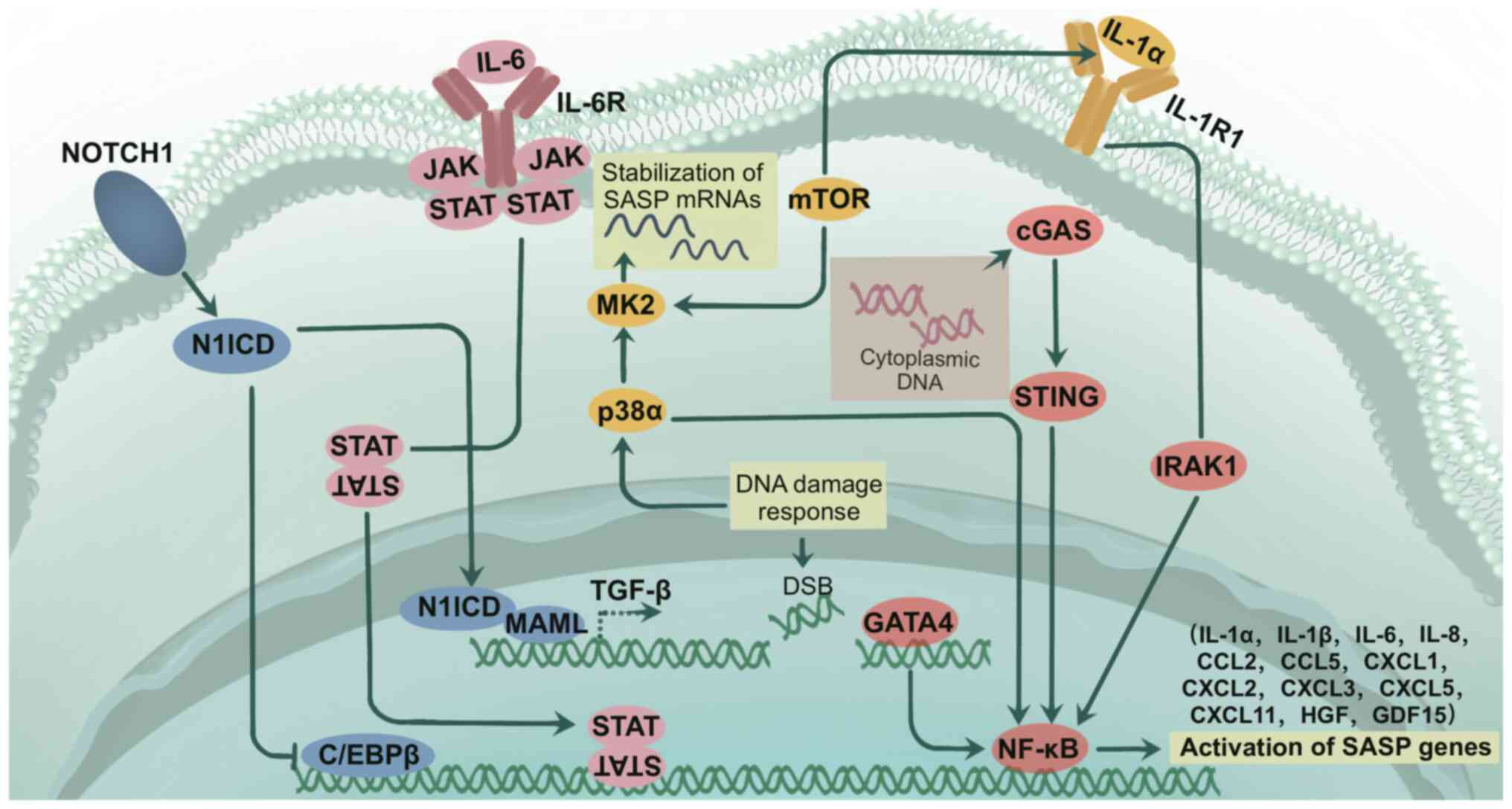
The concept of the SASP was first introduced by Coppé et al (22) in 2008, who reported that senescent cells can induce the oncogenic transformation of neighboring precancerous cells by secreting inflammatory and oncogenic gene-associated factors, which was termed as the SASP. The SASP generally refers to a complex group of secreted factors associated with senescent cells (23), including proteogenic inflammatory cytokines and chemokines, proteases, growth factors, biologically active lipids and extracellular vesicles (24,25). Under chronic stress conditions, such as DNA damage and oncogene expression, pathways, such as the p53 and p16 pathways, are activated, which leads to the activation of downstream effectors, such as p38, NF-κB and CCAAT/enhancer-binding protein β (C/EBPβ), and results in increased activation of the SASP and cellular senescence (26,27). The SASP can be induced by a number of treatments, such as chemotherapy and ionizing radiation, and develops from an acute stress-associated phenotype (ASAP), which typically manifests 5–8 days after the initiation of PCa treatment (11). The SASP is regulated by several signaling pathways. Cytoplasmic chromatin fragments, recognized by the cytoplasmic DNA sensor cyclic GMP-AMP synthase (cGAS), promote the SASP through the activation of the cGAS-interferon gene stimulating protein (STING) pathway (28). The cGAS-STING signaling pathway, which consists of cGAS, STING and downstream signaling adaptors, is able to sense and respond to the abnormal presence of double-stranded DNA in the cytoplasm of cells (29). IL-1α/IL-1 receptor signaling, positioned upstream of NF-κB, activates mTOR signaling, which reactivates NF-κB. Neutralizing antibodies against IL-1α or its receptor reduce the transcriptional activity of NF-κB, thereby inhibiting the production of inflammatory factors (30). In addition, the production of SASP factors can be regulated by the Janus kinase (JAK)/STAT pathway (21). A previous study demonstrated that PTEN deficiency leads to the production of inflammatory factors and activation of the JAK/STAT pathway, whereas inhibition of the JAK/STAT pathway attenuates the growth of PTEN-deficient prostate tumors and reduces resistance to chemotherapy (31). Lastly, NOTCH1 specifically inhibits C/EBPβ activity in fibroblasts in vitro and in vivo, which prevents the secretion of proinflammatory SASP factors (Fig. 2) (32).
The SASP serves a dual role in tumorigenesis and progression. Positive regulatory effects involve some SASP factors, such as IL-6 and IL-8, which increase the immunosurveillance of senescent cells. This stimulation prompts the immune system to eliminate precancerous senescent cells and promotes tissue repair (33). By contrast, negative regulation involves the secretion of soluble factors, such as ILs, chemokines, growth factors and degradative enzymes, including matrix metalloproteinases (MMPs), as well as insoluble proteins/extracellular matrix (ECM) components of the SASP. These factors can influence the tumor microenvironment (TME) to promote tumor progression (34). The TME is a complex, dynamic environment formed from the interactions of tumor cells with immune cells, mesenchymal stromal cells and the extracellular environment, such as the ECM and soluble biomolecules secreted by tumor cells (35). The effects of the SASP on the PCa microenvironment are predominantly in the form of immune reactivity and matrix or vascular remodeling (36). IL-6 can recruit myeloid-derived suppressor cells into the TME; these cells can block IL-1-mediated cellular senescence and reduce immune surveillance by inhibiting antitumor cells, such as CD8+ T cells and natural killer (NK) cells (37). Furthermore, in addition to direct immunomodulation through the release of cytokines and chemokines, the SASP may indirectly affect immunoreactivity by affecting other stromal cells in the TME, particularly endothelial cells (38). Senescent cells produce high levels of proangiogenic SASP factors from the VEGF, platelet-derived growth factor and fibroblast growth factor families, which mediate neointimal formation and vascular remodeling, thereby promoting cancer development (39).
Overall, the production of SASP factors is diverse and dynamic. A number of SASP factors exhibit distinct functions, although even the same SASP factors can exert opposing effects on tumors at different stages. Resolving the contradictory mechanisms between the SASP and PCa treatment in important to advance research on the therapeutic strategies for metastatic PCa in the future.
Association between senescence and PCa
Senescence inhibits PCa growth
In the early-stage of PCa progression, treatment-induced SASP, such as that caused by chemotherapy and radiotherapy, evolves from an ASAP, which typically appears 5–8 days after the initiation of PCa treatment (11). Core factors of the SASP include IL-6, IL-8, colony stimulating factor 1 and chemokine (C-C motif) ligand 2. The p53 and NF-κB signaling pathways can synergistically regulate these SASP factors, activate macrophages to form an oncogenic microenvironment, which promotes senescence of tumor cells, prevent the proliferation and division of senescent tumor cells and increase the oncogenic effect of PCa therapeutic drugs (40).
Existing PCa treatments, such as ADT, radiotherapy and chemotherapy, have been shown to induce senescence of PCa cells, a phenomenon termed treatment-induced senescence (TIS) (41). TIS can lead to reduced or enhanced tumor growth, and investigation of its role in a number of treatments could help to address drug resistance in PCa therapy (42). Initially, advanced stage PCa can be effectively treated with ADT, which rapidly diminishes serum testosterone levels through the reduction of testicular androgen production or inhibition of the androgen receptor (AR). This is achieved by the modulation of luteinizing hormone-releasing hormone (LHRH) production or activity using LHRH agonists, such as goserelin, leuprolide and trenbolone, or LHRH antagonists, such as degareli (43). Gilbert et al (44) demonstrated an association between the tumor suppressor mechanism of ADT and the induction of senescence in an IKKε-deficient PC-3 cell line and a xenograft PCa mouse model. Pernicová et al (45) reported that ADT may regulate the tissue microenvironment through senescent cells, which potentially promote the development of androgen-independent PCa. Mirzakhani et al (20) demonstrated the effects of the interaction between the AR and long non-coding (lnc)RNASAT1 on chromatin level using RNA-chromatin immunoprecipitation experiments, and identified a novel AR-lncRNASAT1-AKT-p15INK4b signaling axis that may mediate supraphysiological androgen level-induced cellular senescence. Coppé et al (22) analyzed biopsy samples from patients with PCa who were treated with the antitumor drug mitoxantrone and demonstrated upregulation of the SASP factors IL-6 and IL-8. In addition, Blute et al (46) demonstrated that in patients with PCa treated with chemotherapy and ADT, the reactive oxygen species-ERK-ETS-p16INK4a and p27Kip1-retinoblastoma protein pathways were activated to induce senescence. This highlights the variation in intracellular signaling within PCa cells that can lead to the induction of TIS.
In addition to the diverse aforementioned actions of the SASP, senescent cells also exert their tumor-suppressive effects through the activation of immune surveillance pathways. Oncogene-induced cellular senescence (OIS) represents a specific type of senescence mechanism during tumorigenesis, which inhibits the oncogenic transformation of tumors, such as prostate, ovarian and colorectal cancer, and serves as an initial barrier to cancer development in vivo (47–49). OIS is triggered by the activation of oncogenes, such as Ras and BRAF, or the inactivation of tumor suppressor genes, such as PTEN (50). In OIS, oncogene activation induces DNA damage, which in turn activates p53 and leads to senescence. By contrast, PTEN deletion-induced cellular senescence lacks apparent DNA damage and, in mouse models and human tumor xenograft models, p53 may be activated via the PI3K/AKT/mTOR pathway after PTEN deletion (51). The tumor suppressor gene PTEN is frequently absent or mutated in human PCa, which leads to the activation of the PI3K/AKT signaling pathway and tumorigenesis (52). In addition, PTEN has an AKT-independent function and directly interacts with p53 to regulate its transcriptional activity and stability (53). Parisotto et al (54) demonstrated that PTEN deficiency in adult mouse prostate luminal epithelial cells stimulated PCa epithelial proliferation, followed by progressive growth arrest characterized by cellular senescence. In a PCa mouse model, Chen et al (55) demonstrated that double mutant mice with specific inactivation of PTEN and p53 in the prostate epithelium consistently developed PCa and died by 7 months of age. This was attributed to the acceleration of tumor progression in PTEN-deficient mice through promoting hyperproliferation and transformation, and eliminating tumor cell senescence.
Overall, PTEN loss-induced progression of PCa intraepithelial neoplasia is counteracted by cellular senescence in mouse models of PCa. However, due to the replicative stress associated with PTEN deletion, strategies to promote PTEN loss-induced senescence are high-risk in cancer prevention and treatment (54). The aforementioned studies provide in vivo evidence to support the role of OIS as a key ‘brake’ on prostate tumorigenesis. At the same time, senescent cells can induce senescence in neighboring cells through the SASP and direct cell-to-cell interactions, which limit the proliferation of nearby precancerous or fully malignant cells that have not yet undergone senescence and enhances the tumor suppressor function of senescent cells (56). In certain instances, SASP factors in the TME recruit a variety of immune cells to eliminate tumor cells undergoing senescence, which inhibit tumor cell growth. For example, senescent tumor cells secrete IL-6, IL-8 and insulin-like growth factor binding protein 7 into the TME, which initiates a proinflammatory response. These factors recruit immune cells, such as T lymphocytes, to the site of tumorigenesis, where these immune cells recognize and eliminate senescent and tumor cells, inhibit tumor growth and impede tumor progression (57). In summary, although the molecular pathways that induce cell cycle arrest can vary among different therapies, their tumor suppression mechanisms are generally related to senescence.
Senescence promotes PCa metastasis
In the later stages of PCa, radiotherapy, chemotherapy and targeted therapies result in a large number of senescent cells remaining in the body; the continuous accumulation of senescent cells promotes the formation of a senescent microenvironment, which enhances SASP secretion and senescence of immune cells, which can lead to metastasis and drug resistance of PCa (11). Notably, there is experimental evidence on the role of SASP factor components in promoting PCa proliferation. In the present review, the SASP regulatory and effector factors associated with PCa metastasis pathways, the targeting of these pathways and the impact of the regulation of SASP factors on metastatic PCa treatment is discussed.
IL-6 and IL-8
IL-6 and IL-8 are two proinflammatory cytokines, and the main components of the SASP in human senescent fibroblasts, which have a notable impact on PCa metastasis (58). High levels of IL-6 and IL-8 have been indicated as biomarkers for metastatic PCa, which promote angiogenesis and are closely related to the colonization of metastatic PCa (59). González-Ochoa et al (60) demonstrated a positive association between the secretion levels of IL-8 and IL-6, and the invasiveness and metastatic potential of PCa using the DU145 PCa cell line. In addition, IL-6 was shown to promote PCa metastasis by facilitating tumor cell EMT. Méndez-Clemente et al (61) demonstrated that IL-6 receptor (IL-6R)-mediated IL-6-dependent STAT3 activation, through the formation of an IL-6/IL-6R/STAT3 feedback loop, could promote EMT in PCa cells. In summary, IL-6 and IL-8 are two marked contributors to PCa metastasis.
MMPs
Members of the MMP superfamily are a component of the SASP. MMPs, secreted by stromal cells, serve an integral role in the progression of PCa and are closely associated with bone metastasis of PCa (62). Park et al (63) reported that MMPs, such as MMP2 and MMP9, can mediate the degradation of the ECM. This process can increase the protein expression levels of MMPs and VEGF to promote angiogenesis and thus tumor growth (64). MT1-MMP, a transmembrane member of the MMP family, is upregulated as PCa progresses from normal progression to prostatic intraepithelial neoplasia to invasive cancer, which indicates its role in the invasive process of prostate adenocarcinoma (65). Wei et al (66) reported that MMP activity, particularly through MMP2 and MMP9, is associated with the invasive and metastatic potential of cancer cells. It was reported that osteonectin induces MMP activity, which led to the degradation of the ECM and enabling the invasion of cancer cells. In cell specimens obtained from metastatic lesions in the bone marrow of patients with PCa, Miftakhov et al (67) demonstrated that MMP production in bone marrow could create a suitable microenvironment for the metastatic growth of PCa cells. In a previous study, it was revealed that Timp1 deletion led to the activation of downstream MMPs, which reprogrammed and initiated the SASP in PTEN-deficient cells; furthermore, PTEN, and TIMP1-knockout cells could promote the migration of non-senescent mouse and human tumor cells by paracrine means (68). Therefore, TIS should potentially be used with caution in patients with PCa and a background of TIMP1 gene deletion, to spare a number of patients from the toxicity of chemotherapy. The aforementioned studies demonstrate that MMPs may influence PCa metastasis and further studies should be conducted in the future to identify the specific pathways involved in the metastatic transformation of primary tumors, which could serve to develop strategies to prevent the development of metastatic PCa.
P53
P53 is a key regulator of SASP expression (22,69). Studies of the integrated transcriptomic and genomic characterization of PCa have shown that the p53 gene is one of the most commonly mutated driver genes in primary PCa and p53 mutations occur at a high frequency in patients with metastatic PCa (70). The mutation or activation of the PTEN gene is strongly associated with activation of p53 signaling. An analysis of primary and metastatic PCa samples by Aggarwal et al (71) demonstrated that PTEN deletion resulted in decreased expression or inactivation of p53 in metastatic tumors, accompanied by increased PI3K/AKT signaling. Wanjala et al (72) developed a genetically engineered mouse model of PCa and demonstrated that the inactivation of PTEN and p53 promoted the development of an aggressive phenotype of PCa, characterized by accelerated tumor growth, increased invasion and enhanced development of metastatic disease, which resembles advanced stage PCa in humans. A follow-up of one patient with CRPC, who initially presented with nine prostate tumor foci, demonstrated that only tumor clones with mutations in PTEN, p53 and SPOP acquired additional genetic alterations and resulted in fatal metastatic tumors over 17 years of tumor progression (73). Recently, Ding et al (74) demonstrated that gain-of-function mutations in p53 and erythroblast transformation-specific-related gene (ERG) can collectively promote the β-catenin signaling pathway and increase the expression levels of pyrimidine synthesis-related genes, which contributes to the progression of PCa. The aforementioned study provides insights into the gene expression profile of a patient with PCa carrying both p53 mutations and ERG fusions, and suggests a novel direction for precision treatment of this molecular subtype of PCa. In summary, the downregulation of p53 may be considered a hallmark feature in metastatic PCa, which suggests a close association between PCa metastasis and p53.
mTOR
As an additional crucial regulator of the SASP, mTOR serves a pivotal role in promoting the SASP through its involvement in translation. A previous study demonstrated that both mTOR complex (mTORC)1 and mTORC2 are frequently overactivated in PCa cells, and are associated with the process of cancer metastasis (75). The oncogenic role of PI3K-AKT-mTOR signaling and the common genetic alterations in this pathway are well established (76). Shi et al (77) identified a novel circular (circ)RNA, circMBOAT29, the overexpression of which led to the activation of the PI3K/Akt pathway through the upregulation of mTOR, which promoted the metastasis of PCa. Since PCa is a highly heterogeneous tumor and its biological behavior may vary within the same stage of disease, the efficacy of these compounds may also vary. Based on the expression characteristics and mechanisms of circRNAs in PCa, it may be possible to utilize precision therapy for tumor treatment and develop personalized treatment plans for individual patients (78). Existing mTORC1 inhibitors are effective in the treatment of PCa (79). mTORC1 inhibitors, such as temsirolimus and everolimus, are U.S. Food and Drug Agency (FDA)-approved for phase III clinical trials in kidney cancer, neuroendocrine tumors and metastatic breast cancer (80). Ongoing clinical trials investigating the combination of mTORC1 inhibitors with standard anticancer therapies, such as chemotherapy and ADT, in PCa treatment have achieved improved efficacy compared to the therapies alone (81–83). Rapamycin, a selective inhibitor of mTORC1, is well tolerated in patients with PCa according to preliminary analysis from a number of clinical trials, as mTORC1 displays sensitivity to rapamycin treatment (84,85). The therapeutic benefit of the mTORC1/2 inhibitor rapalink-1 has been reported in patient-derived organoid and xenograft models of bone metastatic PCa, which supports the involvement of the mTOR pathway in bone metastatic cancers (86). In addition, mTORC2 is required for PTEN loss-induced PCa development in mice and serves a central role in mediating PI3K-dependent carcinogenesis (87). mTORC2 signaling promotes the growth of PTEN-deficient PCa, which indicates that mTORC2 inhibition may be clinically effective in PTEN-deficient PCa (88). This suggests that targeting mTORC2 in patients with PTEN-deficient PCa may potentially be able to provide novel therapeutic strategies for those patients with PCa. To the best of our knowledge, there is no effective treatment for metastatic PCa, in particular for PCa cases where hormonal ablation therapy has failed, and highly selective drugs that target the PI3K-AKT-mTOR pathway have a more favorable therapeutic index in such cases (89). However, off-target effects of the dual inhibition of PI3K and mTOR pathways may cause unacceptable toxicity with dose-dependent adverse effects and drugs with more favorable therapeutic indexes, as well as the efficacy and appropriate dosage of different drug combinations, should be explored in future research.
NF-κB
NF-κB is required to achieve full transcriptional activation of the SASP. Activation of the NF-κB pathway results in enhanced secretion of proinflammatory cytokines and chemokines from senescent cells, resulting in the SASP (90). Li et al (91) identified that the IKKβ/AT-rich interaction domain 1A/NF-κB feedback axis integrates inflammation and immunosuppression to promote PCa progression, which supports the suggestion that anti-NF-κB antibodies or targeting IL-8 receptor b, in combination with immune checkpoint blockade therapies, such as blockade of PD-1 binding to PD-L1, might serve as a therapeutic strategy for advanced PCa. Previous studies have demonstrated that NF-κB expression not only promotes the expression of cell adhesion molecules, but also promotes the expression of molecules that facilitate tumor metastasis, which results in increased resistance to tumor therapy (92). A recent study demonstrated that NF-κB activation is required for tumor cell EMT, the process through which tumor epithelial cells acquire mesenchymal features, and become highly invasive and metastatic (93). Inhibition of NF-κB signaling prevents EMT, which reduces the metastatic potential of PCa. In addition, NF-κB regulates metastatic activity by controlling the transcriptional activity of MMPs and angiogenic enzymes (94). Ayala et al (95) conducted a neoadjuvant clinical trial of bortezomib, an NF-κB inhibitor, in male patients with PCa classed as having a high risk of recurrence and demonstrated that bortezomib was generally safe preoperatively and that, in vitro, the combination of bortezomib and perifosine, an AKT inhibitor, was more effective compared with either therapy alone. It was previously reported that the FDA-approved therapeutic combinations of artesunate (AS) or NF-κB inhibitors with AR antagonists may improve the clinical efficacy of treatment in patients with CRPC (96). Future clinical trials using AS and AR antagonist therapies in patients with CRPC are needed for further research advances. In summary, the activation of the NF-κB pathway may promote PCa metastasis through a number of mechanisms, such as enhancing immunosuppression, expression of tumor metastatic molecules and the promotion of angiogenesis by facilitating the development of EMT.
Small extracellular vesicles (sEVs)
In addition to cytokines, sEVs have emerged as contributors to PCa metastasis, as part of the SASP (97). sEVs constitute a diverse population of membrane-secreting vesicles containing exosomes (98). PCa cell-derived sEVs contain cytokines that stimulate the differentiation of bone marrow mesenchymal stem cells into myofibroblasts (99). These sEVs release elevated expression levels of VEGF-A, hepatocyte growth factor and MMPs that possess proangiogenic properties, which increase tumor cell proliferation and metastasis (100). Ma et al (101) analyzed the microRNA (miR) profiles of tumor-derived sEVs and osteoclasts together and reported that miR-152-3p, carried by PCa-derived sEVs, could transmit osteolytic signals from tumor cells to osteoclasts; this process may promote osteolysis in bone metastasis. sEVs may serve as biomarkers of PCa progression, and represent potential therapeutic targets in the prediction and prevention of PCa metastasis.
Therapeutic approaches and future outlook
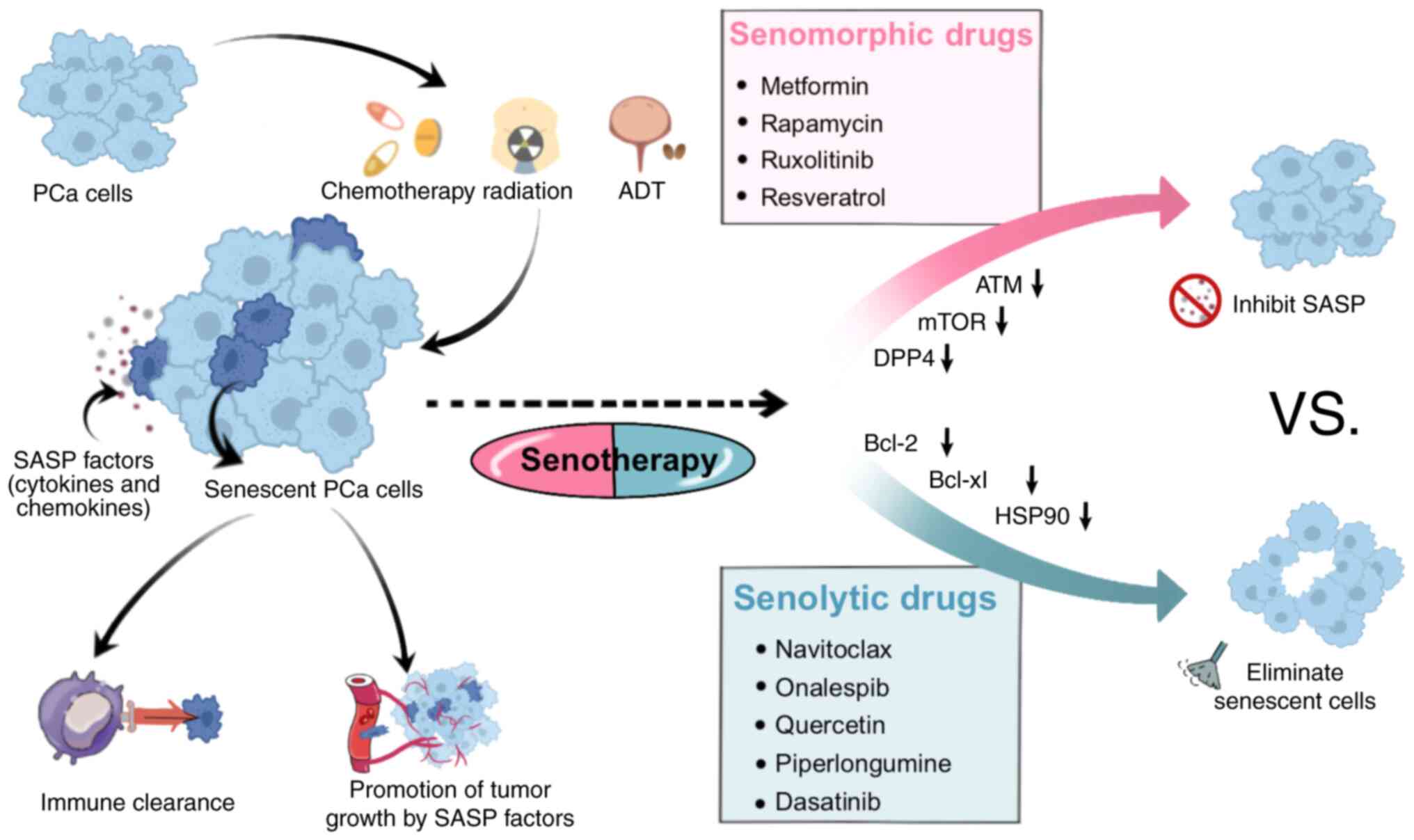
The use of senescence-inducing therapy as a standalone treatment for tumors is currently controversial. A number of studies have shown that the SASP can exhibit a dual effect: i) Inhibition of the tumor growth by inducing paracrine senescence in tumor cells through inflammatory mediator-containing extracellular vesicle-mediated mechanisms; and ii) influencing the TME through the secretion of inflammatory cytokines and chemokines, which potentially promotes the progression of tumors. In recent years, research has been devoted to the emerging concept of ‘senotherapy’ (102–104) (Fig. 3). Senotherapy involves initially inducing senescence in tumor cells through radiotherapy and chemotherapy to exert tumor-suppressing effects. Subsequently, adjuvant drugs are employed to eliminate senescent cells, preventing the release of SASP factors. This dual-phase strategy aims to avoid the possible tumor-promoting effects of senescent cells (105). Through the use of adjuvant drugs to eliminate senescent cells and inhibit the release of SASP factors from these cells, the potential tumor-promoting effect of senescent cells is effectively avoided (106). Current therapeutic approaches for PCa have been expanded to include the use of senomorphic drugs to mitigate SASP activity and senolytic drugs to eliminate senescent cells (107). Contemporary senotherapeutic drugs not only inhibit the SASP, but also leverage the characteristics of senescent cells to enhance the ability of the immune system to clear senescent cells. For example, the application of chimeric antigen receptor-T targeting antigens specific to senescent cells allows for selective removal of these senescent cells (108). While clinical trials validating the efficacy of chimeric antigen receptor-T targeting antigens specific to senescent cells in PCa treatment are still lacking, the results of the aforementioned preclinical studies provide support for the integration of senotherapeutic drugs with conventional radiotherapy in the management of metastatic PCa (105). The range of drugs used for the treatment of senescent cells are listed in Table I (109–129), providing an overview of the current drug mechanisms of action in the context of PCa treatment.
Senolytics
Senolytics are a class of drugs designed to induce apoptosis specifically in senescent cells, and have potential as a treatment for PCa (130). It has been suggested that the potential benefits of senolytic therapy are through the intrinsic anti-aging system present in organisms, i.e. the immune surveillance of senescent cells (131). Transgenic animal models for in vivo senescence studies mainly include INK-ATTAC, p16-3MR, p16-Cre and p21-ATD mice (132–135), and have shown that multiple components of the immune system, including NK cells, T cells and macrophages, are involved in controlling and eliminating the presence of senescent cells in tissues (26). Pathways implicated in this process include activation of the PI3K/AKT and/or Bcl-2/Bcl-xl pathways (11). Zhan et al (136) reported evidence from two cell models with deletions in the ATRX gene (a common molecular marker for glioma) suggesting the efficacy of senolytic drugs in targeting senescent tumor cells and precancerous cells. Arai et al (109) demonstrated the potential of BH3 mimetics, a novel class of antitumor drugs that targets Bcl-2 family proteins, as a monotherapy or in combination with other agents for the treatment of PCa cells. A combination of navitoclax, a BH3 mimetic, with taxane-based chemotherapy, such as docetaxel and paclitaxel, increased the rate of apoptotic cell death in human PCa cells compared with the drug alone. In a murine model of PCa with PTEN deficiency, Guccini et al (68) demonstrated that the absence of TIMP1 shifted senescence from a tumor-suppressive process to a metastasis-promoting process. Furthermore, the elimination of senescent cells using senolytic Bcl-2 inhibitors attenuated this transition. Mechanistically, the loss of TIMP1 reprograms the SASP of senescent tumor cells by activation of MMPs (68). The deletion of TIMP1, either alone or in PTEN and TIMP1 double deletions, is common in PCa and is associated with docetaxel resistance (137). A major limitation of navitoclax identified in early senolytic experiments was the risk of thrombocytopenia when used in excess, which limited its use (138). However, it has been demonstrated that the platelet toxicity can be reduced by converting navitoclax to PZ15227, with the use of protein hydrolysis-targeted chimerism technology, through a conversion process that reduces its toxicity and improves its effectiveness against senescent cells, which results in the regeneration of tissue stem and progenitor cells in senescent mice (139). This further supports the future feasibility of navitoclax in combination with conventional radiotherapy agents for the treatment of metastatic PCa (140). Bioinformatics analysis of whole transcriptome RNA sequencing data performed by Ferraldeschi et al (141) confirmed that the second-generation heat shock protein 90 (HSP90) inhibitor, onalespib, altered the splicing of ≥557 genes, including AR, in PCa cells, which may be beneficial for PCa and suggests HSP90 inhibitors as a class of drugs that could potentially be evaluated further in metastatic PCa in the future. Lu et al (113) demonstrated that the HSP90 inhibitor quercetin could reverse proliferation, colony formation, migration, invasion and apoptosis resistance to doxorubicin in PCa, using doxorubicin-resistant cells (LNCaP/R, PC-3/R) established from doxorubicin-sensitive cell lines (LNCaP, PC-3). A recent study reported that the retinoic acid receptor agonist adapalene can be used as a new senolytic agent, and that in a preclinical mouse model of PCa, the combination of adapalene and docetaxel could promote a tumor-suppressive SASP, which prompts NK cells to mediate tumor clearance more effectively than either drug alone (142). This supports the therapeutic potential of senolytic therapy in PCa and provide insights into the mitigation of the side effects associated with senolytic therapy.
Currently, there are >20 clinical trials related to senolytics. Due to a limited understanding of the side effects of senolytics in humans, a large proportion of clinical trials have been conducted in patients with serious health conditions, which aimed to optimize the benefit-risk ratio. A number of studies have suggested that senolytics contribute positively to the improvement of somatic function, which reduces senescent cellular load and ameliorates inflammatory states (143–146); however, a number of studies have reported unsuccessful outcomes (147,148). For example, Spetsieris et al (149) conducted a phase II clinical trial of abiraterone followed by randomized assignment to the addition of dasatinib or sunitinib, for the treatment of patients with metastatic desmoplasia-resistant PCa. No difference in overall survival or time to treatment failure between dasatinib and sunitinib in combination with abiraterone for the treatment of patients with metastatic CRPC in the bone was found. In the future, preclinical studies are needed to further elucidate the markers and mechanisms of action of senescence in patients with PCa, and to perform more extensive validation of senolytic drugs, to prioritize disease-specific drug candidates for clinical use.
Senomorphic compounds
Senomorphic compounds, also referred to as SASP inhibitors, can modulate the senescence phenotype and inhibit generation of the SASP without eliminating senescent cells (150). Representative drugs categorized as senomorphic compounds include rapamycin and metformin, which can directly or indirectly attenuate the SASP in senescent cells by inhibiting NF-κB, the JAK-STAT signaling pathway and the serine/threonine protein kinase mTOR (151). Currently, a large proportion of the data on the efficacy of senotherapeutic drugs come from in vitro experiments using human cell cultures. Studies have shown that the use of metformin after ADT induces apoptosis, attenuates mTOR activation and reduces the number of senescent cells in PCa in vitro and in vivo (122). Several preclinical studies have also indicated that the mTOR inhibitor rapamycin can inhibit the androgen-dependent growth of human PCa cells by downregulating the expression levels of AR-activated downstream genes (100–102). The findings from the aforementioned trials suggest that senescence therapies may provide novel leads for the treatment of metastatic PCa and desmoplasia-resistant PCa. In addition, specific JAK/STAT small molecule inhibitors, such as ruxolitinib and fludarabine, have demonstrated efficacy in the treatment of both curative and desmoplasia-resistant PCa (127).
In general, senolytic therapies may offer additional advantages compared with senomorphic therapies. Senolytic drugs are not only effective, but because they administered intermittently, the side effects are effectively controlled and they are considered to be safe (152). Unlike senolytic drugs that are administered intermittently, a large proportion of SASP inhibitors require continuous treatment to maintain SASP inhibition, which potentially increases the occurrence of side effects associated with the drug (153). Secondly, the complete removal of all senescent cells or total inhibition of the SASP may be detrimental in a number of cases (154,155), as it can cause chronic inflammation, immunosuppression, stimulate the EMT, and even promote tumor migration and metastasis (156). By contrast, senolytic drugs have the advantage of specifically targeting senescent cells at the site of the lesion and can induce apoptosis in these cells. Future drug studies should prioritize intervening in senescent cells consistently expressing tissue-damaging SASP, and aim to develop drugs with enhanced therapeutic potential and minimized off-target effects. Novel therapeutic options could potentially be based on the expression characteristics and mechanisms of circRNAs in PCa, thus bringing PCa tumor treatment into a precision therapy paradigm. Rigorous clinical trials are necessary to demonstrate the safety and efficacy of senolytics or SASP inhibitors in PCa; despite currently available preclinical data, advancements in relevant research in this field are challenging.
Summary
In summary, the microenvironment of senescence can markedly influence prostate carcinogenesis and metastasis, which highlights the importance of cellular senescence in PCa treatment. The intricate composition of SASP factors contributes to the dual impact of senescence, which acts as both a tumor suppressor and promoter. Effectively harnessing the tumor-suppressive effects of SASP, while mitigating its tumor-promoting effects, remains a challenge in this field. Currently, the main obstacle in the treatment of metastatic PCa is the development of resistance to existing therapies and progression to an incurable state. Future research should focus on identifying the intricate associations among cellular senescence, the SASP and the TME. A comprehensive exploration into the types of PCa cell senescence induced by drug treatments, along with the investigation of specific molecular mechanisms and their interactions with the immune microenvironment could potentially advance the current understanding of PCa, and has ramifications for the precise treatment of metastatic PCa, alleviation of chemotherapy resistance, minimization of the toxic side effects of chemotherapy drugs and enhancement of therapeutic outcomes for metastatic PCa in the future.
Acknowledgements
Not applicable.
Funding
The present review was supported by the College Students' Innovation and Entrepreneurship Training Program (grant no. S202210660072) and the Undergraduate Teaching Research Project of Guizhou Medical University (grant no. JG2023046). Partial support was provided by the Natural Science Foundation Project of Guizhou Provincial Science and Technology Department (grant no. Qiankehe Foundation-ZK 2024 General 182).
Availability of data and materials
Not applicable.
Authors' contributions
CJ, SL and GL wrote the original draft and prepared the figures. DY and GG wrote, reviewed and edited the manuscript. BG, ZY and JX edited the review and were responsible for editing the references. All authors have read and approved the final version of the manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
|
Xie J, Xiao X, Dong Z and Wang Q: The systemic inflammation score is associated with the survival of patients with prostate cancer. J Inflamm Res. 16:963–975. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Siegel RL, Miller KD, Wagle NS and Jemal A: Cancer statistics, 2023. CA Cancer J Clin. 73:17–48. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Tan Y, Wang L, Du Y, Liu X, Chen Z, Weng X, Guo J, Chen H, Wang M and Wang X: Inhibition of BRD4 suppresses tumor growth in prostate cancer via the enhancement of FOXO1 expression. Int J Oncol. 53:2503–2517. 2018.PubMed/NCBI | |
|
Dong D, Zhang L, Bai C, Ma N, Ji W, Jia L, Zhang A, Zhang P, Ren L and Zhou Y: UNC5D, suppressed by promoter hypermethylation, inhibits cell metastasis by activating death-associated protein kinase 1 in prostate cancer. Cancer Sci. 110:1244–1255. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Belmonte M, Saia G, Zugni F, Alessi S, Colombo A, Summers PE, Luzzago S, Marvaso G, Musi G, De Cobelli O, et al: The role of MRI in the management of a prostate cancer patient with bone and lymph nodes metastases. A case report. Acta Biomed. 92:e20212142021.PubMed/NCBI | |
|
Witt K, Evans-Axelsson S, Lundqvist A, Johansson M, Bjartell A and Hellsten R: Inhibition of STAT3 augments antitumor efficacy of anti-CTLA-4 treatment against prostate cancer. Cancer Immunol Immunother. 70:3155–3166. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Sun CY, Talukder M, Cao D and Chen CW: Gilteritinib enhances Anti-tumor efficacy of CDK4/6 inhibitor, abemaciclib in lung cancer cells. Front Pharmacol. 13:8297592022. View Article : Google Scholar : PubMed/NCBI | |
|
Rysanek D, Vasicova P, Kolla JN, Sedlak D, Andera L, Bartek J and Hodny Z: Synergism of BCL-2 family inhibitors facilitates selective elimination of senescent cells. Aging. 14:6381–6414. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Rhinn M, Zapata-Bodalo I, Klein A, Plassat JL, Knauer-Meyer T and Keyes WM: Aberrant induction of p19Arf-mediated cellular senescence contributes to neurodevelopmental defects. PLoS Biol. 20:e30016642022. View Article : Google Scholar : PubMed/NCBI | |
|
Alessio N, Aprile D, Squillaro T, Di Bernardo G, Finicelli M, Melone MA, Peluso G and Galderisi U: The senescence-associated secretory phenotype (SASP) from mesenchymal stromal cells impairs growth of immortalized prostate cells but has no effect on metastatic prostatic cancer cells. Aging (Albany NY). 11:5817–5828. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Kallenbach J, Atri Roozbahani G, Heidari Horestani M and Baniahmad A: Distinct mechanisms mediating therapy-induced cellular senescence in prostate cancer. Cell Biosci. 12:2002022. View Article : Google Scholar : PubMed/NCBI | |
|
Mori JO, Elhussin I, Brennen WN, Graham MK, Lotan TL, Yates CC, De Marzo AM, Denmeade SR, Yegnasubramanian S, Nelson WG, et al: Prognostic and therapeutic potential of senescent stromal fibroblasts in prostate cancer. Nat Rev Urol. 21:258–273. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Takemoto K, Kobatake K, Miura K, Fukushima T, Babasaki T, Miyamoto S, Sekino Y, Kitano H, Goto K, Ikeda K, et al: BACH1 promotes clear cell renal cell carcinoma progression by upregulating oxidative stress-related tumorigenicity. Cancer Sci. 114:436–448. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Hayflick L and Moorhead PS: The serial cultivation of human diploid cell strains. Exp Cell Res. 25:585–621. 1961. View Article : Google Scholar : PubMed/NCBI | |
|
Huang Y, Ge MX, Li YH, Li JL, Yu Q, Xiao FH, Ao HS, Yang LQ, Li J, He Y and Kong QP: Longevity-associated transcription factor ATF7 promotes Healthspan by suppressing cellular senescence and systematic inflammation. Aging Dis. 14:1374–1389. 2023.PubMed/NCBI | |
|
Ngoi NY, Liew AQx, Chong SJF, Davids MS, Clement MV and Pervaiz S: The redox-senescence axis and its therapeutic targeting. Redox Biol. 45:1020322021. View Article : Google Scholar : PubMed/NCBI | |
|
Park SS, Choi YW, Kim JH, Kim HS and Park TJ: Senescent tumor cells: An overlooked adversary in the battle against cancer. Exp Mol Med. 53:1834–1841. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Alhaddad L, Nofal Z, Pustovalova M, Osipov AN and Leonov S: Long-term cultured human glioblastoma multiforme cells demonstrate increased radiosensitivity and senescence-associated secretory phenotype in response to irradiation. Int J Mol Sci. 24:20022023. View Article : Google Scholar : PubMed/NCBI | |
|
Wu Z, Uhl B, Gires O and Reichel CA: A transcriptomic pan-cancer signature for survival prognostication and prediction of immunotherapy response based on endothelial senescence. J Biomed Sci. 30:212023. View Article : Google Scholar : PubMed/NCBI | |
|
Mirzakhani K, Kallenbach J, Rasa SMM, Ribaudo F, Ungelenk M, Ehsani M, Gong W, Gassler N, Leeder M, Grimm MO, et al: The androgen receptor-lncRNASAT1-AKT-p15 axis mediates androgen-induced cellular senescence in prostate cancer cells. Oncogene. 41:943–959. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Birch J and Gil J: Senescence and the SASP: Many therapeutic avenues. Genes Dev. 34:1565–1576. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Coppé JP, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J, Nelson PS, Desprez PY and Campisi J: Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6:2853–2868. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Georgilis A, Klotz S, Hanley CJ, Herranz N, Weirich B, Morancho B, Leote AC, D'Artista L, Gallage S, Seehawer M, et al: PTBP1-mediated alternative splicing regulates the inflammatory Secretome and the Pro-tumorigenic effects of senescent cells. Cancer Cell. 34:85–102.e9. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Yue Z, Nie L, Zhao P, Ji N, Liao G and Wang Q: Senescence-associated secretory phenotype and its impact on oral immune homeostasis. Front Immunol. 13:10193132022. View Article : Google Scholar : PubMed/NCBI | |
|
Liu L, Yue X, Sun Z, Hambright WS, Wei J, Li Y, Matre P, Cui Y, Wang Z, Rodney G, et al: Reduction of senescent fibro-adipogenic progenitors in progeria-aged muscle by senolytics rescues the function of muscle stem cells. J Cachexia Sarcopenia Muscle. 13:3137–3148. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Khalil R, Diab-Assaf M and Lemaitre JM: Emerging therapeutic approaches to target the dark side of senescent cells: New hopes to treat aging as a disease and to delay age-related pathologies. Cells. 12:9152023. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang L, Pitcher LE, Yousefzadeh MJ, Niedernhofer LJ, Robbins PD and Zhu Y: Cellular senescence: A key therapeutic target in aging and diseases. J Clin Invest. 132:e1584502022. View Article : Google Scholar : PubMed/NCBI | |
|
Duan D, Shang M, Han Y, Liu J, Liu J, Kong SH, Hou J, Huang B, Lu J and Zhang Y: EZH2-CCF-cGAS axis promotes breast cancer metastasis. Int J Mol Sci. 23:17882022. View Article : Google Scholar : PubMed/NCBI | |
|
Huang M, Cha Z, Liu R, Lin M, Gafoor NA, Kong T, Ge F and Chen W: Enhancing immunotherapy outcomes by targeted remodeling of the tumor microenvironment via combined cGAS-STING pathway strategies. Front Immunol. 15:13999262024. View Article : Google Scholar : PubMed/NCBI | |
|
Lee KS, Lin S, Copland DA, Dick AD and Liu J: Cellular senescence in the aging retina and developments of senotherapies for age-related macular degeneration. J Neuroinflammation. 18:322021. View Article : Google Scholar : PubMed/NCBI | |
|
Toso A, Revandkar A, Di Mitri D, Guccini I, Proietti M, Sarti M, Pinton S, Zhang J, Kalathur M, Civenni G, et al: Enhancing chemotherapy efficacy in Pten-Deficient prostate tumors by activating the senescence-associated antitumor immunity. Cell Rep. 9:75–89. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Parry AJ, Hoare M, Bihary D, Hänsel-Hertsch R, Smith S, Tomimatsu K, Mannion E, Smith A, D'Santos P, Russell IA, et al: NOTCH-mediated non-cell autonomous regulation of chromatin structure during senescence. Nat Commun. 9:18402018. View Article : Google Scholar : PubMed/NCBI | |
|
Tan SYX, Zhang J and Tee WW: Epigenetic regulation of inflammatory signaling and inflammation-induced cancer. Front Cell Dev Biol. 10:9314932022. View Article : Google Scholar : PubMed/NCBI | |
|
Chandrasekaran A, Idelchik MDPS and Melendez JA: Redox control of senescence and age-related disease. Redox Biol. 11:91–102. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Takasugi M, Yoshida Y and Ohtani N: Cellular senescence and the tumour microenvironment. Mol Oncol. 16:3333–3351. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Wan R, Long S, Ma S, Yan P, Li Z, Xu K, Lian H, Li W, Duan Y, Zhu M, et al: NR2F2 alleviates pulmonary fibrosis by inhibition of epithelial cell senescence. Respir Res. 25:1542024. View Article : Google Scholar : PubMed/NCBI | |
|
Zhao B, Wu B, Feng N, Zhang X, Zhang X, Wei Y and Zhang W: Aging microenvironment and antitumor immunity for geriatric oncology: the landscape and future implications. J Hematol OncolJ Hematol Oncol. 16:282023. View Article : Google Scholar : PubMed/NCBI | |
|
Hwang HJ, Lee YR, Kang D, Lee HC, Seo HR, Ryu JK, Kim YN, Ko YG, Park HJ and Lee JS: Endothelial cells under therapy-induced senescence secrete CXCL11, which increases aggressiveness of breast cancer cells. Cancer Lett. 490:100–110. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Chibaya L, Snyder J and Ruscetti M: Senescence and the tumor-immune landscape: Implications for cancer immunotherapy. Semin Cancer Biol. 86:827–845. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Volonte D and Galbiati F: Caveolin-1, a master regulator of cellular senescence. Cancer Metastasis Rev. 39:397–414. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Pardella E, Pranzini E, Nesi I, Parri M, Spatafora P, Torre E, Muccilli A, Castiglione F, Fambrini M, Sorbi F, et al: Therapy-induced stromal senescence promoting aggressiveness of prostate and ovarian cancer. Cells. 11:40262022. View Article : Google Scholar : PubMed/NCBI | |
|
Xu MY, Xia ZY, Sun JX, Liu CQ, An Y, Xu JZ, Zhang SH, Zhong XY, Zeng N, Ma SY, et al: A new perspective on prostate cancer treatment: The interplay between cellular senescence and treatment resistance. Front Immunol. 15:13950472024. View Article : Google Scholar : PubMed/NCBI | |
|
Meng F, Han X, Min Z, He X and Zhu S: Prognostic signatures associated with high infiltration of Tregs in bone metastatic prostate cancer. Aging. 13:17442–17461. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Gilbert S, Péant B, Malaquin N, Tu V, Fleury H, Leclerc-Desaulniers K, Rodier F, Mes-Masson AM and Saad F: Targeting IKKε in androgen-independent prostate cancer causes phenotypic senescence and genomic instability. Mol Cancer Ther. 21:407–418. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Pernicová Z, Slabáková E, Kharaishvili G, Bouchal J, Král M, Kunická Z, Machala M, Kozubík A and Souček K: Androgen depletion induces senescence in prostate cancer cells through down-regulation of Skp2. Neoplasia. 13:526–536. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Blute ML, Damaschke N, Wagner J, Yang B, Gleave M, Fazli L, Shi F, Abel EJ, Downs TM, Huang W and Jarrard DF: Persistence of senescent prostate cancer cells following prolonged neoadjuvant androgen deprivation therapy. PLoS One. 12:e01720482017. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang X, Peng Y, Yuan Y, Gao Y, Hu F, Wang J, Zhu X, Feng X, Cheng Y, Wei Y, et al: Histone methyltransferase SET8 is regulated by miR-192/215 and induces oncogene-induced senescence via p53-dependent DNA damage in human gastric carcinoma cells. Cell Death Dis. 11:9372020. View Article : Google Scholar : PubMed/NCBI | |
|
Tao YP, Zhu HY, Shi QY, Wang CX, Hua YX, Hu HY, Zhou QY, Zhou ZL, Sun Y, Wang XM, et al: S1PR1 regulates ovarian cancer cell senescence through the PDK1-LATS1/2-YAP pathway. Oncogene. 42:3491–3502. 2023. View Article : Google Scholar : PubMed/NCBI | |
|
Fang L, Li D, Yin J, Pan H, Ye H, Bowman J, Capaldo B and Kelly K: TMPRSS2-ERG promotes the initiation of prostate cancer by suppressing oncogene-induced senescence. Cancer Gene Ther. 29:1463–1476. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Saleh T, Khasawneh AI, Himsawi N, Abu-Raideh J, Ejeilat V, Elshazly AM and Gewirtz DA: Senolytic therapy: A potential approach for the elimination of oncogene-induced senescent HPV-positive cells. Int J Mol Sci. 23:155122022. View Article : Google Scholar : PubMed/NCBI | |
|
Ye M, Huang X, Wu Q and Liu F: Senescent stromal cells in the tumor microenvironment: Victims or accomplices? Cancers. 15:19272023. View Article : Google Scholar : PubMed/NCBI | |
|
Brandmaier A, Hou SQ and Shen WH: Cell cycle control by PTEN. J Mol Biol. 429:2265–2277. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Zhou X, Yang X, Sun X, Xu X, Li X, Guo Y, Wang J, Li X, Yao L, Wang H and Shen L: Effect of PTEN loss on metabolic reprogramming in prostate cancer cells. Oncol Lett. 17:2856–2866. 2019.PubMed/NCBI | |
|
Parisotto M, Grelet E, El Bizri R, Dai Y, Terzic J, Eckert D, Gargowitsch L, Bornert JM and Metzger D: PTEN deletion in luminal cells of mature prostate induces replication stress and senescence in vivo. J Exp Med. 215:1749–1763. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Z, Carracedo A, Lin HK, Koutcher JA, Behrendt N, Egia A, Alimonti A, Carver BS, Gerald W, Teruya-Feldstein J, et al: Differential p53-independent outcomes of p19(Arf) loss in oncogenesis. Sci Signal. 2:ra442009. View Article : Google Scholar : PubMed/NCBI | |
|
Guo J, Huang X, Dou L, Yan M, Shen T, Tang W and Li J: Aging and aging-related diseases: From molecular mechanisms to interventions and treatments. Signal Transduct Target Ther. 7:3912022. View Article : Google Scholar : PubMed/NCBI | |
|
Hua H, Zheng C, Fan J, Li X, Xie W, Chen J and Yu C: The senescence-related signature predicts prognosis and characterization of tumor microenvironment infiltration in pancreatic cancer. BioMed Res Int. 2022:1–28. 2022. View Article : Google Scholar | |
|
Dyachkova U, Vigovskiy M, Basalova N, Efimenko A and Grigorieva O: M2-Macrophage-induced chronic inflammation promotes reversible mesenchymal stromal cell senescence and reduces their anti-fibrotic properties. Int J Mol Sci. 24:170892023. View Article : Google Scholar : PubMed/NCBI | |
|
Stanojković TP, Matić IZ, Petrović N, Stanković V, Kopčalić K, Besu I, Đorđić Crnogorac M, Mališić E, Mirjačić-Martinović K, Vuletić A, et al: Evaluation of cytokine expression and circulating immune cell subsets as potential parameters of acute radiation toxicity in prostate cancer patients. Sci Rep. 10:190022020. View Article : Google Scholar : PubMed/NCBI | |
|
González-Ochoa S, Tellez-Bañuelos MC, Méndez-Clemente AS, Bravo-Cuellar A, Hernández Flores G, Palafox-Mariscal LA, Haramati J, Pedraza-Brindis EJ, Sánchez-Reyes K and Ortiz-Lazareno PC: Combination blockade of the IL6R/STAT-3 Axis with TIGIT and its impact on the functional activity of NK cells against prostate cancer cells. J Immunol Res. 2022:18108042022. View Article : Google Scholar : PubMed/NCBI | |
|
Méndez-Clemente A, Bravo-Cuellar A, González-Ochoa S, Santiago-Mercado M, Palafox-Mariscal L, Jave-Suárez L, Solorzano-Ibarra F, Villaseñor-García M, Ortiz-Lazareno P and Hernández-Flores G: Dual STAT-3 and IL-6R inhibition with stattic and tocilizumab decreases migration, invasion and proliferation of prostate cancer cells by targeting the IL-6/IL-6R/STAT-3 axis. Oncol Rep. 48:1382022. View Article : Google Scholar : PubMed/NCBI | |
|
Silk N, Reich J, Sinha R, Chawla S, Geary K and Zhang D: The effects of resveratrol on prostate cancer through targeting the tumor microenvironment. J Xenobiotics. 11:16–32. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Park SY, Cui Z, Kim B, Park G and Choi YW: Treatment with gold nanoparticles using cudrania tricuspidata root extract induced downregulation of MMP-2/-9 and PLD1 and inhibited the invasiveness of human U87 Glioblastoma cells. Int J Mol Sci. 21:12822020. View Article : Google Scholar : PubMed/NCBI | |
|
Fahs A, Hussein N, Zalzali H, Ramadan F, Ghamloush F, Tamim H, El Homsi M, Badran B, Boulos F, Tawil A, et al: CD147 promotes tumorigenesis via Exosome-mediated signaling in rhabdomyosarcoma. Cells. 11:22672022. View Article : Google Scholar : PubMed/NCBI | |
|
Bair EL, Chen ML, McDaniel K, Sekiguchi K, Cress AE, Nagle RB and Bowden GT: Membrane type 1 Matrix Metalloprotease cleaves Laminin-10 and promotes prostate cancer cell migration. Neoplasia. 7:380–389. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Wei R, Wong JPC, Lyu P, Xi X, Tong O, Zhang SD, Yuen HF, Shirasawa S and Kwok HF: In vitro and clinical data analysis of Osteopontin as a prognostic indicator in colorectal cancer. J Cell Mol Med. 22:4097–4105. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Miftakhova R, Hedblom A, Semenas J, Robinson B, Simoulis A, Malm J, Rizvanov A, Heery DM, Mongan NP, Maitland NJ, et al: Cyclin A1 and P450 aromatase promote metastatic homing and growth of Stem-like prostate cancer cells in the bone marrow. Cancer Res. 76:2453–2464. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Guccini I, Revandkar A, D'Ambrosio M, Colucci M, Pasquini E, Mosole S, Troiani M, Brina D, Sheibani-Tezerji R, Elia AR, et al: Senescence reprogramming by TIMP1 deficiency promotes prostate cancer metastasis. Cancer Cell. 39:68–82.e9. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Rodier F, Coppé JP, Patil CK, Hoeijmakers WA, Muñoz DP, Raza SR, Freund A, Campeau E, Davalos AR and Campisi J: Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol. 11:973–979. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
van Dessel LF, van Riet J, Smits M, Zhu Y, Hamberg P, van der Heijden MS, Bergman AM, van Oort IM, de Wit R, Voest EE, et al: The genomic landscape of metastatic castration-resistant prostate cancers reveals multiple distinct genotypes with potential clinical impact. Nat Commun. 10:52512019. View Article : Google Scholar : PubMed/NCBI | |
|
Aggarwal M, Saxena R, Asif N, Sinclair E, Tan J, Cruz I, Berry D, Kallakury B, Pham Q, Wang TTY and Chung FL: p53 mutant-type in human prostate cancer cells determines the sensitivity to phenethyl isothiocyanate induced growth inhibition. J Exp Clin Cancer Res. 38:3072019. View Article : Google Scholar : PubMed/NCBI | |
|
Wanjala J, Taylor BS, Chapinski C, Hieronymus H, Wongvipat J, Chen Y, Nanjangud GJ, Schultz N, Xie Y, Liu S, et al: Identifying actionable targets through integrative analyses of GEM model and human prostate cancer genomic profiling. Mol Cancer Ther. 14:278–288. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Haffner MC, Mosbruger T, Esopi DM, Fedor H, Heaphy CM, Walker DA, Adejola N, Gürel M, Hicks J, Meeker AK, et al: Tracking the clonal origin of lethal prostate cancer. J Clin Invest. 123:4918–4922. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Ding D, Blee AM, Zhang J, Pan Y, Becker NA, Maher LJ III, Jimenez R, Wang L and Huang H: Gain-of-function mutant p53 together with ERG proto-oncogene drive prostate cancer by beta-catenin activation and pyrimidine synthesis. Nat Commun. 14:46712023. View Article : Google Scholar : PubMed/NCBI | |
|
Jiang SJ and Wang S: Dual targeting of mTORC1 and mTORC2 by INK-128 potently inhibits human prostate cancer cell growth in vitro and in vivo. Tumour Biol. 36:8177–8184. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Shorning BY, Dass MS, Smalley MJ and Pearson HB: The PI3K-AKT-mTOR pathway and prostate cancer: At the crossroads of AR, MAPK, and WNT signaling. Int J Mol Sci. 21:45072020. View Article : Google Scholar : PubMed/NCBI | |
|
Shi J, Liu C, Chen C, Guo K, Tang Z, Luo Y, Chen L, Su Y and Xu K: Circular RNA circMBOAT2 promotes prostate cancer progression via a miR-1271-5p/mTOR axis. Aging (Albany NY). 12:13255–13280. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Li Y, Fan A, Zhang Y, Guo Z, Meng W, Pan W, Ma Z and Chen W: Cellular senescence: A potential mode of circular RNAs regulating prostate cancer. MedComm-Oncol. 2:e612023. View Article : Google Scholar | |
|
Ellis L, Lehet K, Ramakrishnan S, Adelaiye R, Miles KM, Wang D, Liu S, Atadja P, Carducci MA and Pili R: Concurrent HDAC and mTORC1 inhibition attenuate androgen receptor and hypoxia signaling associated with alterations in microRNA expression. PLoS One. 6:e271782011. View Article : Google Scholar : PubMed/NCBI | |
|
Park H, Williams K, Trikalinos NA, Larson S, Tan B, Waqar S, Suresh R, Morgensztern D, Van Tine BA, Govindan R, et al: A phase I trial of temsirolimus and erlotinib in patients with refractory solid tumors. Cancer Chemother Pharmacol. 87:337–347. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Bendell JC, Kurkjian C, Infante JR, Bauer TM, Burris HA III, Greco FA, Shih KC, Thompson DS, Lane CM, Finney LH and Jones SF: A phase 1 study of the sachet formulation of the oral dual PI3K/mTOR inhibitor BEZ235 given twice daily (BID) in patients with advanced solid tumors. Invest New Drugs. 33:463–471. 2015. View Article : Google Scholar : PubMed/NCBI | |
|
Li S, Sheng J, Liu Z, Fan Y, Zhang C, Lv T, Hu S, Jin J, Yu W and Song Y: Potent antitumour of the mTORC1/2 dual inhibitor AZD2014 in docetaxel-sensitive and docetaxel-resistant castration-resistant prostate cancer cells. J Cell Mol Med. 25:2436–2449. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Jin Y, Qu S, Tesikova M, Wang L, Kristian A, Mælandsmo GM, Kong H, Zhang T, Jerónimo C, Teixeira MR, et al: Molecular circuit involving KLK4 integrates androgen and mTOR signaling in prostate cancer. Proc Natl Acad Sci USA. 110:E2572–E2581. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Pan HY and Valapala M: Regulation of autophagy by the glycogen synthase Kinase-3 (GSK-3) signaling pathway. Int J Mol Sci. 23:17092022. View Article : Google Scholar : PubMed/NCBI | |
|
Sun Y, Li Z and Song K: AR-mTOR-SRF axis regulates HMMR expression in human prostate cancer cells. Biomol Ther. 29:667–677. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Valenti MT, Mottes M, Dalle Carbonare L and Feron O: Editorial: Bone metastases. Front Oncol. 11:7415152021. View Article : Google Scholar : PubMed/NCBI | |
|
Tanaka K, Babic I, Nathanson D, Akhavan D, Guo D, Gini B, Dang J, Zhu S, Yang H, De Jesus J, et al: Oncogenic EGFR signaling activates an mTORC2-NF-κB pathway that promotes chemotherapy resistance. Cancer Discov. 1:524–538. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Wang Q, Tang Y, Yu H, Yin Q, Li M, Shi L, Zhang W, Li D and Li L: CCL18 from tumor-cells promotes epithelial ovarian cancer metastasis via mTOR signaling pathway. Mol Carcinog. 55:1688–1699. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Wei XX, Hsieh AC, Kim W, Friedlander T, Lin AM, Louttit M and Ryan CJ: A phase I study of abiraterone acetate combined with BEZ235, a dual PI3K/mTOR inhibitor, in metastatic castration resistant prostate cancer. Oncologist. 22:503–e43. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Raynard C, Ma X, Huna A, Tessier N, Massemin A, Zhu K, Flaman JM, Moulin F, Goehrig D, Medard JJ, et al: NF-κB-dependent secretome of senescent cells can trigger neuroendocrine transdifferentiation of breast cancer cells. Aging Cell. 21:e136322022. View Article : Google Scholar : PubMed/NCBI | |
|
Li N, Liu Q, Han Y, Pei S, Cheng B, Xu J, Miao X, Pan Q, Wang H, Guo J, et al: ARID1A loss induces polymorphonuclear myeloid-derived suppressor cell chemotaxis and promotes prostate cancer progression. Nat Commun. 13:72812022. View Article : Google Scholar : PubMed/NCBI | |
|
Dushyanthen S, Cossigny DAF and Quan GMY: The osteoblastic and osteoclastic interactions in spinal metastases secondary to prostate cancer. Cancer Growth Metastasis. 6:61–80. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Chen Q, Du X, Hu S and Huang Q: NF-κB-related metabolic gene signature predicts the prognosis and immunotherapy response in gastric cancer. Biomed Res Int. 2022:50925052022.PubMed/NCBI | |
|
Dewdney B, Jenkins MR, Best SA, Freytag S, Prasad K, Holst J, Endersby R and Johns TG: From signalling pathways to targeted therapies: Unravelling glioblastoma's secrets and harnessing two decades of progress. Signal Transduct Target Ther. 8:4002023. View Article : Google Scholar : PubMed/NCBI | |
|
Ayala G, Yan J, Li R, Ding Y, Thompson TC, Mims MP, Hayes TG, MacDonnell V, Lynch RG, Frolov A, et al: Bortezomib-mediated inhibition of steroid receptor coactivator-3 degradation leads to activated Akt. Clin Cancer Res. 14:7511–7518. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Nunes JJ, Pandey SK, Yadav A, Goel S and Ateeq B: Targeting NF-kappa B signaling by artesunate restores sensitivity of castrate-resistant prostate cancer cells to antiandrogens. Neoplasia. 19:333–345. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Chen H, Pang B, Zhou C, Han M, Gong J, Li Y and Jiang J: Prostate cancer-derived small extracellular vesicle proteins: The hope in diagnosis, prognosis, and therapeutics. J Nanobiotechnology. 21:4802023. View Article : Google Scholar : PubMed/NCBI | |
|
Rickard BP, Overchuk M, Chappell VA, Kemal Ruhi M, Sinawang PD, Nguyen Hoang TT, Akin D, Demirci U, Franco W, Fenton SE, et al: Methods to evaluate changes in mitochondrial structure and function in cancer. Cancers (Basel). 15:25642023. View Article : Google Scholar : PubMed/NCBI | |
|
Gong L, Chen B, Zhang J, Sun Y, Yuan J, Niu X, Hu G, Chen Y, Xie Z, Deng Z, et al: Human ESC-sEVs alleviate age-related bone loss by rejuvenating senescent bone marrow-derived mesenchymal stem cells. J Extracell Vesicles. 9:18009712020. View Article : Google Scholar : PubMed/NCBI | |
|
Martinez-Vidal L, Murdica V, Venegoni C, Pederzoli F, Bandini M, Necchi A, Salonia A and Alfano M: Causal contributors to tissue stiffness and clinical relevance in urology. Commun Biol. 4:10112021. View Article : Google Scholar : PubMed/NCBI | |
|
Ma Q, Liang M, Wu Y, Dou C, Xu J, Dong S and Luo F: Small extracellular vesicles deliver osteolytic effectors and mediate cancer-induced osteolysis in bone metastatic niche. J Extracell Vesicles. 10:e120682021. View Article : Google Scholar : PubMed/NCBI | |
|
Mongelli A, Atlante S, Barbi V, Bachetti T, Martelli F, Farsetti A and Gaetano C: Treating senescence like cancer: Novel perspectives in senotherapy of chronic diseases. Int J Mol Sci. 21:79842020. View Article : Google Scholar : PubMed/NCBI | |
|
Gazzillo A, Volponi C, Soldani C, Polidoro MA, Franceschini B, Lleo A, Bonavita E and Donadon M: Cellular senescence in liver cancer: How dying cells become ‘Zombie’ enemies. Biomedicines. 12:262023. View Article : Google Scholar : PubMed/NCBI | |
|
Du D, Tang X, Li Y, Gao Y, Chen R, Chen Q, Wen J, Wu T, Zhang Y, Lu H, et al: Senotherapy protects against Cisplatin-induced ovarian injury by removing senescent cells and alleviating DNA damage. Oxid Med Cell Longev. 2022:91446442022. View Article : Google Scholar : PubMed/NCBI | |
|
Gasek NS, Kuchel GA, Kirkland JL and Xu M: Strategies for targeting senescent cells in human disease. Nat Aging. 1:870–879. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Liu Y, Zhang Q, Ni W, Ji G and Xu H: A strategy for the treatment of gastrointestinal cancer: Targeting tumor senescent cells. Front Mol Biosci. 10:11398402023. View Article : Google Scholar : PubMed/NCBI | |
|
Ramírez R, Ceprian N, Figuer A, Valera G, Bodega G, Alique M and Carracedo J: Endothelial senescence and the chronic vascular diseases: Challenges and therapeutic opportunities in atherosclerosis. J Pers Med. 12:2152022. View Article : Google Scholar : PubMed/NCBI | |
|
Fedorov VD, Themeli M and Sadelain M: PD-1- and CTLA-4-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med. 5:215ra1722013. View Article : Google Scholar : PubMed/NCBI | |
|
Arai S, Varkaris A, Nouri M, Chen S, Xie L and Balk SP: MARCH5 mediates NOXA-dependent MCL1 degradation driven by kinase inhibitors and integrated stress response activation. eLife. 9:e549542020. View Article : Google Scholar : PubMed/NCBI | |
|
Arai S, Jonas O, Whitman MA, Corey E, Balk SP and Chen S: Tyrosine kinase inhibitors increase MCL1 degradation and in combination with BCLXL/BCL2 inhibitors drive prostate cancer apoptosis. Clin Cancer Res. 24:5458–5470. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Ferraldeschi R, Welti J, Powers MV, Yuan W, Smyth T, Seed G, Riisnaes R, Hedayat S, Wang H, Crespo M, et al: Second-generation HSP90 inhibitor Onalespib blocks mRNA splicing of androgen receptor variant 7 in prostate cancer cells. Cancer Res. 76:2731–2742. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Slovin S, Hussain S, Saad F, Garcia J, Picus J, Ferraldeschi R, Crespo M, Flohr P, Riisnaes R, Lin C, et al: Pharmacodynamic and clinical results from a phase I/II study of the HSP90 Inhibitor Onalespib in combination with abiraterone acetate in prostate cancer. Clin Cancer Res. 25:4624–4633. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Lu X, Yang F, Chen D, Zhao Q, Chen D, Ping H and Xing N: Quercetin reverses docetaxel resistance in prostate cancer via androgen receptor and PI3K/Akt signaling pathways. Int J Biol Sci. 16:1121–1134. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Ward AB, Mir H, Kapur N, Gales DN, Carriere PP and Singh S: Quercetin inhibits prostate cancer by attenuating cell survival and inhibiting anti-apoptotic pathways. World J Surg Oncol. 16:1082018. View Article : Google Scholar : PubMed/NCBI | |
|
Pratheeshkumar P, Budhraja A, Son YO, Wang X, Zhang Z, Ding S, Wang L, Hitron A, Lee JC, Xu M, et al: Quercetin inhibits angiogenesis mediated human prostate tumor growth by targeting VEGFR-2 regulated AKT/mTOR/P70S6K signaling pathways. PLoS One. 7:e475162012. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang DF, Yang ZC, Chen JQ, Jin XX, Qiu YD, Chen XJ, Shi HY, Liu ZG, Wang MS, Liang G and Zheng XH: Piperlongumine inhibits migration and proliferation of castration-resistant prostate cancer cells via triggering persistent DNA damage. BMC Complement Med Ther. 21:1952021. View Article : Google Scholar : PubMed/NCBI | |
|
Makhov P, Golovine K, Teper E, Kutikov A, Mehrazin R, Corcoran A, Tulin A, Uzzo RG and Kolenko VM: Piperlongumine promotes autophagy via inhibition of Akt/mTOR signalling and mediates cancer cell death. Br J Cancer. 110:899–907. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Golovine KV, Makhov PB, Teper E, Kutikov A, Canter D, Uzzo RG and Kolenko VM: Piperlongumine induces rapid depletion of the androgen receptor in human prostate cancer cells. Prostate. 73:23–30. 2013. View Article : Google Scholar : PubMed/NCBI | |
|
Liu G, Jin Z and Lu X: Differential targeting of Gr-MDSCs, T cells and prostate cancer cells by dactolisib and dasatinib. Int J Mol Sci. 21:23372020. View Article : Google Scholar : PubMed/NCBI | |
|
Araujo JC, Poblenz A, Corn P, Parikh NU, Starbuck MW, Thompson JT, Lee F, Logothetis CJ and Darnay BG: Dasatinib inhibits both osteoclast activation and prostate cancer PC-3-cell-induced osteoclast formation. Cancer Biol Ther. 8:2153–2159. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Cuyàs E, Verdura S, Llorach-Pares L, Fernández-Arroyo S, Luciano-Mateo F, Cabré N, Stursa J, Werner L, Martin-Castillo B, Viollet B, et al: Metformin directly targets the H3K27me3 demethylase KDM6A/UTX. Aging Cell. 17:e127722018. View Article : Google Scholar : PubMed/NCBI | |
|
Hua Y, Zheng Y, Yao Y, Jia R, Ge S and Zhuang A: Metformin and cancer hallmarks: Shedding new lights on therapeutic repurposing. J Transl Med. 21:4032023. View Article : Google Scholar : PubMed/NCBI | |
|
Wang ZS, Huang HR, Zhang LY, Kim S, He Y, Li DL, Farischon C, Zhang K, Zheng X, Du ZY and Goodin S: Mechanistic study of inhibitory effects of metformin and atorvastatin in combination on prostate cancer cells in vitro and in vivo. Biol Pharm Bull. 40:1247–1254. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Wang J, Lu Y, Wang J, Koch AE, Zhang J and Taichman RS: Retraction: CXCR6 Induces prostate cancer progression by the AKT/mammalian target of rapamycin signaling pathway. Cancer Res. 82:3406. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Zhang J, Wu D, He Y, Li L, Liu S, Lu J, Gui H, Wang Y, Tao Y, Wang H, et al: Rapamycin inhibits AR signaling pathway in prostate cancer by interacting with the FK1 domain of FKBP51. Biochem Biophys Rep. 23:1007782020.PubMed/NCBI | |
|
Shorning BY, Dass MS, Smalley MJ and Pearson HB: The PI3K-AKT-mTOR pathway and prostate cancer: At the crossroads of AR, MAPK, and WNT signaling. Int J Mol Sci. 21:45072020. View Article : Google Scholar : PubMed/NCBI | |
|
Lo U, Chen Y, Cen J, Deng S, Luo J, Zhau H, Ho L, Lai CH, Mu P, Chung LWK and Hsieh JT: The driver role of JAK-STAT signalling in cancer stemness capabilities leading to new therapeutic strategies for therapy- and castration-resistant prostate cancer. Clin Transl Med. 12:e9782022. View Article : Google Scholar : PubMed/NCBI | |
|
Sheth S, Jajoo S, Kaur T, Mukherjea D, Sheehan K, Rybak LP and Ramkumar V: Resveratrol reduces prostate cancer growth and metastasis by inhibiting the Akt/MicroRNA-21 pathway. PLoS One. 7:e516552012. View Article : Google Scholar : PubMed/NCBI | |
|
Fenner A: Prostate cancer: Resveratrol inhibits the AR. Nat Rev Urol. 14:642. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Hickson LJ, Langhi Prata LGP, Bobart SA, Evans TK, Giorgadze N, Hashmi SK, Herrmann SM, Jensen MD, Jia Q, Jordan KL, et al: Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 47:446–456. 2019. View Article : Google Scholar : PubMed/NCBI | |
|
Di Micco R, Krizhanovsky V, Baker D and d'Adda di Fagagna F: Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 22:75–95. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Kaur G, Sundar IK and Rahman I: p16-3MR: A novel model to study cellular senescence in cigarette smoke-induced lung injuries. Int J Mol Sci. 22:48342021. View Article : Google Scholar : PubMed/NCBI | |
|
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, Kirkland JL and van Deursen JM: Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 479:232–236. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Song P, Duan JL, Ding J, Liu JJ, Fang ZQ, Xu H, Li ZW, Du W, Xu M, Ling YW, et al: Cellular senescence primes liver fibrosis regression through Notch-EZH2. MedComm (2020). 4:e3462023.PubMed/NCBI | |
|
Chen M, Wu G, Lu Y, Sun S, Yu Z, Pan X, Chen W, Xu H, Qiu H, He W, et al: A p21-ATD mouse model for monitoring and eliminating senescent cells and its application in liver regeneration post injury. Mol Ther. Apr 6–2024.(Epub ahead of print). View Article : Google Scholar | |
|
Zhan D, Ma D, Wei S, Lal B, Fu Y, Eberhart C, Laterra J, Ying M, Li Y, Meeker A, et al: Monoallelic IDH1 R132H mutation mediates glioma cell response to anticancer therapies via induction of senescence. Mol Cancer Res. 19:1878–1888. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Chen WC, Chang TC, Chou HH, Cheng MH, Hong JJ, Hsieh YS and Cheng CM: Peritoneal fluid analysis of advanced ovarian cancers after hyperthermic intraperitoneal chemotherapy. Int J Mol Sci. 24:97482023. View Article : Google Scholar : PubMed/NCBI | |
|
D'Aguanno S and Del Bufalo D: Inhibition of Anti-apoptotic Bcl-2 proteins in preclinical and clinical studies: Current overview in cancer. Cells. 9:12872020. View Article : Google Scholar : PubMed/NCBI | |
|
Harrison CN, Garcia JS, Somervaille TCP, Foran JM, Verstovsek S, Jamieson C, Mesa R, Ritchie EK, Tantravahi SK, Vachhani P, et al: Addition of Navitoclax to ongoing Ruxolitinib therapy for patients with myelofibrosis with progression or suboptimal response: Phase II safety and efficacy. J Clin Oncol. 40:1671–1680. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
He Y, Zhang X, Chang J, Kim HN, Zhang P, Wang Y, Khan S, Liu X, Zhang X, Lv D, et al: Using proteolysis-targeting chimera technology to reduce navitoclax platelet toxicity and improve its senolytic activity. Nat Commun. 11:19962020. View Article : Google Scholar : PubMed/NCBI | |
|
Ferraldeschi R, Welti J, Powers MV, Yuan W, Smyth T, Seed G, Riisnaes R, Hedayat S, Wang H, Crespo M, et al: Second-generation HSP90 inhibitor Onalespib blocks mRNA splicing of androgen receptor variant 7 in prostate cancer cells. Cancer Res. 76:2731–2742. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Colucci M, Zumerle S, Bressan S, Gianfanti F, Troiani M, Valdata A, D'Ambrosio M, Pasquini E, Varesi A, Cogo F, et al: Retinoic acid receptor activation reprograms senescence response and enhances anti-tumor activity of natural killer cells. Cancer Cell. 42:646–661.e9. 2024. View Article : Google Scholar : PubMed/NCBI | |
|
Chi KN, Gleave ME, Klasa R, Murray N, Bryce C, Lopes de Menezes DE, D'Aloisio S and Tolcher AW: A phase I dose-finding study of combined treatment with an antisense Bcl-2 oligonucleotide (Genasense) and mitoxantrone in patients with metastatic hormone-refractory prostate cancer. Clin Cancer Res. 7:3920–3927. 2001.PubMed/NCBI | |
|
Tolcher AW, Kuhn J, Schwartz G, Patnaik A, Hammond LA, Thompson I, Fingert H, Bushnell D, Malik S, Kreisberg J, et al: A Phase I pharmacokinetic and biological correlative study of oblimersen sodium (genasense, g3139), an antisense oligonucleotide to the bcl-2 mRNA, and of docetaxel in patients with hormone-refractory prostate cancer. Clin Cancer Res. 10:5048–5057. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Stein MN, Goodin S, Gounder M, Gibbon D, Moss R, Portal D, Lindquist D, Zhao Y, Takebe N, Tan A, et al: A phase I study of AT-101, a BH3 mimetic, in combination with paclitaxel and carboplatin in solid tumors. Invest New Drugs. 38:855–865. 2020. View Article : Google Scholar : PubMed/NCBI | |
|
Osada T, Crosby EJ, Kaneko K, Snyder JC, Ginzel JD, Acharya CR, Yang XY, Polascik TJ, Spasojevic I, Nelson RC, et al: HSP90-specific nIR Probe identifies aggressive prostate cancers: Translation from preclinical models to a human phase I study. Mol Cancer Ther. 21:217–226. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Suh GA, Lodise TP, Tamma PD, Knisely JM, Alexander J, Aslam S, Barton KD, Bizzell E, Totten KMC, Campbell JL, et al: Considerations for the use of phage therapy in clinical practice. Antimicrob Agents Chemother. 66:e02071212022. View Article : Google Scholar : PubMed/NCBI | |
|
Kolodkin-Gal D, Roitman L, Ovadya Y, Azazmeh N, Assouline B, Schlesinger Y, Kalifa R, Horwitz S, Khalatnik Y, Hochner-Ger A, et al: Senolytic elimination of Cox2-expressing senescent cells inhibits the growth of premalignant pancreatic lesions. Gut. 71:345–355. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Spetsieris N, Boukovala M, Weldon JA, Tsikkinis A, Hoang A, Aparicio A, Tu SM, Araujo JC, Zurita AJ, Corn PG, et al: A Phase 2 trial of abiraterone followed by randomization to addition of dasatinib or sunitinib in men with metastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 19:22–31.e5. 2021. View Article : Google Scholar : PubMed/NCBI | |
|
Rossi M, Anerillas C, Idda ML, Munk R, Shin CH, Donega S, Tsitsipatis D, Herman AB, Martindale JL, Yang X, et al: Pleiotropic effects of BAFF on the senescence-associated secretome and growth arrest. Elife. 12:e842382023. View Article : Google Scholar : PubMed/NCBI | |
|
Chaib S, Tchkonia T and Kirkland JL: Cellular senescence and senolytics: The path to the clinic. Nat Med. 28:1556–1568. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Kirkland JL, Tchkonia T, Zhu Y, Niedernhofer LJ and Robbins PD: The clinical potential of senolytic drugs. J Am Geriatr Soc. 65:2297–2301. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Ji S, Xiong M, Chen H, Liu Y, Zhou L, Hong Y, Wang M, Wang C, Fu X and Sun X: Cellular rejuvenation: Molecular mechanisms and potential therapeutic interventions for diseases. Signal Transduct Target Ther. 8:1162023. View Article : Google Scholar : PubMed/NCBI | |
|
Kreienkamp R, Graziano S, Coll-Bonfill N, Bedia-Diaz G, Cybulla E, Vindigni A, Dorsett D, Kubben N, Batista LFZ and Gonzalo S: A cell-intrinsic interferon-like response links replication stress to cellular aging caused by progerin. Cell Rep. 22:2006–2015. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Krtolica A, Parrinello S, Lockett S, Desprez PY and Campisi J: Senescent fibroblasts promote epithelial cell growth and tumorigenesis: A link between cancer and aging. Proc Natl Acad Sci USA. 98:12072–12077. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Wang L, Jin H, Jochems F, Wang S, Lieftink C, Martinez IM, De Conti G, Edwards F, de Oliveira RL, Schepers A, et al: cFLIP suppression and DR5 activation sensitize senescent cancer cells to senolysis. Nat Cancer. 3:1284–1299. 2022. View Article : Google Scholar : PubMed/NCBI |